Abstract
Divergent evolution leads to variation among populations and thus promotes diversification. In plants, adaptation to different soils, pollinator guilds, and herbivores is thought to be a key ecological driver of adaptive divergence, but few studies have investigated this process experimentally. Here we use experimental evolution with fast cycling Brassica rapa plants to study the impact of soil, pollination, herbivory, and their interactions on divergent evolution in various traits during eight generations of selection. We found significant evolutionary changes in plant phenotypes caused by all three factors and their interactions. In the richer soil type, plants showed higher evolutionary rates, especially with bumblebee-pollination, which led to the evolution of increased attractiveness of plants to bumblebees. Plants that had experienced aphid-herbivory showed lower attractiveness. We found the strongest evolutionary divergence when plants evolved in different soils with bee-pollination rather than hand-pollination, irrespective of herbivory. This “soil-pollinator effect” impacted divergence in diverse suites of traits, for example leaf size, flowering time, flower petal length, some floral volatiles and leaf glucosinolates. We conclude that the interaction between soil and biotic pollination may be an important cause for divergent evolution of plants growing on different soil types, even without a shift in pollinator guilds.
Similar content being viewed by others
Introduction
The first step in plant speciation is the establishment of genotypic and phenotypic differences among populations, sometimes called plant races1. Although both neutral (i.e. genetic drift) and adaptive evolutionary processes can cause such variations, adaptation driven by selection is thought to be of prime importance for divergent evolution2. Factors that cause adaptation to local conditions include abiotic factors such as climate3, mixed abiotic-biotic factors like soil and soil-born microorganisms4, as well as biotic interactions such as those between plants, herbivores, and pollinators5,6,7. Whereas shifts in physiological-, root- and overall growth-related traits are thought to be driven primarily by soil adaptation8, divergence in defense mechanisms is expected to be selected for by variable herbivore communities9, and floral divergence is thought to be driven by shifts in pollinator species or pollinator guild composition10,11.
Divergent evolution in response to soil-type is common in plants and is well documented12. Plant adaptations to soil may be driven by physical and/or chemical parameters such as water-holding capacity, nutrient content13,14, salinity15,16, calcium ions in calcareous soil14,17, or heavy metal content in serpentine-18 and other metalliferous soils19,20. Soil represents the major source of nutrients for plants21,22, and selection favors mechanisms that improve resource acquisition and/or utilization23. Adaptation to physical properties typically causes alterations of the root system, and re-allocations between above- and below ground biomass24. On the other hand, physiological mechanisms to cope with chemical properties like the exclusion or tolerance of high concentration of toxic compounds in plant tissue have also been demonstrated12,25.
Plants in their natural environment are involved in multiple interspecific biotic interactions, for example with animals that pollinate flowers and/or feed on plant tissue26,27. By feeding on plants, herbivores induce strong selection on defense mechanisms in plants that may differ depending on the feeding mode and sensitivity of herbivore taxa to defense compounds9,28,29. Pollinators, in contrast, act mutualistically and select for signals and morphological traits ensuring optimal pollen transfer26,30. As pollinators differ in both their sensory preferences and morphological features, selection imposed by different pollinators can lead to divergent evolution among plant populations/species31,32.
Multiple lines of evidence show that different ecological factors such as soil-type and plant-animal interactions influence each other at various levels. First, soil-type strongly impacts the morphology and physiology of plants through phenotypic plasticity, and thereby changes the plants’ interactions with pollinators and herbivores33. For example, soil nutrients have been shown to impact plant phenology, flower production, flower shape, nectar production, as well as nectar and pollen chemistry34,35,36, all with high relevance for pollinator behavior. Plants growing on serpentine soils have been shown to receive less pollinator visitations and less herbivory37, whereas fertile soils can increase plant attractiveness to herbivores38. Hyperaccumulation of toxic metals is known to negatively affect herbivores and may also have an impact on pollinators and plant-fertility related traits39,40,41. Such environmentally-induced changes in plant traits likely change patterns of phenotypic selection caused by the behavior of pollinators and herbivores33,42, leading to evolutionary changes driven by interactions between soil and animal interactors.
In addition, although different ecological factors may typically select for different plant traits, the evolution of those traits is often not independent because of ecological and/or genetic mechanisms43,44,45,46, potentially resulting in trade-offs for plants47. For instance, soil chemistry can modify adaptive responses to both plant-herbivore and plant-pollinator interactions through resource limitation. Soil fertility can determine the evolution of plant growth and defense as predicted by the resource availability hypothesis48,49. In poor growing conditions, plants are expected to evolve low growth-rates but high levels of constitutive defense, a strategy driven by the lack of resources necessary to compensate for herbivore damage or produce effective herbivore-induced defenses48,50,51,52,53. In contrast, where resource availability is higher, plants may invest less in defense but evolve an ability to compensate for or tolerate herbivore damage54,55,56. In addition, traits that evolve in response to herbivores are intricately linked with those that evolve under selection by pollinators and vice versa57. For example, evolution of defense or herbivore-induced plasticity can compromise plant attractiveness to pollinators by decreasing the attractiveness of floral signals58,59, and rewards60,61, thereby causing a trade-off between pollinator attractiveness and defense against herbivores.
Finally, patterns of macroevolutionary diversification suggest combined effects of soil-type and pollinators on plant speciation in some plant lineages and floristic provinces8. Evidence for this is found in closely related plant species being adapted to different pollinators as well as to different soil types62,63,64,65. The evolution of such combined adaptions can be explained by adaptation to one factor, e.g. soil-type, driving the subsequent adaptation to the other (e.g. pollinator), either because pollinator distribution co-vary with soil-type in a mosaic-like fashion (i.e. the Grant-Stebbins model of pollinator driven speciation6,10), or soil-adaptation reinforces differential pollinator attraction upon secondary contact63. Both scenarios assume a shift in pollinator guilds as the primary force for divergence in flower-types and the subsequent evolution of pollination ecotypes32,66. Other studies, however, suggest that pollination ecotypes can also arise as a byproduct of habitat adaptation, such as to elevation ranges67,68,69. Generally, however, we know little about the combined effects of various ecological factors, such as soil, pollinators and/or herbivores in adaptive diversification, and their importance in promoting divergent evolution at the onset of the diversification process70.
A powerful way to study the process of diversification is experimental evolution71, which has, however, up to now only rarely been applied to plants, and most studies that do, focus on the effects of only one ecological factor31,61,72,73,74. Here we aim to contribute to filling this gap by disentangling the effects of soil, bee-pollination, and aphid-herbivory in shaping divergent evolution, by performing an eight-generation evolution experiment with fast cycling Brassica rapa plants as an outcrossing model with short generation time and high standing genetic variation75,76. In order to apply as-natural-as-possible ecological parameters in the experiment, we mimicked natural conditions found in populations of a Southern Italian Brassica species, Brassica incana, that grows on limestone- or tuff soil, the latter with higher nutrient availability than the former, with bumblebees and aphids being major biotic interactors. Nevertheless, the objective of the study was not to investigate evolutionary patterns specific to any plant species, but to provide overall insights and create new testable hypotheses about how soil and animal interactors impact plant divergent evolution. We used a 3-way factorial design with two different soil types (limestone- and tuff soil) that were collected in natural populations of Brassica incana, with- and without aphid-herbivory, and with either bumblebee-pollination or hand-pollination (Fig. 1). Hand pollination was included as a control for pollinator-mediated selection, as it represents random pollination without pollinator-mediated selection. Each treatment comprised two replicates with 49 plants each, evolving independently during eight consecutive generations; to reduce maternal effects, two generations without insects were included before plants were phenotyped. The soil used in the experiment was not sterilized, because sterilized soil can hardly be kept sterile during the experiment, with unnatural re-establishment of microbes being the possible consequence. We assessed the evolutionary responses of plants after the experiment by phenotyping plants in a common garden environment with standardized soil. We measured morphological as well as chemical traits, namely flower volatiles collected by headspace sorption and analyzed by gas chromatography, and leaf glucosinolates analyzed by liquid chromatography. These different traits are traditionally thought to evolve in response to different ecological factors such as pollinators (floral scent, flower morphology), herbivores (defense chemistry, i.e. glucosinolates), and soil (vegetative traits such as leaves). We performed various statistical analyzes, such as principle component analysis, linear discriminant function analysis and general linear models, and calculated evolutionary rates in haldanes. We addressed the following questions: (1) How do plant morphology, nectar, floral scent, defense traits, and attractiveness to pollinators evolve in response to different soil types, pollination and herbivory treatments? (2) Do interactions between these factors lead to distinct evolutionary trajectories and which combinations lead to the strongest divergence? For (1) we predicted that low nutrient soil should limit evolutionary response to bee-pollination and aphid-herbivory, as resource limitation is expected to induce strong trade-offs between traits. For (2) we predicted that because the ecological factors likely selected for different traits, more factors should cause more divergence77, and thus the interaction of both herbivory and pollination lead to the strongest divergence among plant populations on different soil types.
