Abstract
Among pediatric malignancies, solid tumors, particularly within the central nervous system (CNS), are common. Thiotepa, a myeloablative, high-dose chemotherapeutic (HDT) treatment administered prior to autologous hematopoietic stem cell transplantation (HSCT), can cross the blood-brain barrier and rapidly penetrate the CNS. We evaluated thiotepa HDT in conjunction with melphalan in Japanese patients with pediatric CNS/non-CNS solid tumors in a multicenter, open-label, non-comparative study. Thiotepa (200 mg/m2/day) was administered intravenously (IV) over 24 h on days −12, −11, −5, and −4 before scheduled HSCT. Melphalan (70 mg/m2/day) was administered IV over 1 h on days −11, −5, and −4. The safety analysis population comprised 41 patients, of whom 16 (39.0%) had solid tumors and 25 (61.0%) had brain tumors. The most frequently reported adverse events were diarrhea (40/41 [97.6%] patients) and febrile neutropenia (34/41 [82.9%]). No unexpected safety events were observed, and no events resulted in death or treatment discontinuation. All patients experienced bone marrow suppression and 39/41 (95.1%) achieved engraftment (neutrophil count ≥500/mm3 for 3 consecutive days after HSCT). The survival rate at day 100 post-autologous HSCT was 100%. These data confirm the safety of IV thiotepa plus melphalan HDT prior to autologous HSCT for patients with pediatric CNS/non-CNS solid tumors. Trial registration: JapicCTI-173654.
Similar content being viewed by others
Introduction
Globally, cancer is a major cause of childhood mortality, and incidence rates appear to be increasing over time [1]. Around 2500 children are newly affected by cancer every year in Japan [2], corresponding to an incidence of 1.23/million for ages 0–14 years and 142/million for ages 15–19 years [3].
After leukemia, solid tumors, particularly those occurring within the central nervous system (CNS) are the most common pediatric cancers [4]. In 2015, 904 children with solid tumors were newly registered to the Japanese Society of Pediatric Hematology/Oncology database [5]. High-dose chemotherapy (HDT) and hematopoietic stem cell transplantation (HSCT) allows intensive treatment of such malignancies [6]. Thus, doses of cytotoxic therapies can be escalated beyond marrow tolerance. It is estimated that in Japan, 3 323 children <16 years underwent autologous HSCT for pediatric solid tumors between 1991–2018 [7].
Thiotepa is a myeloablative HDT treatment administered prior to autologous HSCT to treat solid tumors and hematological malignancies [8]. It is an antitumor alkylating agent belonging to the ethyleneimine group, and inhibits DNA synthesis [9]. Importantly, it has the capacity to cross the blood-brain barrier and penetrate rapidly into the CNS, producing concentrations in the cerebrospinal fluid >90% of those observed in serum [8, 10]. Thiotepa has been in clinical use since the 1950s [11, 12], and was approved for standard-dose chemotherapy in Japan in 1958. Its use as HDT prior to HSCT was approved in Europe in 2010 [13]. However, thiotepa was not available for this purpose in Japan, despite the critical medical need, because manufacturing had been discontinued in 2008 [14, 15].
A recent phase I study established the pharmacokinetics of thiotepa as HDT with autologous HSCT in a Japanese population of nine pediatric and 10 adult patients [8]. Treatment was well tolerated and survival rates were high (77.8% for pediatric solid tumors and 100% for malignant lymphoma) [8]. Thiotepa is now approved in Japan as HDT before autologous HSCT for pediatric malignant solid tumors [16, 17].
An expanded access program, including patients with pediatric solid tumors or brain tumors, and patients with malignant lymphoma, was initiated to further evaluate the safety and efficacy of thiotepa as HDT before autologous HSCT. We report here the data from the population with pediatric solid tumors or brain tumors.
Patients and methods
Patients
This expanded access study included two groups of patients scheduled to undergo autologous HSCT. This report focuses on patients with pediatric solid tumors or brain tumors; data from the group of patients with malignant lymphoma are reported elsewhere [18].
