Abstract
Aim
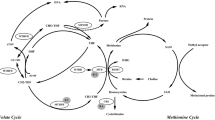
Age and lower folate concentrations are well-known risk factors for cardiovascular disease (CVD), but the potential roles of age and folate deficiency in hyperhomocysteinemia (HHcy), especially in HHcy patients with abnormal methylation levels of key enzyme genes promoter in homocysteinemia (Hcy) pathway, have not been thoroughly evaluated. The purpose of this study was to evaluate the relationship between the promoter methylation levels of six key enzyme genes and age and serum folate level to better understand the pathogenesis of HHcy.
Methods
In 299 HHcy patients, six key enzyme genes promoter methylation was analyzed by PCR amplification and MethylTargetTM methods.
Results
The betaine homocysteine methyltransferase (BHMT), Cystathionine β-synthase (CBS), and Methionine synthasegene (MTR) promoter methylation levels were positively associated with age and a negative correlation was found between CBS promoter methylation level and folate levels. However, these associations were not significant after Bonferroni correction. The stratified analysis showed that the methylation level of CBS gene promoter was positively correlated with age in males, and a positive correlation was also found between BHMT gene promoter methylation level and age in HHcy patients with a history of diabetes or hypertension. Moreover, stratified analysis according to sex revealed that the methylation levels of three CpG regions of BHMT_2, CBS_2, and CBS_3 were positively correlated with age in males after Bonferroni correction.
Conclusions
Our data suggested that age and folate deficiency may increase the risk of HHcy by mediating methylation of the promoter regions of key enzyme genes in the one-carbon metabolism pathway.


Similar content being viewed by others
References
Wang Y, Jiang Y, Wang N, Zhu M, Liu X, Wang R, et al. Central but not general obesity is positively associated with the risk of hyperhomocysteinemia in middle-aged women. Nutrients. 2019;11:1614.
Mishra PK, Tyagi N, Sen U, Joshua IG, Tyagi SC. Synergism in hyperhomocysteinemia and diabetes: role of PPAR gamma and tempol. Cardiovasc Diabetol. 2010;9:49.
Sharma M, Tiwari M, Tiwari RK. Hyperhomocysteinemia: impact on neurodegenerative diseases. Basic Clin Pharmacol Toxicol. 2015;117:287–96.
Gaiday AN, Tussupkaliyev AB, Bermagambetova SK, Zhumagulova SS, Sarsembayeva LK, Dossimbetova MB, et al. Effect of homocysteine on pregnancy: a systematic review. Chem Biol Interact. 2018;293:70–6.
Yang B, Fan S, Zhi X, Wang Y, Wang Y, Zheng Q. et al. Prevalence of hyperhomocysteinemia in China: a systematic review and meta-analysis. Nutrients. 2014;7:74–90.
Gonzalez-Gross M, Benser J, Breidenassel C, Albers U, Huybrechts I, Valtuena J. et al. Gender and age influence blood folate, vitamin B12, vitamin B6, and homocysteine levels in European adolescents: the Helena Study. Nutr Res. 2012;32:817–26.
Sakuta H, Suzuki T. Alcohol consumption and plasma homocysteine. Alcohol. 2005;37:73–7.
de Bree A, Verschuren WM, Blom HJ, Kromhout D. Lifestyle factors and plasma homocysteine concentrations in a general population sample. AM J Epidemiol. 2001;154:150–4.
Taguchi T, Mori H, Hamada A, Yamori Y, Mori M. Serum folate, total homocysteine levels and methylenetetrahydrofolate reductase 677C>T polymorphism in young healthy female Japanese. Asia Pac J Clin Nutr. 2012;21:291–5.
Du B, Tian H, Tian D, Zhang C, Wang W, Wang L. et al. Genetic polymorphisms of key enzymes in folate metabolism affect the efficacy of folate therapy in patients with hyperhomocysteinaemia. Br J Nutr. 2018;119:887–95.
Deng Q, Huang W, Peng C, Gao J, Li Z, Qiu X. et al. Genomic 5-mC contents in peripheral blood leukocytes were independent protective factors for coronary artery disease with a specific profile in different leukocyte subtypes. Clin Epigenet. 2018;10:9.
Illingworth RS, Gruenewald-Schneider U, Webb S, Kerr AR, James KD, Turner DJ. et al. Orphan CpG islands identify numerous conserved promoters in the mammalian genome. PLOS Genet. 2010;6:e1001134.
Bellacosa A, Drohat AC. Role of base excision repair in maintaining the genetic and epigenetic integrity of CpG sites. DNA Repair (Amst). 2015;32:33–42.
Wong KY, Chim CS. DNA methylation of tumor suppressor protein-coding and non-coding genes in multiple myeloma. Epigenomics-Uk. 2015;7:985–1001.
Sansom OJ, Maddison K, Clarke AR. Mechanisms of disease: methyl-binding domain proteins as potential therapeutic targets in cancer. Nat Clin Pr Oncol. 2007;4:305–15.
Menafra R, Stunnenberg HG. MBD2 and MBD3: elusive functions and mechanisms. FRONT Genet. 2014;5:428.