Our results show rapid divergent evolution in many traits driven by soil, pollination, herbivory and their interactions in our model system. We document that divergent evolution in response to different soil types was more pronounced with bee-pollination rather than hand pollination. This shows that pollination, the biotic factor with the strongest fitness-impact in our experiment, interacted with soil differences leading to the evolution of different plant phenotypes without a switch in pollinator quantity or quality. We conclude that divergent evolution driven by different soils can be accelerated by biotic pollination, which may act as a starting point for soil-pollinator driven diversification in plants.
98 full-sib seed families of fast cycling Brassica rapa plants were divided into two replicates (A and B), and spread among treatment groups, with each seed family being represented in each treatment (49 plants were used per treatment per replicate). Differently colored squares represent the two types of soil, tuff- (pale green) and limestone soil (pale blue).
Results
Throughout this Result section, unless explicitly stated otherwise, only statistically significant findings are reported (P < 0.05).
Divergence in individual traits
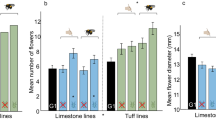
Soil: The morphology of plants that evolved in tuff during the experiment diverged from plants evolving in limestone soil, with increases in nectar production (1.30-fold), leaf size (1.23-fold), flower number (1.09-fold), plant size (1.08-fold), and flowering time (1.04-fold; Table 1, Supplementary Table 7, Supplementary Data 1). In terms of floral scent, plants that evolved in tuff exhibited distinctive emission profiles compared to those that evolved in limestone, with increases in the emission of benzaldehyde (1.21-fold) and (E,E)-α-farnesene (1.13-fold), but decreases in the emission of 2-aminobenzaldehyde (0.83-fold), benzyl nitrile (0.76-fold), phenylethyl alcohol (0.62-fold), phenylacetaldehyde (0.48-fold), and methyl anthranilate (0.38-fold; Table 1, Supplementary Table 7). Plants that evolved in tuff exhibited a higher leaf glucosinolates production (1.12-fold) than those that evolved in limestone. This change was particularly pronounced for glucobrassicanapin (1.38-fold), gluconapin (1.15-fold), and glucoasturtiin (1.15-fold; Fig. 2, Table 1, Supplementary Table 7, Supplementary Data 1).
Evolution of phenology and morphology (a: time to flower (N = 647 plants), b: leaf size (N = 647 plants), c: flower diameter (N = 636 plants)), floral scent (d: benzyl nitrile emission (N = 635)), leaf glucosinolates (e: methoxyglucobrassicin (N = 576 plants)), and plant attractiveness (f: plant attractiveness to bumblebees (N = 213 bees for 639 plants)) in plants of generation 10 (i.e. after 8 generations of selection) in different soil types and plants of generation 1 (G1, i.e. before selection). The effects of ecological factors (soil, pollination, herbivory) on evolutionary changes were estimated by two-sided linear mixed models (LMM). Asterisks indicate significant effects of the factors soil, herbivory, or pollination (P < 0.05; detailed statistical values are shown in Supplementary Table 6). Bars show mean trait values (±s.e.m.) among the different treatments.
Pollination: Bee-pollinated plants evolved 1.07-fold bigger flowers, with changes in floral scent observed in benzyl nitrile (1.23-fold) and in phenylethyl alcohol (0.79-fold), as well as lower amounts of leaf glucosinolates (0.92-fold), such as gluconapin (0.91-fold), glucoraphanin (0.78-fold), glucobrassicin (0.78-fold), and gluconasturtiin (0.64-fold; Fig. 2, Supplementary Tables 6, 7, Supplementary Data 1).
Herbivory: Plants experiencing aphid-herbivory evolved smaller petal width (0.96-fold) and sepal length (0.97-fold). Plants that experienced aphid-herbivory also evolved distinctive floral scent emission with variations in indole (1.20-fold), 1-butene-4-isothiocyanate (0.79-fold), and phenylethyl alcohol (0.78-fold; Supplementary Tables 6, 7, Supplementary Data 1).
Pollination × soil: Plants evolving in limestone soil with bee-pollination evolved smaller plant size (0.91-fold), lower flower production rate (0.90-fold) with lower nectar amount (0.82-fold), and smaller leaves (0.89-fold) but with higher amounts of leaf methoxyglucobrassicin (1.17-fold). In contrast, plants that evolved in tuff soil with bee-pollination evolved to stand taller (1.05-fold), with higher flower production rate (1.05-fold), and bigger leaves (1.16-fold) but with lower leaf methoxyglucobrassicin (0.57-fold; Fig. 2, Supplementary Tables 6–8, Supplementary Data 1). In addition, plants in limestone soil with bee-pollination evolved lower 2-aminobenzaldehyde emission (0.88-fold), but higher leaf neoglucobrassicin production (1.66-fold) than plants in tuff soil with bee-pollination (Table 1, Supplementary Table 7, Supplementary Data 1). In plants that evolved in tuff soil with bee-pollination, we observed a later flowering time (1.10-fold) compared to plants in limestone soil with bee-pollination, as well as an increased emission in methyl salicylate (1.25-fold), and in (E,E)-α-farnesene (1.24-fold), whereas leaf glucosinolates were characterized by lower amounts in hydroxyglucobrassicin (0.81-fold) and methoxyglucobrassicin (0.49-fold; Supplementary Tables 7, 8, Supplementary Data 1).
Herbivory × soil: Among the two soil evolutionary lines, aphid-herbivory had a very different effect on plant evolution (Supplementary Table 8, Supplementary Data 1). In plants evolving in limestone soil, herbivory mainly affected the plants’ floral scent emission with variations in methyl anthranilate (1.89-fold), 2-aminobenzaldehyde (1.31-fold), (E,E)-α-farnesene (0.72-fold), phenylethyl alcohol (0.69-fold), benzaldehyde (0.64-fold), and 1-butene-4-isothiocyanate (0.63-fold; Supplementary Table 8, Supplementary Data 1). In addition, we observed a 1.14-fold change in plant branching and 0.35-fold lower leaf glucoalyssin amount (Supplementary Table 8, Supplementary Data 1). When plants evolved in tuff soil, aphid-herbivory affected mostly the evolution of plant morphology with 0.88-fold smaller leaf, reduced plant height (0.91-fold), and reduced flower production rate (0.87-fold), but increases in p-anisaldehyde (1.52-fold), and benzaldehyde emissions (1.38-fold, Supplementary Table 8, Supplementary Data 1).
Pollination × herbivory × soil: Plants that evolved in limestone soil with bee-pollination and aphid-herbivory increased their production in 4 aromatic compounds (2.96, 1.98, 1.43, 1.13 for methyl anthranilate, methyl benzoate, methyl salicylate, phenylacetaldehyde, respectively) and produced 1.38-fold more leaf neoglucobrassicin than plants that evolved in absence of herbivory but with bee-pollination (Supplementary Tables 6, 8, 9, Supplementary Data 1). In contrast, plants that evolved in tuff soil with aphid-herbivory and bee-pollination were smaller (0.88-fold), with flowers emitting 0.34-fold lower (Z)−3-hexen-1-ol-acetate than bee-pollinated plants that evolved without herbivory (Supplementary Tables 6–9, Supplementary Data 1).
Overall patterns of divergence between soil types
To study the phenotypic differences between soil types, we analyzed F-values of univariate ANOVAs, and multivariate linear discriminant function analyses (DFA). The F-values were calculated for 33 traits of plants that evolved in different soil types, with larger F-values indicating stronger differentiation. The analyses showed that the factor “pollination” led to higher F-values between soil types in plant morphological traits only (Fig. 3; Table 2, Supplementary Data 2). In support of this, using multivariate DFA with 12 principal components summarizing all plant traits, we showed that the divergence between soil types was stronger when plants had evolved with bee-pollination as compared to hand-pollination (Fig. 4, Supplementary Data 3).
The graph shows the effect of pollination (bee-pollination or hand-pollination) and herbivory (aphid-herbivory or no herbivory) on phenotypic differences across different trait categories (plant morphology (black bars), floral scent (dark grey bars), and leaf glucosinolates (light grey bars)), between plants that evolved in different soil types. Differences were measured by F-values of univariate ANOVAs (N = 264): F-values were calculated between treatment groups with the same biotic interactions, growing on different soil types. Thus, greater F-values indicate more differentiation between traits of plants that evolved in different soil types (see Table 2 for statistical details). Bars show mean F-values (±s.e.m.) for the three trait categories.
