Abstract
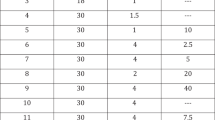
To investigate the efficacy, tolerability, and patient’s preference of alprostadil cream for topical use administered within the urethral meatus versus the standard administration route, in erectile dysfunction (ED) treatment. Seventy-one patients (mean age 59.7 ± 9.0 years) affected by ED were analyzed in this multicenter, randomized, two-administration routes, cross-over trial. All patients received a single dose of alprostadil cream applying the dispenser to the tip of the penis (without contacting the urethral meatus) (Standard administration route or ST.AR) alternating with a single dose of alprostadil cream applying the dispenser within the urethral meatus (New administration route or NEW.AR) separated by a one-week washout period, according to randomization. The primary objective of the study was to evaluate the change in International Index of Erectile Function (IIEF-5) total score from baseline to the control visit by comparing the ST.AR and NEW.AR. Secondary objectives of the study were to compare the different methods of administration by evaluating the change in the Sexual Encounter Profile (SEP-2 and SEP-3) questionnaire score and the Patient Reported Outcomes (PROs) by scoring the Patient Self-Assessment of Erection (PSAE) questionnaire. The treatment safety profile was assessed by analysis of adverse events (AEs). Based on the study findings it is evident that the NEW.AR is more efficacious than the ST.AR in improving IIEF-5 and SEP scores from baseline to control visit (IIEF-5: +3.8 vs +6.3; p < 0.001; positive response to SEP-2: 10 vs 27; p = 0.002) and in terms of PSAE (a significant improvement from the baseline in 31% of patients; p < 0.001). As regards the safety profile, no difference in terms of local and systemic side effects was found.


Similar content being viewed by others
References
Braun M, Wassmer G, Klotz T, Reifenrath B, Mathers M, Engelmann U. Epidemiology of erectile dysfunction: results of the Cologne Male Survey. Int J Impot Res. 2000;12:305–11.
Liu LH, Zhang T, Zhang YR, Liu TS, Zhang HB, Chen FZ, et al. Metabolic syndrome and risk for ED: a meta-analysis. Int J Impot Res. 2014;26:196–200.
NIH Consensus Development Panel on Impotence. JAMA.1993;270:83–90.
Latini DM, Penson DF, Lubeck DP, Wallace KL, Henning JM, Lue TF. Longitudinal differences in disease specific quality of life in men with erectile dysfunction: results from the exploratory comprehensive evaluation of erectile dysfunction study. J Urol. 2003;169:1437–42.
Hatzimouratidis K (Chair), Giuliano F, Moncada I, Muneer A, Salonia A (Vice-chair), Verze P. EAU Guidelines on Erectile Dysfunction, Premature Ejaculation, Penile Curvature and Priapism http://uroweb.org/guideline/male-sexual-dysfunction/ UPDATE MARCH 2018
Carvalheira AA, Pereira NM, Maroco J, Forjaz V. Dropout in the treatment of erectile dysfunction with PDE5: a study on predictors and a qualitative analysis of reasons for discontinuation. J Sex Med. 2012;9:2361–9.
El-Sakka AI. Intracavernosal prostaglandin E1 self vs office injection therapy in patients with erectile dysfunction. Int J Impot Res. 2006;18:180–5.
Jannini EA, Sternbach N, Limoncin E, Ciocca G, Gravina GL, Tripodi F, et al. Health-related characteristics and unmet needs of men with erectile dysfunction: a survey in five European countries. J Sex Med. 2014;11:40–50.
Rooney M, Pfister W, Mahoney M, Nelson M, Yeager J, Steidle C. Long-term, Multicenter Study of the Safety and Efficacy of Topical Alprostadil Cream in Male Patients with Erectile Dysfunction. J Sex Med. 2009;6:520–34.
Padma-Nathan H, Steidle C, Salem S, Tayse N, Yeager J, Harning R. The efficacy and safety of a topical alprostadil cream, Alprox-TD, for the treatment of erectile dysfunction: two phase 2 studies in mild-to-moderate and severe ED. Int J Impot Res. 2003;15:10–17.
Steidle C, Padma-Nathan H, Salem S, Tayse N, Thwing D, Fendl J, et al. Topical alprostadil cream for the treatment of erectile dysfunction: a combined analysis of the phase II program. Urology. 2002;60:1077–82.
Padma-Nathan H, Yeager JL. An integrated analysis of alprostadil topical cream for the treatment of erectile dysfunction in 1732 patients. Urology. 2006;68:386–91.
Moncada I, Cuzin B. Clinical efficacy and safety of Vitaros©/Virirec© (Alprostadil cream) for the treatment of erectile dysfunction. Urologia. 2015;82:84–92.
D’Elia C, Cerruto MA, Cavicchioli FM, Cardarelli S, Molinari A, Artibani W. Critical points in understanding the Italian version of the IIEF 5 questionnaire. Arch Ital Urol Androl. 2012;84:197–201.
Araujo AB, Allen KR, Ni X, Rosen RC. Minimal clinically important differences in the vaginal insertion and successful intercourse items of the sexual encounter profile. J Sex Med. 2012;9:169–79.
Cappelleri JC, Stecher VJ. An assessment of patient-reported outcomes for men with erectile dysfunction: Pfizer’s perspective. Int J Impot Res. 2008;20:343–57.
Machin D, Campbell MJ, Fayers P, Pinol A. Statistical tables for the design of clinical studies. edn. 3. Oxford, UK: Blackwell; 1998.
Kim ED, McVary KT. Topical prostaglandin-E1 for the treatment of erectile dysfunction. J Urol. 1995;153:1828–30.
Montorsi F, Guazzoni C, Barbieri L, et al. Clinical and hemodynamic effects of transdermal alprostadil for mild arteriogenic impotence: a double-blind placebo controlled study. Int J Impot Res. 1995;7:10–11.
Goldstein I, Payton TR, Schechter PJ. A double-blind, placebo-controlled, efficacy and safety study of topical gel formulation of 1% alprostadil (Topiglan) for the in-office treatment of erectile dysfunction. Urology. 2001;57:301–5.
Campbell HE. Clinical monograph for drug formulary review: erectile dysfunction agents. J Manag Care Pharm. 2005;11:151–71.
Hanchanale V, Eardley I. Alprostadil for the treatment of impotence. Expert Opin Pharmacother. 2014;15:421–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Cai, T., Palumbo, F., Liguori, G. et al. The intra-meatal application of alprostadil cream (Vitaros®) improves drug efficacy and patient’s satisfaction: results from a randomized, two-administration route, cross-over clinical trial. Int J Impot Res 31, 119–125 (2019). https://doi.org/10.1038/s41443-018-0087-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41443-018-0087-6
- Springer Nature Limited
This article is cited by
-
Conservative Non-surgical Options for Erectile Dysfunction
Current Urology Reports (2023)
-
The Italian Society of Andrology and Sexual Medicine (SIAMS), along with ten other Italian Scientific Societies, guidelines on the diagnosis and management of erectile dysfunction
Journal of Endocrinological Investigation (2023)
-
Combination therapy with topical alprostadil and phosphodiesterase-5 inhibitors after failure of oral therapy in patients with erectile dysfunction: a prospective, two-arm, open-label, non-randomized study
International Journal of Impotence Research (2022)
-
Hard flaccid syndrome: initial report of four cases
International Journal of Impotence Research (2020)