The key inclusion criteria were patients aged ≥2 years with solid tumors or brain tumors who had completed hematopoietic cell collection for autologous HSCT; Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–2 assessed within 14 days before enrollment; negative pregnancy test, and willingness and ability to use appropriate contraception until 90 days after the end of study treatment (if age appropriate); and normal hepatic, renal, and cardiac function based on tests performed within 14 days before enrollment. In addition, the estimated glomerular filtration rate (eGFR), calculated using the levels of serum creatinine or cystatin C measured within 14 days before enrollment was required to be ≥60 ml/min/1.73 m2 (patients aged ≥18 years), or ≥100 ml/min/1.73 m2 (patients aged <18 years). Each patient (for those aged ≥20 years) and/or their legal representative (for those aged <20 years) provided written informed consent prior to study enrollment.
Key exclusion criteria were patients who had undergone any treatment (other than hematopoietic cell collection) for the underlying disease within 13 days before the start of study treatment; previous HSCT within the 6 months prior to the study; receipt of live attenuated vaccine within 90 days or any investigational agent within 27 days before the start of study treatment; pregnancy or lactation; present or previous history of complications affecting drug metabolism or excretion; active infection; presence of hepatitis B surface antigen or antibody, hepatitis B core antibody, or human immunodeficiency virus antibody (although patients positive for hepatitis B surface antigen or antibody, or hepatitis B core antibody could be enrolled if vaccinated for type B hepatitis); uncontrolled intercurrent illness; hypersensitivity to the study drugs or their excipients; and any other reason which may endanger the patient or confound the study outcomes based on the judgment of the study investigator. Patients who were considered likely to have difficulty in receiving an adequate volume of replacement fluid and frequent blood transfusions (often required in association with concomitant melphalan) were also excluded.
Study design and treatments
This multicenter, open-label, non-comparative, expanded access program (JapicCTI-173654) was conducted between September 2017 and June 2020 at eight sites in Japan (of which four enrolled patients with malignant lymphoma and four enrolled patients with pediatric solid tumors). Following the approval of thiotepa in Japan as HDT before autologous HSCT for pediatric malignant solid tumors (on March 26, 2019), the study continued as a post-marketing clinical study. The study protocol and related documentation were approved by the ethics committee or independent review board of each study center. The study was conducted in accordance with the Declaration of Helsinki, good clinical practice guidelines, and all applicable legal and regulatory requirements.
The study design is shown in Fig. 1. The study comprised a HDT period and a transplant period, with the day of HSCT defined as day 0. Thiotepa (200 mg/m2/day) was administered intravenously (IV) over 24 h on days −12, −11, −5, and −4 before scheduled HSCT. Commercially available melphalan (70 mg/m2/day) was administered IV over 1 h on days −11, −5, and −4 before scheduled HSCT. Melphalan was chosen for the combination treatment regimen in both the phase I study [8] and the current expanded access program based on its regulatory approval status in Japan for pretreatment prior to autologous HSCT in patients with pediatric solid tumors, and its widespread domestic use for this purpose. The doses of thiotepa and melphalan could be reduced or interrupted if deemed necessary by the investigator. Both drugs were to be discontinued if eGFR was <45 ml/min/1.73 m2 (patients aged ≥18 years) or <75 ml/min/1.73 m2 (patients aged <18 years) on day −7 before scheduled HSCT (i.e., scheduled treatment administration on days −5 and −4 did not occur under these circumstances).
Prohibited concomitant medications and therapies during the study period included any cancer therapy (other than study treatments), any other investigational drugs, and live vaccines.
For these patients with pediatric solid tumors or brain tumors, skin management was recommended to avoid severe dermatologic toxicity such as skin peeling. In addition, as patients were receiving concomitant melphalan, replacement fluid (≥2000 ml/day) and diuretics were supplied to ensure adequate urine volume (≥100 ml/h). Volumes of replacement fluid could be adjusted depending on the age and condition of the patient.
Endpoints
The primary study objective was to assess the safety of IV thiotepa HDT in combination with melphalan before autologous HSCT in patients with pediatric solid tumors or brain tumors. Safety was assessed by recording treatment-emergent adverse events (TEAEs) and adverse drug reactions occurring between the start of study treatment and day 28 post HSCT. TEAEs were classified using the Medical Dictionary for Regulatory Activities, version 19.1. Ascertainment of causality was undertaken solely by individual treating investigators, without study monitoring or review/modification by the primary investigators. Additional safety evaluations included ECOG PS at each visit, and physical and laboratory test results, including 12-lead electrocardiogram, left ventricular ejection fraction, laboratory measures, vital signs, and weight.