The figure shows multivariate comparisons of plants before and after experimental evolution (plants of generation 1 (initial population) and 10), with a plants that evolved with bee-pollination (N = 221) and b plants that evolved with hand-pollination (N = 219), using linear discriminant function analyses (DFA). Blue dots: plants that evolved in limestone, green dots: plants that evolved in tuff; circles: plants that evolved without herbivory, triangles: plants that evolved with aphid-herbivory, black squares: plants of generation 1; filled, enlarged symbols are group centroids of replicates. In bee-pollinated treatment groups, all comparisons between soil types were significantly different, whereas in the hand-pollinated treatment groups, only plants that evolved in tuff without herbivores were significantly different from limestone lines (see post hoc comparisons between evolutionary lines in Supplementary Table 4). We used replicates of all five treatment groups as factor in the DFA leading to 10 groups for the hand-pollination and bee-pollination group, and 9 discriminant functions. (N = 283 for bee-pollinated plants and generation one: functions: 1–9 χ2 = 445.00, 2–9: 241.46, 3–9: 166.53, 4–9: 103.63, all P < 0.001, 5–9: 50.95, 6–9: 29.81, 7–9: 13.39, 8–9: 5.14, 9: 0.58 with P > 0.05, N = 281 for hand-pollinated plants and generation one: functions: 1–9 χ2 = 327.12, 2–9: 237.02, 3–9: 148.10, 4–9: 42.90 all P < 0.001, 5–9: 48.23, 6–9: 30.22, 7–9: 16.39, 8–9: 8.98, 9: 2.50 P > 0.05).
Evolution of plant attractiveness
Because we documented strong evolutionary changes in plant traits driven by soil, pollinators, and herbivory, we investigated how these changes affected plant attractiveness to pollinators and herbivores, by performing choice tests with plants of each treatment group and generation one (i.e. 9 plants were assayed together). In terms of the plants’ attractiveness to bees, we found that plants that evolved in tuff soil with bee-pollination without herbivory were more attractive than plants of all other treatment groups (Fig. 2, Supplementary Data 1). There was no statistical difference in attractiveness between the other plant groups. We did not find significant variation in plant attractiveness to alate aphids among our treatment groups.
Evolutionary rates
Plants that evolved with bee-pollination and in tuff soil showed the highest evolutionary rates, with haldanes (=s.d. per generation) being on average 1.36-fold higher than in plants evolving with hand-pollination or in limestone soil (Fig. 5, Supplementary Tables 2, 3, Supplementary Data 4).
The graphs show the impact of pollination (bee-pollination or hand-pollination), herbivory (aphid-herbivory or no herbivory), and soil (tuff or limestone) on evolutionary changes between generation one and ten (i.e. evolutionary rate), measured in absolute values of haldanes (=s.d. per generation). a overall phenotypic change, b plant morphology, c floral scent and d leaf glucosinolates. Blue bars: plants that evolved in limestone soil, green bars: plants that evolved in tuff soil. Note that bars represent absolute evolutionary changes (mean ±s.e.m.) and indicate evolutionary rates of different traits, so similar bar height does not necessarily indicate evolutionary changes in the same direction or similar evolutionary rates in the same traits. Asterisks indicate significant difference between the factors pollination, herbivory and soil (P < 0.05, see Supplementary Tables 2 and 3 for statistical details).
Discussion
The identification of mechanisms driving among-population divergence in plant traits remains one of the most important research questions in plant ecological speciation9,32. It has long been known that soil can affect plant evolution through imposing selection on mechanisms for nutrient gain, potentially leading to edaphic specialization and the formation of plant races1,8,78. We show here that soil-type also impacts the way pollinators shape plant evolution, and that the interaction between bee-pollination and soil-type causes the strongest phenotypic divergence after 8 generations of experimental evolution. Thus, our data show that soil and bee-pollination can drive rapid plant divergent evolution without a shift in pollinators, thereby affecting not only floral phenotype, but a range of traits related to reproduction, growth, as well as defense. This “soil-pollinator effect” may be caused by i) trade-offs between growth, defense and pollinator attraction; such trade-offs should be more pronounced in nutrient-limited environments, thus slowing down pollinator-driven adaptation. Indeed, in our experiment, plants in the more nutrient-limited limestone soil showed less adaptive evolution and divergence in response to bee-pollination. In addition, ii) different patterns of phenotypic plasticity caused by different soils can change patterns of pollinator-mediated selection, thus also impacting trajectories of adaptive evolution33. We argue that the soil-pollinator effect deserves more attention as a potential driver of phenotypic divergence in plants, and perhaps as a starting point and/or enhancer for subsequent shifts in pollinators leading to the evolution of pollination ecotypes.
Whereas the role of individual ecological factors for plant divergent evolution has been investigated in considerable detail, interactions among factors have generally received less attention61,79,80. In our study, both the effects of herbivory and pollination on plant evolution were soil-dependent, highlighting the importance of considering multiple factors for trait evolution in plants79,81. The importance of herbivory × soil interactions for the evolution of plant defense has been conceptualized in the “growth-defense trade-off”, which has received some attention in previous research. Whereas this hypothesis predicts that plants in low-nutrient soils evolve higher defenses48,55,82, we did not find higher amounts of glucosinolate defense compounds in plants that evolved in limestone soil (the poorer soil type, see ref. 33). Plants that evolved in limestone soil with aphid-herbivory and bee-pollination actually showed the lowest production of glucosinolates. A reason for this may be that the specialist herbivore used in our experiment does not efficiently select for increased glucosinolates, because of its tolerance against aliphatic and to some degree also indolic glucosinolates83. In addition, animal pollinators are thought to select against chemical plant defenses, because the presence of defensive compounds in nectar or pollen may make floral rewards less palatable to them81,84,85. Indeed, in our study, bee-pollination led to a reduction in the amounts of most glucosinolates, in both soil types. This shows, as suggested by earlier studies61,85,86 that biotic pollination can lead to lower degrees of chemical defense, when selection for elevated defense chemistry is absent or weak.
Although aphid-herbivory in our experiment did not lead to consistently higher glucosinolate production, it still compromised the evolution of flower attractiveness to pollinators. This confirms earlier experimental evolution studies using chewing herbivores61, as well as numerous single-generation studies that showed decreased flower attractiveness as a pleiotropic effect of herbivore-induced plasticity59,80,87. In our study, aphid-herbivory likely decreased flower attractiveness through diminishing floral scent emission and plant height, that both signal plant attractiveness to pollinators in the here used as well as in other plant-pollinator systems31,88.
Different patterns and strengths of trait evolution among plant populations lead to population-level differences in plant phenotypes, called plant races, or ecotypes, in case population differences are also associated to local adaptation89. In our experiment, the combination of soil difference and bee-pollination led to the strongest divergence among our experimental populations. Whereas both soil and pollinators are considered key drivers of plant diversification62,90,91, the interaction between the two factors has received little attention, despite closely related plants growing on different soil types often differ in floral traits62,92,93,94,95, suggesting a link between soil- and flower divergence. Van der Niet63 interpreted the combined divergence between soil specialization and floral trait divergence as the outcome of reinforcement, because this pattern was more pronounced when plants occurred in sympatry than in allopatry. In our experiment, the mechanism for soil × pollinator interactions driving divergence are likely twofold. First, similar to the growth-defense trade-off, plants under pollinator-mediated selection face different trade-offs when growing on soils with different nutrient availability. In low-nutrient soil, such as our limestone soil, a decrease in above-ground traits, as observed in our study, yet not predicted by pollinator-mediated selection, may result from biomass trade-offs, where plants allocate more resources into below-ground and less into above-ground biomass96. Such reallocations allow for an optimal nutrient investment, as they lead to a direct fitness benefit or an increase in overall resource uptake97,98,99. When nutrients are less limited, as in our tuff soil, such trade-offs may be less important and plant evolutionary trajectories may follow more directly selection imposed by pollinators. Secondly, and in addition to trade-offs, Dorey and Schiestl33 showed that pollinator-mediated selection can change depending on patterns of phenotypic plasticity induced by different soil types. The patterns of divergence documented here are likely the outcome of the combined effects of different selection and different trade-offs as a consequence of growing on different soil types.
Floral diversification has been assumed to be tightly connected to either quantitative or qualitative shifts in pollinators, typically driven by pollen limitation10,11,31,100,101,102,103. Nevertheless, flowers may also diversify without pollinator shifts104, and mechanisms underlying this phenomenon may relate to different use of the same pollinators, abiotic factors, antagonists, or plant community composition68,81. Our study adds the soil-pollinator effect to the mechanisms leading to floral (and general plant) divergence without pollinator shifts. As the soil-pollinator effect forms a starting point for population divergence, it may subsequently be followed by a shift in pollinator guilds, as traits related to pollinator attraction (such as floral scent and petal size) or pollinator-flower fit (such as flower- or reproductive organ size) diverge between populations. The consequence may be a change in pollinator effectiveness (sensu78), thus further reinforcing divergent selection caused by different pollinators attracted, and/or their different efficiency in flowers of plants growing on different soil types. In this way, the soil-pollinator effect may form the starting point for pollinator-driven plant diversification, however, independent of a geographic pollinator mosaic.
Methods
Study system
In this experiment we used annual “fast cycling” Brassica rapa plants (Fast Plants® Standard Seed with maximum genetic diversity). These plants are fully outcrossing (self-incompatible), have short generation time, harbor a high standing genetic variation and have previously been shown to be suitable for selection- and experimental evolution studies31,61,76,105.