Ehrlich M, Lacey M. DNA methylation and differentiation: silencing, upregulation and modulation of gene expression. Epigenomics-Uk. 2013;5:553–68.
Mann DA. Epigenetics in liver disease. Hepatology. 2014;60:1418–25.
Ozanne SE, Constancia M.Mechanisms of disease: the developmental origins of disease and the role of the epigenotype.Nat Clin Pract Endocrinol Metab. 2007;3:539–46.
Singh N, Rashid S, Rashid S, Dash NR, Gupta S, Saraya A. Clinical significance of promoter methylation status of tumor suppressor genes in circulating DNA of pancreatic cancer patients. J Cancer Res Clin Oncol. 2020;146:897–907.
Wang C, Xu G, Wen Q, Peng X, Chen H, Zhang J, et al. CBS promoter hypermethylation increases the risk of hypertension and stroke. Clin (Sao Paulo). 2019;74:e630.
Yao Y, Gao LJ, Zhou Y, Zhao JH, Lv Q, Dong JZ, et al. Effect of advanced age on plasma homocysteine levels and its association with ischemic stroke in non-valvular atrial fibrillation. J Geriatr Cardiol. 2017;14:743–9.
Jud P, Hafner F, Verheyen N, Gary T, Meinitzer A, Brodmann M, et al. Age-dependent effects of homocysteine and dimethylarginines on cardiovascular mortality in claudicant patients with lower extremity arterial disease. Heart Vessels. 2018;33:1453–62.
Xie W, Kagiampakis I, Pan L, Zhang YW, Murphy L, Tao Y. et al. DNA methylation patterns separate senescence from transformation potential and indicate cancer risk. Cancer Cell. 2018;33:309–21.
Fan R, Mao SQ, Gu TL, Zhong FD, Gong ML, Hao LM, et al. Preliminary analysis of the association between methylation of the ACE2 promoter and essential hypertension. Mol Med Rep. 2017;15:3905–11.
Jiang D, Zheng D, Wang L, Huang Y, Liu H, Xu L. et al. Elevated PLA2G7 gene promoter methylation as a gender-specific marker of aging increases the risk of coronary heart disease in females. PLoS ONE. 2013;8:e59752.
Dong Y, Yi X, Yujie Z, Huixia Z, Yan C. Relationship between the Methylation of Folic Acid Metabolism-related Genes and the Incidence and Prognosis of Esophageal Cancer among Ethnic Kazakhs. J Cancer. 2018;9:2865–75.
Zhang Z, He Y, Tu X, Huang S, Chen Z, Wang L, et al. Mapping of DNA hypermethylation and hypomethylation induced by folate deficiency in sporadic colorectal cancer and clinical implication analysis of hypermethylation pattern in CBS promoter. Clin Lab. 2017;63:733–48.
Andersson A, Brattstrom L, Israelsson B, Isaksson A, Hamfelt A, Hultberg B. Plasma homocysteine before and after methionine loading with regard to age, gender, and menopausal status. Eur J Clin Investig. 1992;22:79–87.
Jacobsen DW, Gatautis VJ, Green R, Robinson K, Savon SR, Secic M. et al. Rapid HPLC determination of total homocysteine and other thiols in serum and plasma: sex differences and correlation with cobalamin and folate concentrations in healthy subjects. Clin Chem. 1994;40:873–81.
Vitvitsky V, Prudova A, Stabler S, Dayal S, Lentz SR, Banerjee R. Testosterone regulation of renal cystathionine beta-synthase: implications for sex-dependent differences in plasma homocysteine levels. Am J Physiol Renal Physiol. 2007;293:F594–600.
Giltay EJ, Hoogeveen EK, Elbers JM, Gooren LJ, Asscheman H, Stehouwer CD. Effects of sex steroids on plasma total homocysteine levels: a study in transsexual males and females. J Clin Endocrinol Metab. 1998;83:550–3.
Acknowledgements
This project was funded by the Department of Science and Technology of Henan Province (no. 132102310431). The funder did not participate in the design, analysis or writing, or approval of the paper. All authors thank the staff from the Department of Neurology, the Fifth Affiliated Hospital of Zhengzhou University, for their assistance and support.
Author information
Authors and Affiliations
Contributions
All authors’ responsibilities were as follows: XH contributed to the writing of this paper. QZ and DL were involved in the study concept and design. BR, LY, FS, and XW acquired data of this study. CZ, XC and CZ carried out data analysis and interpretation. WZ was in charge of study supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, X., Zhao, Q., Li, D. et al. Association between gene promoter methylation of the one-carbon metabolism pathway and serum folate among patients with hyperhomocysteinemia. Eur J Clin Nutr 74, 1677–1684 (2020). https://doi.org/10.1038/s41430-020-0657-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-020-0657-9
- Springer Nature Limited
This article is cited by
-
Using the optimal method—explained variance weighted genetic risk score to predict the efficacy of folic acid therapy to hyperhomocysteinemia
European Journal of Clinical Nutrition (2022)
-
Combining genetic risk score with artificial neural network to predict the efficacy of folic acid therapy to hyperhomocysteinemia
Scientific Reports (2021)
-
The regulation mechanisms and the Lamarckian inheritance property of DNA methylation in animals
Mammalian Genome (2021)