The secondary objective was to assess the clinical outcomes following the use of IV thiotepa in this patient population. Endpoints included the bone marrow suppression rate (defined as the proportion of patients with a neutrophil count <500/mm3 at least once during the 28 days after HSCT), the engraftment rate (defined as the proportion of patients with a neutrophil count ≥500/mm3 for 3 consecutive days after bone marrow suppression and HSCT), time to engraftment (defined as the number of days between HSCT and the first of three consecutive days with a neutrophil count ≥500/mm3 after bone marrow suppression and HSCT), and the survival rate at day 100 post-HSCT.
Statistical methods
No formal study size calculations or hypothesis testing was performed. The overall sample size for the expanded access program was approximately 100 patients, comprising both those with pediatric solid tumors or brain tumors (reported herein) plus adults with malignant lymphoma (reported elsewhere [18]) based on the expected number of participants.
The safety analysis population included all patients who received at least a single dose of thiotepa. The number and frequency of adverse events and adverse drug reactions were summarized; physical and laboratory test results were reported as summary statistics and electrocardiogram interpretations as shift tables.
For the efficacy analyses of bone marrow suppression rate, engraftment rate and time to engraftment, missing data were not imputed. The rate of survival at day 100 post-HSCT was estimated using Kaplan–Meier methodology. Death after HSCT (regardless of cause of death) was defined as an event, and observations were censored on the latest date of confirmed survival. Statistical analyses were conducted using SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patients
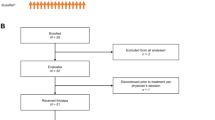
A total of 41 patients with pediatric solid tumors or brain tumors were enrolled into the study. Of these, 38 were enrolled during the expanded access phase and three during the post-marketing phase; data from all 41 patients were summarized together. All patients received thiotepa and were included in the safety analysis population. All 41 patients underwent peripheral blood HSCT.
Baseline demographics and clinical characteristics are shown in Table 1. Overall, 22/41 (53.7%) patients were female, the median age was 4 years, and 31/41 (75.6%) patients were aged between ≥2 and <12 years. Most patients (30/41 [73.2%]) had an ECOG PS of 0. A total of 16/41 (39.0%) patients had a pediatric non-CNS solid tumor, of which the most common was neuroblastoma (5/41 [12.2%]), and 25/41 (61.0%) had a pediatric brain tumor, of which the most common was medulloblastoma (12/41 [29.3%]). Tumors were newly diagnosed in 20/41 (48.8%) patients and relapsed in 21/41 (51.2%) patients; 4/41 (9.8%) patients had received one prior HSCT transplantation and 2/41 (4.9%) had received two or more.
Treatments
Dosing frequency and dose administered for thiotepa and melphalan are shown in Supplementary Table S1. All patients (100%) received at least three doses of thiotepa and 39 (95.1%) received all four scheduled doses. All patients (100%) received at least two doses of melphalan and 32 (78.0%) received all three scheduled doses.
Safety
TEAEs occurring in ≥10% of patients are shown in Table 2. The most frequently reported TEAEs (in ≥50% of patients) were diarrhea (40/41 [97.6%]), febrile neutropenia (34/41 [82.9%]), vomiting (31/41 [75.6%]), stomatitis (26/41 [63.4%]), and nausea (21/41 [51.2%]). Grade III febrile neutropenia was reported in 34/41 (82.9%) patients. The majority of other TEAEs were grade I or II in intensity.
No TEAE resulting in death or treatment discontinuation/dose reduction was reported during the study. TEAEs requiring treatment interruption were observed in 2/41 (4.9%) patients. One patient had an event of bacteremia (this developed on HSCT day −9 and recovered on day 16 post-HSCT) and the other presented with herpes zoster (this developed on day −6 and recovered on day 14 post-HSCT); neither event was considered to be related to the study treatment. In both patients, the scheduled thiotepa dose on day −5 was missed although the other scheduled doses were administered.
Three patients reported serious TEAEs (sepsis grade IV, n = 1; tumor hemorrhage grade III, n = 1; bacterial enteritis grade IV, n = 1). The sepsis was considered unrelated to the study treatment (it was thought to be associated with neutropenia), while the other two events were considered possibly related to treatment. All events were reported as resolved/recovered at the end of the study.