Experimental design
In 2018, 440 seeds of fast cycling Brassica rapa obtained from Carolina Biological Supply (Burlington, NC, USA) were sown out on standardized soil (commercially obtained soil with a homogeneous texture and distribution of nutrients) in a phytotron under standardized humidity, temperature and water conditions. Of these 440 seeds, 410 germinated and were used to produce full sib seed families by artificial crossings. Artificial crossings consisted of manually crossing 205 randomly assigned plant pairs, which resulted in a total of 163 seed families (only pairs where both parents produced seeds were used as seed families). Of these 163 families, 98 families were randomly selected for use as the starting population of our experimental evolution.
Experimental evolution
Our experimental evolution included 8 different treatments, which encompassed a full factorial combination of three different factors: growing in either limestone- or tuff soil (see Supplementary Table 11 for chemical and physical properties of each soil type), hand-pollination or bumblebee-pollination, with- or without aphid-herbivory (Fig. 1). The 98 families were randomly assigned to two replicates (A and B) and 8 treatment groups, so that each replicate included 49 plants (allowing for a squared 7 × 7 setup during pollination). Plants were first grown in a phytotron under 24 h of light, 21 °C, 60% humidity and were watered once a day (at 8:00 h). After repotting, we moved the plants to an air-conditioned greenhouse with natural and additional artificial illumination to achieve growing conditions with 16 h of light and a constant temperature of 23 °C, with uniform watering. The evolution experiment lasted for 8 generations with insects, followed by two more generations without any insects, including one in a common garden where plants were grown in standardized soil. This was done to avoid plasticity due to herbivory and to reduce maternal effects carried throughout the experiment (e.g., in aphid-herbivory groups). Because no sign of inbreeding was found after 8 generations (see below), replicates were not crossed but kept as isolated lines during the 10 generations to be able to assess independent and reproducible evolutionary changes. Inbreeding effects were assessed after eight generations of selection by crossing plants within or between replicates. We showed that plants crossed between replicates produced a similar amount of seeds than those gained from within-replicate crosses (ANOVA: F271 = 0.05, P = 0.817, Supplementary Data 5). Plants grown from the seeds of the crosses within and between replicates also showed similar phenotypes (MANOVA including five traits that were analyzed: F254 = 1.58, P = 0.167, Supplementary Data 5), suggesting little or no inbreeding effects on plant phenotypes.
Selection treatments
Our evolution experiment was set up to mirror natural conditions found in natural Southern Italian populations of Brassica spp., especially B. incana, a close relative to Brassica oleracea and B. rapa (Arrigo and Schiestl, unpublished data). The Mediterranean region, and especially Southern Italy is a center of diversity for Brassica spp. with three species strictly endemic to this region106. Both soil types used throughout the experiment were collected in Brassica incana populations in Campania (Italy) and represent two of the major types (tuff, limestone) of soil on which Brassica spp. grow (Arrigo and Schiestl, unpublished data). Soil was collected in July and October 2018, at Valico di Chiunze (40.719°N, 14.619°E) for limestone, and at Monte di Procida (40.809°N, 14.045°E) for tuff soil. We collected soil from the surface layer (0–15 cm, ca. 500 kg per soil type), sifted it (using a mesh of 1 cm of diameter), and stored it in textile bags to be shipped to Switzerland. The chemical composition of the soils was analyzed (see ref. 33). The soil was not sterilized before use. Because at generation three and four our experimental B. rapa plants produced few seeds, from generation five onwards we added fertilizer to both soil types in the form of 10 mL of an universal garden fertilizer diluted in 10 L of water (NPK: 8-6-6 with traces of B, Cu, Fe, Mn, Mo, Zn; Wuxal, Maag Agro, Dielsdorf, Switzerland). These nutrient additions were first performed during sowing and then a second time after pricking. Because resource limitation is likely an important component of soil-driven evolution, we analyzed resource limitation after adding fertilizer. We showed that reproduction and growth in our experimental plants was still resource limited even with fertilizer, as plants produced fewer flowers, fewer seeds, smaller flowers as well as had a lower growth rate than plants grown on standardized soil with optimal fertilization (Supplementary Fig. 1, Supplementary Data 6).
Herbivory
Plants were exposed either to pre-flowering aphid-herbivory (Brevicoryne brassicae) or kept uninfested (as indicated by the symbols, H: herbivory, NH: no herbivory). Brevicoryne brassicae is a notorious agricultural pest that specializes on Brassicaceae by sequestrating glucosinolates for self-defense. In the wild, Brevicoryne brassicae is one the most common herbivores on crucifers such as Brassica incana (Arrigo and Schiestl, unpublished data). To start an aphid colony, we collected individuals from different plants at the Botanical Garden of the University of Zurich (Switzerland) in the summer of 2018 and reared them in a climatic chamber on unselected lines of fast cycling Brassica rapa plants under 16 h lights at a temperature of 23 °C and humidity of 70%. In our evolution experiment, aphid-herbivory started at “the two true leaves stages”, which occurred around 13–14 days after sowing out for plants growing in tuff and 17–18 days after sowing out for plants growing in limestone. Each individual plant was infested on the leaves with 10 wingless individuals, which were allowed to feed and reproduce on plants for 72 h. Previous studies showed that 10 aphids are enough to induce strong plastic responses in plants within the first hours of the infestation107 and to lead to different levels of infestation depending on plant defenses108. To avoid aphid migration to more suitable plants, infested-plants as well as non-infested plants were covered by a net (7 × 7 × 15 cm) with mesh size of 680 µm (Bugdorm, model DC0901-W). After the three days of herbivory, nets and aphids were carefully removed from the plants and from this day on were checked every day for left-over aphids, which were subsequently removed as well.
Pollination
For pollination, we either hand-pollinated plants or used bumblebees (Bombus terrestris61 for pollination (as indicated by the symbols, H: hand-pollination, B: bee-pollination). Bombus terrestris is a common and efficient pollinator of crucifers and is also one of the most common pollinator of populations of Brassica incana (Arrigo and Schiestl unpublished data109,110;). For all pollination including pollinator bioassays at the end of the experiment, we used only bumblebee workers. Bees were purchased in Switzerland (Andermatt Biocontrol Suisse AG) and hives were kept inside the greenhouse in a flight cage (l × w × h: 75 × 75 × 115 cm). One to two colonies of B. terrestris were used every generation for pollination, and new colonies were used for each generation. Because bumblebees had not experienced any plants or flowers before being delivered, we fed them for a week with Brassica rapa “fast cycling” plants to gain experience with these flowers. In addition to plant flowers, bumblebees were fed with supplemental pollen (Biorex, Ebnat-Kappel, Switzerland) and sugar water (Biogluc sugar solution, Biobest). To avoid bias in pollinator choice, “feeding plants” were from unselected lines and were grown on standardized soil. Three days before pollination and choices test, we removed all plants from the cages and supplemental pollen and nectar solution were the only source of food left for bumblebees. To enhance bumblebee’s foraging activities, bees were starved by removing all food 16 h before pollination and choices tests. Pollination as well as bee choice tests were done 10 days after herbivory (when all aphids had been removed) and when most of the plants were flowering simultaneously on both soil types. Pollination was performed between 8.30am and 5.30 pm for each replicate and in each generation. For pollination, plants were randomly set up in a square of 7 × 7 in a flight cage (l × w × h: 2.5 m × 1.8 m × 1.2 m) with a distance of 20 cm between plants. A total of seven bee workers were released individually and sequentially, and recaptured after visiting a total of five different plants; each bee was only used once throughout the experiment. As a consequence of bee-pollination, for each replicate in each generation, around 17 plants produced seeds (replicate A, mean: 17.41 ± 2.63; range of 11–23, replicate B, mean 16.57 ± 2.27; range of 12–22). The number of bee visits was limited to ensure pollen limitation and thus pollinator-mediated selection. The level of pollen limitation at the replicate level in our experiment was considered at the upper range of pollen limitation in natural environments, where many more plants interact with fewer pollinators111,112. For the hand-pollinated plant groups, we randomly selected 28 plants per replicate for pollination to achieve a similar proportion of pollinated plants. In these control groups, we randomly assigned one father plant to a mother plant of the same replicate, sampled one long stamen that we used to deposit an excess of pollen on stigmas of four flowers per mother plant. Because in the bee-pollinated treatments, visited plants had an average visitation (bees per plant) between 1 and 2 (mean ± s.d.: TNHB: 1.55 ± 0.13; THB: 1.78 ± 0.22; LNHB: 1.66 ± 0.23; LHB: 1.71 ± 0.25), a single father plant for the hand-pollinated treatments was considered leading to similar average number of paternities compared to the bee-pollinated treatments. Because in Brassica rapa flowering occurs in acropetal succession, we marked the lowest and highest open flowers of inflorescences of visited plants and only fruits that developed from between these marks were collected and counted after ripening. After pollination, plants were kept in the same standardized conditions for four weeks after which plants were deprived of water, to dry for seed maturation. Once dried, fruits and seeds of the different treatment groups were harvested, counted, and weighted. Seed set per plant was then used to calculate the contribution of each plant to the next generation, to ensure that all individuals contributed proportional to their seed set in the replicate to the next generation. Plant relative seed set, calculated as the seed production of one individual divided by the mean seed set of the replicate, was used for calculating a plant’s contribution to the next generation as it indicates the contribution of each individual to the total seed set pool in the replicate. Seed contribution defined as relative seed set was adjusted to achieve a sample size of 49 plants per replicate. The seed contribution of each pollinated plant into next generation can therefore be summarized with the following formula:
Evolutionary changes
Because our experimental plants did not experience any phenotypic signs of inbreeding effects, in generation 9, we manually and randomly cross-pollinated all plants within each replicate, so that each plant was a pollen donor and also a pollen recipient. Of the resulting seeds (generation 10), we picked randomly one seed of 36 families to obtain 36 plants per replicate per treatment. We also selected 36 families of the ancestral population (generation one) for each replicate. To assess evolutionary changes, plants of generation 10 were grown simultaneously with generation one (i.e. as a resurrection experiment) on standardized soil (with optimal fertilization; Patzer Erden, Sinntal Germany) and in standardized greenhouse conditions.
