Abstract
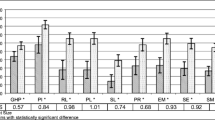
The Peyronie’s Disease Questionnaire (PDQ) is a 15-question self-reported questionnaire that evaluates the severity and physical and psychosexual issues of Peyronie’s disease (PD) symptoms in three scales: “psychological and physical symptoms,” “penile pain,” and “symptom bother.” Previous studies validated the PDQ US version and confirmed its test–retest reliability and responsiveness. The aim is to translate and validate the Spanish version of the PDQ to be used in the clinical practice and in PD research studies in Spain. A non-interventional, observational study with 160 PD patients was conducted. Patients included from four healthcare centers in Spain and completed the PDQ in two study visits separated for a period of 4–7 days from March 2018 to June 2019. Patients received no type of treatment or intervention. Different statistical tests were applied to the data in order to validate the structural and construct of the PDQ, as well as its internal reliability, temporal stability reliability, reliability between observers, and test–retest reliability. Cronbach’s alpha over 0.9 showed good internal consistency. We found an ICC agreement of 0.82 (test–retest) for the three scales of the Spanish version of the PDQ, which demonstrates good reliability. When comparing Visit 1 and Visit 2 questionnaires mean scores, the PDQ showed non-significant differences, as expected because no intervention or treatment was administered to the patients between visits. Translation and validation of the PDQ for the Spanish population makes available a valid, useful, and reliable tool to properly evaluate quality of life of men suffering PD.
Similar content being viewed by others
Change history
18 July 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41443-023-00734-0
References
Rhoden EL, Teloken C, Ting HY, Lucas ML, Teodósio da Ros C, Ary Vargas Souto C. Prevalence of Peyronie’s disease in men over 50-y-old from Southern Brazil. Int J Impot Res. 2001;13:291–3.
Ralph D, Gonzalez-Cadavid N, Mirone V, Perovic S, Sohn M, Usta M, et al. The management of Peyronie’s disease: evidence-based 2010 guidelines. J Sex Med. 2010;7:2359–74.
Arafa M, Eid H, El-Badry A, Ezz-Eldine K, Shamloul R. The prevalence of Peyronie’s disease in diabetic patients with erectile dysfunction. Int J Impot Res. 2007;19:213–7.
Peyronie’s Disease Guideline – American Urological Association. https://www.auanet.org/guidelines/peyronies-disease-guideline. Accessed 23 Sept 2020.
Gelbard M, Goldstein I, Hellstrom WJG, McMahon CG, Smith T, Tursi J, et al. Clinical efficacy, safety and tolerability of collagenase clostridium histolyticum for the treatment of peyronie disease in 2 large double-blind, randomized, placebo controlled phase 3 studies. J Urol. 2013;190:199–207.
Hellstrom WJG, Feldman R, Rosen RC, Smith T, Kaufman G, Tursi J. Bother and distress associated with Peyronie’s disease: validation of the Peyronie’s Disease Questionnaire. J Urol. 2013;190:627–34.
Bowling A. The principles of research. 2nd ed. Philadelphia: University Press; 2002. p. 133–62.
Coyne KS, Currie BM, Thompson CL, Smith TM. The test-retest reliability of the Peyronie’s Disease Questionnaire. J Sex Med. 2015;12:543–8.
Coyne KS, Currie BM, Thompson CL, Smith TM. Responsiveness of the Peyronie’s Disease Questionnaire (PDQ). J Sex Med. 2015;12:1072–9.
Smith JF, Walsh TJ, Conti SL, Turek P, Lue T. Risk factors for emotional and relationship problems in Peyronie’s disease. J Sex Med. 2008;5:2179–84.
Burri A, Porst H. The relationship between penile deformity, age, psychological bother, and erectile dysfunction in a sample of men with Peyronie’s Disease (PD). Int J Impot Res. 2018;30:171–8.
Moncada I, Krishnappa P, Romero J, Torremade J, Fraile A, Martinez-Salamanca JI, et al. Penile traction therapy with the new device «Penimaster PRO» is effective and safe in the stable phase of Peyronie’s disease: a controlled multicentre study. BJU Int. 2019;123:694–702.
Acknowledgements
This project has been proposed from the Fundación para la Investigación de Urología (FIU), part of the Spanish Association of Urology, and thanks to a grant from Sobi Spain (Swedish Orphan Biovitrum, SL, Madrid, Spain).
Author information
Authors and Affiliations
Contributions
BGG, JIMS and JRO conceived the paper. BGG, JSG, JJQ, and JRO developed the methodology. BGG, BGG, JIMS, JSG, JJQ, EGR, EFP and ERC collected and validated the data. EGR did the statistical analysis. BGG wrote the original manuscript and BGG, JIMS, JSG and EFP reviewed it and collaborated in rewriting. JRO supervised and validated the process.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: In this article the affiliation details for Author Juan Ignacio Martínez-Salamanca and Esaú Fernández-Pascual were incorrectly given as ‘Lyx Instituto de Urología, Madrid, Spain. 3’ but should have been ‘Lyx Instituto de Urología, Universidad Francisco de Vitoria, Madrid, Spain. 3’.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
García-Gómez, B., Martínez-Salamanca, J.I., Sarquella-Geli, J. et al. Peyronie’s Disease Questionnaire (PDQ): Spanish translation and validation. Int J Impot Res 34, 501–504 (2022). https://doi.org/10.1038/s41443-021-00460-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41443-021-00460-5
- Springer Nature Limited
This article is cited by
-
Validation of the Italian version of the Peyronie’s Disease Questionnaire (PDQ)
International Journal of Impotence Research (2023)