Plant traits
To assess evolutionary changes in plants at the end of the experiment, we measured plant morphology, floral volatile emission (scent), and leaf glucosinolate content in plants of generation one and ten on day 27, three days before bioassays were conducted. All plants were phenotyped on the same day in order to minimize variation between plants due to development. Because volatiles were collected in a non-destructive way (in contrast to some morphology measurements where three flowers were sampled), we performed scent collection first to avoid collecting altered volatile emission from damaged plants. Floral volatiles were collected from plant inflorescences as soon as at least three flowers were open, using headspace sorption with tenax sorbent and a push-pull collection system33. Samples were analyzed by gas chromatrography with mass selective detection, identified by comparison of mass spectra and retention times with synthetic reference standards, and quantified using dose-response curves for all compounds. Amounts of scent compounds were calculated on a per-flower basis for statistical analysis. A detailed description of the collection, identification and analyses protocol can be found in33. Floral morphology (petal display: petal width, petal length, flower diameter, sepal length, long stamen length, pistil length) was measured by sampling 3 flowers per plant and by carefully placing petals and reproductive organs on a white paper sheet. Sheets were immediately scanned before flower display started wilting. Mean values of the three sampled flowers were calculated and used in statistical analyses. Floral nectar was collected from the same three flowers using a 1 μL microcapillary (Blaubrand, Wertheim, Germany). Nectar amount per flower was then calculated by dividing the collected nectar volume by the number of sampled flowers. Plant height was measured at two different times of plants growth, once in the early stage when the first plants started to flower (day 20), and a second time on the day when bioassays were conducted (day 30). On the bioassay day, we counted number of flowers, flower production from flowering onset to bioassays day, cumulated secondary branch length, number of flowering branches, as well as leaf traits such as leaf length and -width. From leaf length and leaf width we estimated leaf area according to the following formula: leaf size = (leaf width × leaf length) divided by 2. Flower production rate was calculated using the time to flower and flower production according to the following formula: flower production rate = flower production from flowering onset to bioassays date/flowering duration, where flowering duration represents the number of days a plant had flowered before bioassays.
Leaf glucosinolates
For leaf glucosinolates analyses, we collected, on day 34, around 50 mg of fresh leaf tissue from 26 to 35 plants of each replicate and treatment of generation ten and one. Leaf tissue was flash frozen in liquid nitrogen to avoid the hydrolysis of glucosinolates by myrosinase. After dry-freezing samples for 48 h (10 bar, −80 °C, alpha 2–4 LSCplus, Christ, Germany), we weighed samples and ca. 20 mg per sample were ground to a fine powder at a speed of 4 m/s with a FastPrep-24TM grinder (MP BiomedicalsTM) using 3 beaded balls. We added 1 ml of ice-cold MeOH: water (70: 30; methanol HPLC grade) with sinalbin (5 μg ml−1) as internal standard to the freshly ground samples. Samples were vortexed for 5 s and immediately incubated for 10 min at 85 °C in a shaking bloc heater (600 rpm, Eppendorf Thermomixer® comfort; Eppendorf, Hamburg, Germany). To assure full extraction of the leaf material, samples were moved in an ultrasonic water bath for 10 minutes (AL 04-04; Advantage-LabTM, Darmstadt, Germany) and then centrifuged at 20,000 × g for 10 min (Sigma 1–16, Sigma Laborzentrifugen GmbH, Osterode am Harz, Germany). We transferred 0.7 mL of the resulting supernatant to an Eppendorf tube stored at −20 °C for two hours. Because the supernatant was not clear enough, we centrifuged it a second time at 20,000 × g for 10 min and 0.35 mL of the supernatant was transferred into a glass vial which was stored at −20 °C until high-pressure liquid chromatography (HPLC) analysis. We injected 15 μL of supernatant into an Agilent 1260 Infinity II Binary LC system, and compounds were separated on an Agilent Poroshell 120 CS-C18 column (2.7 μm, 2.1 × 100 mm). Compounds were eluted at a constant flow of 0.5 mL/min at 40 °C with an acetonitrile–0.25% formic acid gradient as follows: starting at 2% acetonitrile, 5 min at 30% acetonitrile, 7.5 min at 45% acetonitrile, 8–11.5 min at 100% acetonitrile, and reconditioning for 3.5 min at 2% acetonitrile. To quantify glucosinolates, we recorded ultraviolet (UV) absorbance spectra at 229 nm by a diode array detector, and compared spectra to the peak area of the known concentration of the internal standards. We identified 11 candidate glucosinolate compounds with distinct molecular masses and HPLC retention times. Glucosinolates were quantified using four calibration curves with four levels (k = 4) prepared in MeOH: water (70: 30 ratio) at the following concentrations: 1, 5, 10, 20 μg ml−1, for gluconapin (linear curve fit, R2 = 0.9994), glucobrassicanapin (linear curve fit, R2 = 0.9989), gluconasturtiin (linear curve fit, R2 = 0.9997) and sinalbin (linear curve fit, R2 = 0.9998). Calibration curves and quantifications were based on the peak area ratio between each analyte and the internal standard (sinalbin at 5 μg.ml−1). The concentration range was selected to include the expected plant glucosinolate concentration and the calibration curve (gluconapin, glucobrassicanapin, gluconasturtiin, sinalbin) was selected according to glucosinolate side chain (either indole-glucosinolate, aromatic, thioakyl or alkenyl). We identified ten different glucosinolates in our leaf samples and all were considered for statistical analyses. We identified outliers in some of the glucosinolates compounds and decided to consider these values as missing values rather than excluding whole individuals. Final sample size was therefore varying in a range of N = 552 for glucoraphanin to N = 579 for indole glucosinolates.
Pollinator preference assays
To assess differences in plants’ attractiveness to pollinators, we performed bumblebee (Bombus terrestris) preferences (first choice) assays at the usual pollination date of the tuff-soil evolutionary lines (30 day after sowing out) between 8:30am and 5:00 pm. Each run of the behavioral assays included nine plants, with one individual of each treatment group (generation 10) and one plant of the starting population (generation one, G1) assayed together. The nine plants were set up in a flight cage (l × w × h: 2.5 m × 1.8 m × 1.2 m) under controlled light and temperature conditions, in a squared 3 × 3 set up with a distance of 20 cm between individual plants. To ensure independence between choices and plant position, we changed the plant positions between runs, and we also released the bumblebees at different locations. For each test run, a total of three bumblebees were used to assess plant attractiveness. Bees were released individually and caught immediately after they made their first choice. Each bee was only used once, and a successful choice was recorded only when bees landed on an inflorescence and started to collect nectar and/or pollen. In total 71 runs of bioassays were performed, using a total of 639 plants and 213 bees.
Aphid preference assays
To assess differences in plant attractiveness to Brevicoryne brassicae aphids, we performed aphid preferences assays at the pollination date of the evolution experiment (30 day after sowing out). Only morphs of aphids at the migratory stage (alate = winged) were used in behavioral assays. Every test run included the same nine plants that were previously used for pollinator preference assays (one individual of each treatment groups (generation 10) and one plant of the initial population, generation one). Plant attractiveness to aphids was assessed in a flight cage (l × w × h: 0.9 m × 0.6 m × 0.6 m) under controlled light and temperature conditions where plants were placed in a squared 3 × 3 set up with a distance of 10 cm between plants. For each test run, a total of 10 alates were used to assess plant attractiveness to aphids. Aphids were released by deposing them on the cage in the middle of the square. Because alatae can take 48–72 h after release before making their final choice113, we only recorded choices after 72 h by recording how many individuals were found on each plant of the test group. Tests were replicated 71 times for a total of 710 aphids and 639 plants used.
Statistical analysis
Trait evolution
Evolutionary divergence in floral traits was evaluated in generation 10 by one-way ANOVA, linear discriminant function analysis (DFA), and linear mixed models. ANOVA was performed to assess the divergence of traits in plants of different soil types in generation 10. ANOVA was done for 33 individual traits in all treatment groups and replicates separately, using “soil” as independent factor. The resulting F values for the 33 traits (Supplementary Data 2) were compared by a general linear model with F-values as dependent variable, herbivory, pollination, and their interaction as fixed factors, and replicate as random factor. Because some traits were highly correlated (Supplementary Fig. 2), we excluded several traits from this analysis. The following traits were excluded because of high (>0.65) correlation coefficients with other variables: height at day 20 (r2 = 0.71 with flowering time), number of flowering branches (r2 = 0.76 with cumulative branches length), petal length (r2 = 0.81 with flower diameter), petal width (r2 = 0.68 with flower diameter), flowers produced per day (r2 = 0.88 with flower number), phenylethyl alcohol emission (r2 = 0.89 with phenylacetaldehyde), and benzyl nitrile emission (r2 = 0.73 with 2-aminobenzaldehyde). In our selection between correlated terms, we applied a criterion of “significant selection”, wherein all traits known to be under significant selection in our model system were kept. We did not use principle components for this analysis as it would have made the interpretation and separation into categories (morphological, floral scent, glucosinolates) difficult.
For a multivariate analysis of trait divergence, we used DFA in each of the pollination treatment groups separately. To perform DFA on uncorrelated variables, we used principle component scores (PCs) in the DFA (Supplementary Data 3). We conducted principal component analysis on all traits (N = 40, but excluding total volatile emission and total glucosinolate production) of generation 10 and 1 combined. The principal component analysis resulted in 12 PCs with an eigenvalue above 1 explaining 72.1% of the total variation (Supplementary Table 10).
To study the contribution of treatments (soil, pollination and herbivory) on plant evolution, we used linear mixed model (package lme4), with individual traits as dependent variable, with “replicate”, as random factor, and “treatment” and its interaction with replicate as fixed factors (Supplementary Table 5, Supplementary Data 1). Additionally, individual trait values between plants of generation one and ten were also compared using pairwise post hoc comparisons (Supplementary Table 7, Supplementary Data 1).
The impact of “pollination”, “herbivory” and “soil”, and their interactions on evolutionary changes was evaluated in plants of generation 10 only. We used a linear mixed model (package lme4), with individual traits as dependent variables, replicate as random factor, “pollination”, “herbivory”, “soil”, and their interactions as fixed factors (Table 1, Supplementary Table 6, Supplementary Data 1). To simplify the interpretation of the multiple significant interactions, we also assessed the evolutionary changes within each soil line separately (Supplementary Table 8). The values for all floral scent compounds, leaf glucosinolates, and nectar amount were ln(x + 1) transformed before analysis to approach normal distribution.
We assessed evolutionary rates by calculating the haldane index for synchronic comparisons between evolved lines and ancestral lines114:
where \(\bar{{x}_{e}}\) represents the mean trait value for one evolved line (G10), and \(\bar{{x}_{a}}\) represents the mean value of the same trait for the ancestral population (G1), \({\sigma }_{p}\) represents the pooled standard deviation of the evolved and ancestral lines, and g is the number of generations during the experiment (g = 10 for the current study). Evolutionary changes in haldanes for individual traits are reported in Supplementary Table 1 and Supplementary Data 4. Because a single evolutionary line had both negative and positive evolutionary rates, we used absolute values for statistical analyses. Therefore, absolute values of haldanes were used in linear mixed models to assess how “pollination”, “herbivory”, “soil” and their interactions affected evolutionary rates of different trait groups (morphology, floral scent, leaf glucosinolates). To do so, absolute values of haldanes of each trait was used as dependent variable, “replicate” as random factor, “pollination”, “herbivory”, “soil”, “trait groups” and their interactions as fixed factors (Supplementary Table 2). Because soil was a strong factor driving plant evolution, we later ran linear mixed models within each soil evolutionary line separately. In these analyses, we used absolute values of haldanes as dependent variable, “replicate” as random factor, and “pollination”, “herbivory”, “trait groups” and their interactions as fixed factors (Supplementary Table 3). These analyses were performed using the same 33 traits as for the F-value analysis, to reduce the effect of correlations in the overall assessment of evolutionary rates among trait categories.
Plant attractiveness
Attractiveness of plants to bumblebees and winged aphids was analyzed using generalized linear model with Poisson distribution, with “first choices” as dependent variable, replicate as random factor and “pollination”, “herbivory”, “soil”, and their interactions as fixed factors (Supplementary Data 1). All statistical analyses were performed with R software 4.0.0 (2020, R Foundation for Statistical Computing, Vienna, Austria). Generally, we did not adjust P-values for multiple comparisons (Bonferroni correction), because we compared individual traits between treatment groups, not groups of traits. For individual traits, the likelihood of type I errors is independent of the number of comparisons done.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data generated in this study have been deposited in the ZORA database of the University of Zürich at https://doi.org/10.5167/uzh-257663.
References
Grant, V. Plant speciation. (Columbia University Press, 1981).
Rieseberg, L. H., Widmer, A., Arntz, A. M. & Burke, J. M. Directional selection is the primary cause of phenotypic diversification. Proc. Natl Acad. Sci. USA 99, 12242–12245 (2002).
Hall, M. C. & Willis, J. H. Divergent selection on flowering time contributes to local adaptation in Mimulus guttatus populations. Evolution 60, 2466–2477 (2006).
Johnson, N. C., Wilson, G. W., Bowker, M. A., Wilson, J. A. & Miller, R. M. Resource limitation is a driver of local adaptation in mycorrhizal symbioses. Proc. Natl Acad. Sci. 107, 2093–2098 (2010).
Briscoe Runquist, R. D. et al. Context dependence of local adaptation to abiotic and biotic environments: a quantitative and qualitative synthesis. Am. Naturalist 195, 412–431 (2020).
Gómez, J., Perfectti, F., Bosch, J. & Camacho, J. A geographic selection mosaic in a generalized plant–pollinator–herbivore system. Ecol. Monogr. 79, 245–263 (2009).
Kalske, A., Muola, A., Laukkanen, L., Mutikainen, P. & Leimu, R. Variation and constraints of local adaptation of a long‐lived plant, its pollinators and specialist herbivores. J. Ecol. 100, 1359–1372 (2012).
Rajakaruna, N. Lessons on evolution from the study of edaphic specialization. Botanical Rev. 84, 39–78 (2018).
Marquis, R. J. et al. Ode to Ehrlich and Raven or how herbivorous insects might drive plant speciation. Ecology 97, 2939–2951 (2016).
Johnson, S. D., Harder, L. & Barrett, S. Pollinator-driven speciation in plants (Ecology and evolution of flowers) (Oxford: Oxford University Press, 2006).
van der Niet, T. & Johnson, S. D. Phylogenetic evidence for pollinator-driven diversification of angiosperms. Trends Ecol. Evol. 27, 353–361 (2012).
Cheplick, G. P. Approaches to plant evolutionary ecology (Oxford University Press, 2015).
Marsden-Jones, E. M. & Turrill, W. B. Fifth report of the transplant experiments of the British Ecological Society at Potterne, Wiltshire. J. Ecol. 26, 359–379 (1938).
Silvertown, J. et al. The Park Grass Experiment 1856–2006: its contribution to ecology. J. Ecol. 94, 801–814 (2006).
Cheeseman, J. M. Mechanisms of salinity tolerance in plants. Plant Physiol. 87, 547–550 (1988).
Roy, S. J., Negrão, S. & Tester, M. Salt resistant crop plants. Curr. Opin. Biotechnol. 26, 115–124 (2014).
Terés, J. et al. Soil carbonate drives local adaptation in Arabidopsis thaliana. Plant, cell Environ. 42, 2384–2398 (2019).
Brady, K. U., Kruckeberg, A. R. & Bradshaw, H. D. Jr Evolutionary ecology of plant adaptation to serpentine soils. Annu. Rev. Ecol. Evol. Syst. 36, 243–266 (2005).
Baker, A. Ecophysiological aspects of zinc tolerance in Silene maritima With. N. Phytologist 80, 635–642 (1978).
Bradshaw, A. D. Populations of Agrostis tenuis resistant to lead and zinc poisoning. Nature 169, 1098–1098 (1952).
Field, C. The photosynthesis-nitrogen relationship in wild plants. In On the economy of form and function (Cambridge University Press, 1986).
Wright, I., Reich, P. & Westoby, M. Strategy shifts in leaf physiology, structure and nutrient content between species of high‐and low‐rainfall and high‐and low‐nutrient habitats. Funct. Ecol. 15, 423–434 (2001).
Vance, C. P., Uhde‐Stone, C. & Allan, D. L. Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. N. Phytologist 157, 423–447 (2003).
Lambers, H., Shane, M. W., Cramer, M. D., Pearse, S. J. & Veneklaas, E. J. Root structure and functioning for efficient acquisition of phosphorus: matching morphological and physiological traits. Ann. Bot. 98, 693–713 (2006).
Watanabe, T., Osaki, M., Yano, H. & Rao, I. M. Internal mechanisms of plant adaptation to aluminum toxicity and phosphorus starvation in three tropical forages. J. Plant Nutr. 29, 1243–1255 (2006).
Schiestl, F. P. & Johnson, S. D. Pollinator-mediated evolution of floral signals. Trends Ecol. Evol. 28, 307–315 (2013).
Strauss, S. Y. & Irwin, R. E. Ecological and evolutionary consequences of multispecies plant-animal interactions. Annu. Rev. Ecol. Evol. Syst. 35, 435–466 (2004).
Agrawal, A. A. Induced responses to herbivory in wild radish: effects on several herbivores and plant fitness. Ecology 80, 1713–1723 (1999).
Karban, R. & Baldwin, I. Induced Responses to Herbivory, 319 (University of Chicago Press, 1997).
Fenster, C. B., Armbruster, W. S., Wilson, P., Dudash, M. R. & Thomson, J. D. Pollination syndromes and floral specialization. Annu. Rev. Ecol. Evol. Syst. 35, 375–403 (2004).
Gervasi, D. D. & Schiestl, F. P. Real-time divergent evolution in plants driven by pollinators. Nat. Commun. 8, 14691 (2017).
van der Niet, T., Peakall, R. & Johnson, S. D. Pollinator-driven ecological speciation in plants: new evidence and future perspectives. Ann. Bot. 113, 199–212 (2014).
Dorey, T. & Schiestl, F. P. Plant phenotypic plasticity changes pollinator‐mediated selection. Evolution 76, 2930–2944 (2022).
David, T. I., Storkey, J. & Stevens, C. J. Understanding how changing soil nitrogen affects plant-pollinator interactions. Arthropod Plant Interact. 13, 671–684 (2019).
Adler, L. S., Wink, M., Distl, M. & Lentz, A. J. Leaf herbivory and nutrients increase nectar alkaloids. Ecol. Lett. 9, 960–967 (2006).
Burkle, L. A. & Irwin, R. E. Beyond biomass: measuring the effects of community-level nitrogen enrichment on floral traits, pollinator visitation and plant reproduction. J. Ecol. 98, 705–717 (2010).
Meindl, G. A., Bain, D. J. & Ashman, T. L. Edaphic factors and plant-insect interactions: direct and indirect effects of serpentine soil on florivores and pollinators. Oecologia 173, 1355–1366 (2013).
Alonso, C. & Herrera, C. M. Developmental and spatial covariation of nutrients in growing leaves of Daphne laureola and their relationships with herbivory. N. Phytologist 159, 645–656 (2003).
Boyd, R. S. & Martens, S. N. The significance of metal hyperaccumulation for biotic interactions. Chemoecology 8, 1–7 (1998).
Meindl, G. A. & Ashman, T.-L. Effects of soil metals on pollen germination, fruit production, and seeds per fruit differ between a Ni hyperaccumulator and a congeneric nonaccumulator. Plant Soil 420, 493–503 (2017).
Meindl, G. A. & Ashman, T.-L. Nickel accumulation by Streptanthus polygaloides (Brassicaceae) reduces floral visitation rate. J. Chem. Ecol. 40, 128–135 (2014).
Galen, C. High and dry: Drought stress, sex-allocation trade-offs, and selection on flower size in the alpine wildflower Polemonium viscosum (Polemoniaceae). Am. Naturalist 156, 72–83 (2000).
Galen, C., Kaczorowski, R., Todd, S. L., Geib, J. & Raguso, R. A. Dosage-dependent impacts of a floral volatile compound on pollinators, larcenists, and the potential for floral evolution in the alpine skypilot Polemonium viscosum. Am. Naturalist 177, 258–272 (2011).
González-Teuber, M. & Heil, M. Nectar chemistry is tailored for both attraction of mutualists and protection from exploiters. Plant Signal. Behav. 4, 809–813 (2009).
Rusman, Q., Poelman, E. H., Nowrin, F., Polder, G. & Lucas‐Barbosa, D. Floral plasticity: herbivore‐species‐specific‐induced changes in flower traits with contrasting effects on pollinator visitation. Plant Cell Environ. 42, 1882–1896 (2019).
Theis, N., Lerdau, M. & Raguso, R. A. The challenge of attracting pollinators while evading floral herbivores: patterns of fragrance emission in Cirsium arvense and Cirsium repandum (Asteraceae). Int. J. Plant Sci. 168, 587–601 (2007).
Herrera, C. M. et al. Interaction of pollinators and herbivores on plant fitness suggests a pathway for correlated evolution of mutualism-and antagonism-related traits. Proc. Natl Acad. Sci. 99, 16823–16828 (2002).
Coley, P. D., Bryant, J. P. & Chapin, F. S. III Resource availability and plant antiherbivore defense. Sci. 230, 895–899 (1985).
Endara, M.-J. & Coley, P. D. The resource availability hypothesis revisited: a meta-analysis. Funct. Ecol. 25, 389–398 (2011).
Janzen, D. H. Tropical blackwater rivers, animals, and mast fruiting by the Dipterocarpaceae. Biotropica 6, 69–103 (1974).
McKey, D. Adaptive patterns in alkaloid physiology. Am. Naturalist 108, 305–320 (1974).
Zangerl, A. R. & Bazzaz, F. A. Defense allocation. In Plant resistance to herbivores and pathogens: ecology, evolution, and genetics. 363–392 (University of Chicago Press, 1992).
Van Zandt, P. A. Plant defense, growth, and habitat: A comparative assessment of constitutive and induced resistance. Ecology 88, 1984–1993 (2007).
Agrawal, A. A. Macroevolution of plant defense strategies. Trends Ecol. Evol. 22, 103–109 (2007).
Fine, P. V., Mesones, I. & Coley, P. D. Herbivores promote habitat specialization by trees in Amazonian forests. science 305, 663–665 (2004).
Fine, P. V. et al. The growth–defense trade‐off and habitat specialization by plants in Amazonian forests. Ecology 87, S150–S162 (2006).
Armbruster, W. S. Exaptations link evolution of plant-herbivore and plant- pollinator interactions: a phylogenetic inquiry. Ecology 78, 1661–1672 (1997).
Kessler, A. & Halitschke, R. Testing the potential for conflicting selection on floral chemical traits by pollinators and herbivores: predictions and case study. Functional Ecol. 23, 901–912 (2009).
Schiestl, F. P. Ecology and evolution of floral volatile‐mediated information transfer in plants. N. Phytologist 206, 571–577 (2015).
Irwin, R. E. & Adler, L. S. Correlations among traits associated with herbivore resistance and pollination: implications for pollination and nectar robbing in a distylous plant. Am. J. Bot. 93, 64–72 (2006).
Ramos, S. E. & Schiestl, F. P. Rapid plant evolution driven by the interaction of pollination and herbivory. Science 364, 193–196 (2019).
Anacker, B. L. & Strauss, S. Y. The geography and ecology of plant speciation: range overlap and niche divergence in sister species. Proc. R. Soc. B Biol. Sci. 281, 20132980 (2014).
van der Niet, T., Johnson, S. D. & Linder, H. P. Macroevolutionary data suggest a role for reinforcement in pollination system shifts. Evolution 60, 1596–1601 (2006).
Christie, K. & Strauss, S. Y. Reproductive isolation and the maintenance of species boundaries in two serpentine endemic Jewelflowers. Evolution 73, 1375–1391 (2019).
Gardner, M. & Macnair, M. Factors affecting the co-existence of the serpentine endemic Mimulus nudatus Curran and its presumed progenitor, Mimulus guttatus Fischer ex DC. Biol. J. Linn. Soc. 69, 443–459 (2000).
Gomez, J. M., Munoz-Pajares, A. J., Abdelaziz, M., Lorite, J. & Perfectti, F. Evolution of pollination niches and floral divergence in the generalist plant Erysimum mediohispanicum. Ann. Bot. 113, 237–249 (2014).
Peter, C. I. & Johnson, S. D. A pollinator shift explains floral divergence in an orchid species complex in South Africa. Ann. Bot. 113, 277–288 (2014).
Ellis, A. G. & Anderson, B. Pollinator mediated floral divergence in the absence of pollinator shifts. In Evolution of plant–pollinator relationships, 237–262 (Cambridge University Press, 2012).
Van der Niet, T., Pirie, M. D., Shuttleworth, A., Johnson, S. D. & Midgley, J. J. Do pollinator distributions underlie the evolution of pollination ecotypes in the Cape shrub Erica plukenetii. Ann. Bot. 113, 301–315, (2014).
Antonelli, A. & Sanmartin, I. Why are there so many plant species in the Neotropics? Taxon 60, 403–414 (2011).
Lenski, R. E. What is adaptation by natural selection? Perspectives of an experimental microbiologist. PLOS Genet. 13, e1006668 (2017).
Tonnabel, J., David, P. & Pannell, J. R. Rapid divergence in vegetative morphology of a wind‐pollinated plant between populations at contrasting densities. Evolution 76, 1737–1748 (2022).
Agrawal, A. A., Hastings, A. P., Johnson, M. T., Maron, J. L. & Salminen, J.-P. Insect herbivores drive real-time ecological and evolutionary change in plant populations. Science 338, 113–116 (2012).
Zuest, T. et al. Natural Enemies Drive Geographic Variation in Plant Defenses. Science 338, 116–119 (2012).
Briggs, W. H. & Goldman, I. L. Genetic variation and selection response in model breeding populations of Brassica rapa following a diversity bottleneck. Genetics 172, 457–465 (2006).
Schiestl, F. P., Balmer, A. & Gervasi, D. D. Real‐time evolution supports a unique trajectory for generalized pollination. Evolution 72, 2653–2668 (2018).
White, N. J. et al. Experimental evolution of local adaptation under unidimensional and multidimensional selection. Curr. Biol. 32, 1310 (2022).
Stebbins, L. G. Adaptive radiation of reproductive characteristics in angiosperms, I: pollination mechanisms. Annu Rev. Ecol. Syst. 1, 307–326 (1970).
López-Goldar, X. & Agrawal, A. A. Ecological interactions, environmental gradients, and gene flow in local adaptation. Trends Plant Sci. 26, 796–809 (2021).
Lucas-Barbosa, D. Integrating studies on plant–pollinator and plant–herbivore interactions. Trends plant Sci. 21, 125–133 (2016).
Strauss, S. Y. & Whittall, J. B. Non-pollinator agents of selection on floral traits. Ecol. Evol. Flowers 2006, 120–138 (2006).
Griffiths, J. I., Petchey, O. L., Pennekamp, F. & Childs, D. Z. Linking intraspecific trait variation to community abundance dynamics improves ecological predictability by revealing a growth–defence trade‐off. Funct. Ecol. 32, 496–508 (2018).
Züst, T. & Agrawal, A. A. Mechanisms and evolution of plant resistance to aphids. Nat. Plants 2, 1–9 (2016).
Adler, L. S. The ecological significance of toxic nectar. Oikos 91, 409–420 (2000).
Adler, L. S., Seifert, M. G., Wink, M. & Morse, G. E. Reliance on pollinators predicts defensive chemistry across tobacco species. Ecol. Lett. 15, 1140–1148 (2012).
Bruinsma, M. et al. Folivory affects composition of nectar, floral odor and modifies pollinator behavior. J. Chem. Ecol. 40, 39–49 (2014).
Strauss, S. Y., Rudgers, J. A., Lau, J. A. & Irwin, R. E. Direct and ecological costs of resistance to herbivory. Trends Ecol. Evol. 17, 278–285 (2002).
Knauer, A. C. & Schiestl, F. P. Bees use honest floral signals as indicators of reward when visiting flowers. Ecol. Lett. 18, 135–143 (2015).
VanWallendael, A., Lowry, D. B. & Hamilton, J. A. One hundred years into the study of ecotypes, new advances are being made through large-scale field experiments in perennial plant systems. Curr. Opin. Plant Biol. 66, 102152 (2022).
Schnitzler, J. et al. Causes of plant diversification in the Cape biodiversity hotspot of South Africa. Syst. Biol. 60, 343–357 (2011).
van der Niet, T. & Johnson, S. D. Patterns of plant speciation in the Cape floristic region. Mol. Phylogenet. Evol. 51, 85–93 (2009).
Baldwin, B. G. Origin of the serpentine‐endemic herb Layla discoidea from the widespread L. landulosa (Compositae). Evolution 59, 2473–2479 (2005).
Goldblatt, P. Systematics of Freesia Klatt (Iridaceae). Ji. S. Aft. Bot. 48, 39–91 (1982).
Goldblatt, P. & Manning, J. C. Phylogeny and speciation in Lapeirousia subgenus Lapeirousia (Iridaceae: Ixioideae). Ann. Missouri Botanical Garden 84, 346–361 (1996).
Goldblatt, P. & Manning, J. C. Plant diversity of the Cape region of southern Africa. Ann. Missouri Botanical Garden, 89, 281–302 (2002).
Rehling, F., Sandner, T. M. & Matthies, D. Biomass partitioning in response to intraspecific competition depends on nutrients and species characteristics: A study of 43 plant species. J. Ecol. 109, 2219–2233 (2021).
Jungk, A. Root hairs and the acquisition of plant nutrients from soil. J. Plant Nutr. Soil Sci. 164, 121–129 (2001).
Rengel, Z. & Damon, P. M. Crops and genotypes differ in efficiency of potassium uptake and use. Physiologia Plant. 133, 624–636 (2008).
Smith, S. E. & Read, D. J. Mycorrhizal symbiosis (Academic press, 2010).
Breitkopf, H., Onstein, R. E., Cafasso, D., Schlüter, P. M. & Cozzolino, S. Multiple shifts to different pollinators fuelled rapid diversification in sexually deceptive Ophrys orchids. N. Phytologist 207, 377–389 (2015).
Serrano-Serrano, M. L., Rolland, J., Clark, J. L., Salamin, N. & Perret, M. Hummingbird pollination and the diversification of angiosperms: an old and successful association in Gesneriaceae. Proc. R. Soc. B Biol. Sci. 284, 20162816 (2017).
Valente, L. M., Manning, J. C., Goldblatt, P. & Vargas, P. Did pollination shifts drive diversification in Southern African Gladiolus? Evaluating the model of pollinator-driven speciation. Am. Naturalist 180, 83–98 (2012).
Xiang, G. J., Guo, Y. H. & Yang, C. F. Diversification of floral orientation in Lonicera is associated with pollinator shift and flowering phenology. J. Syst. Evol. 59, 557–566 (2021).
Ellis, A. G. & Johnson, S. D. The evolution of floral variation without pollinator shifts in Gorteria diffusa (Asteraceae). Am. J. Bot. 96, 793–801 (2009).
Tomkins, S. P. & Williams, P. H. Fast plants for finer science—an introduction to the biology of rapid-cycling Brassica campestris (rapa) L. J. Biol. Educ. 24, 239–250 (1990).
Snogerup, S., Gustafsson, M. & Von Bothmer, R. Brassica sect. Brassica (Brassicaceae) I. taxonomy and variation. Willdenowia 19, 271–365 (1990).
Pontoppidan, B., Hopkins, R., Rask, L. & Meijer, J. Infestation by cabbage aphid (Brevicoryne brassicae) on oilseed rape (Brassica napus) causes a long lasting induction of the myrosinase system. Entomolo. Exp. Appl. 109, 55–62 (2003).
Francis, F., Haubruge, E. & Gaspar, C. Influence of host plants on specialist/generalist aphids and on the development of Adalia bipunctata (Coleoptera: Coccinellidae). Eur. J. Entomol. 97, 481–486 (2000).
Jauker, F. & Wolters, V. Hover flies are efficient pollinators of oilseed rape. Oecologia 156, 819–823 (2008).
Rader, R. et al. Alternative pollinator taxa are equally efficient but not as effective as the honeybee in a mass flowering crop. J. Appl. Ecol. 46, 1080–1087 (2009).
Ashman, T.-L. et al. Pollen limitation of plant reproduction: ecological and evolutionary causes and consequences. Ecology 85, 2408–2421 (2004).
Larson, B. M. & Barrett, S. C. A comparative analysis of pollen limitation in flowering plants. Biol. J. Linn. Soc. 69, 503–520 (2000).
Caillaud, M. C. & Via, S. Specialized feeding behavior influences both ecological specialization and assortative mating in sympatric host races of pea aphids. Am. Naturalist 156, 606–621 (2000).
Hendry, A. P. & Kinnison, M. T. Perspective: the pace of modern life: measuring rates of contemporary microevolution. Evolution 53, 1637–1653 (1999).
Acknowledgements
We thank Laura Dällenbach, Franz Huber, Rayko Jonas, Markus Meierhofer, Luca Arrigo for their invaluable help with this study. Timotheus Van Der Niet provided helpful comments to an earlier version of the manuscript. The research was funded by the Swiss National Science Funds (SNF grant no. 31003A_172988 to F.P.S.).
Author information
Authors and Affiliations
Contributions
Conceptualization: F.P.S., T.D. Experimental: T.D. Analysis: T.D., F.P.S. Writing: F.P.S., T.D.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sevan Suni and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dorey, T., Schiestl, F.P. Bee-pollination promotes rapid divergent evolution in plants growing in different soils. Nat Commun 15, 2703 (2024). https://doi.org/10.1038/s41467-024-46841-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-46841-4
- Springer Nature Limited
This article is cited by
-
Biotic interactions promote local adaptation to soil in plants
Nature Communications (2024)