ECOG PS at day 28 post-HSCT remained at 0 in 27 patients (65.9%), and was 1, 2, and 3 in 11 (26.8%), 2 (4.9%), and 1 (2.4%), respectively. No clinically significant change in left ventricular ejection fraction, vital signs or weight was noted during the study.
Efficacy
Efficacy outcomes are summarized in Table 3. All 41 patients (100.0%) experienced bone marrow suppression and 39/41 (95.1%) achieved engraftment. The median time to engraftment in those 39 patients was 11.0 days. The remaining two patients did not meet the narrow definition of engraftment used in this study (neutrophil count ≥500/mm3 for 3 consecutive days), but both patients did achieve a neutrophil count ≥500/mm3 on multiple non-consecutive days after autologous HSCT (Supplementary Table S2). The median (range) follow-up after autologous HSCT was 101.0 (100–129) days. The survival rate in evaluable patients at day 100 post-HSCT was 100% (Supplementary Fig. 1).
Discussion
In this expanded access study, we evaluated the use of thiotepa HDT, in conjunction with melphalan, in Japanese patients with pediatric solid and brain tumors who underwent autologous HSCT. The treatment regimen was found to be tolerable. No unexpected safety outcomes were reported, and there were no discontinuations or deaths due to TEAEs during the study.
The most frequently reported TEAEs in this study were gastrointestinal toxicity and febrile neutropenia, which are commonly associated with chemotherapeutic treatment of pediatric patients [19,20,21,22]. The data were also consistent with those reported in pediatric patients with solid tumors in the phase I study [8], and with the European product label [13] and Japanese package insert [17]. Events of veno-occlusive disease of the liver [23], thrombotic microangiopathy [24], and neurotoxic complications [25, 26], which are commonly reported during pre-HSCT treatment, were not observed in this study population.
In early studies of thiotepa as HDT with autologous HSCT, several problematic and/or dose-limiting toxicities were reported, including mucositis and neurotoxicity [27, 28]. Various regimens have since been evaluated, using combinations of thiotepa plus other alkylating agents, such as melphalan and busulfan [29, 30], in an attempt to increase the dose intensity while minimizing the potential for TEAEs. Nonetheless, toxicities remained problematic with many regimens. In the Head Start study, 37 children with malignant brain tumors received carboplatin, thiotepa, and etoposide as HDT, of whom 3 (8%) died of treatment-related complications [31]; however, this rate decreased in the subsequent Head Start II and III trials, with the reduction attributed by the authors to clinician experience with regimen administration and improved procedural and supportive care [32]. In a French study of 116 children who received a busulfan-thiotepa dual HDT regimen, 31% developed veno-occlusive disease [33]. The treatment regimen used in the current study was slightly different from that used in the most recent prior report [30], and decreased the melphalan dose to three-quarters of the prior dose (no infusion on Day -12). This regimen appears to achieve the aim of high myeloablative exposure with few problematic toxicities. Thus, patients in this study experienced a low frequency of pulmonary toxicity and veno-occlusive disease of the liver, which were a common occurrence in HDT regimens containing busulfan, and few events of nephrotoxicity which were previously reported to be associated with HDT regimens including carboplatin, etoposide, and melphalan [34, 35]. Importantly, no patients died due to treatment-related complications. Thus, while we cannot directly compare different thiotepa-containing HDT regimens, we anticipate that the regimen used herein may be less toxic for patients.
Although no renal dysfunction was noted under clinical trial conditions, patients need to be carefully monitored in routine clinical practice, as renal failure has been reported in association with thiotepa [29, 36]. To date, the effects of thiotepa in patients with renal insufficiency have not been assessed, but caution and careful monitoring should be used in patients with a history of renal disease [13].
Clinical efficacy outcomes were positive. All patients in the study experienced bone marrow suppression and all survived for 100 days post-HSCT. The type of solid tumor (brain or other) and number of prior HSCT procedures did not affect the results, and >95% of patients achieved engraftment.
Although radiotherapy is commonly administered to adult patients, irradiation for pediatric CNS tumors comes with an increased risk of later developmental adverse events [37,38,39]. A recent focus of research has been the use of HDT in combination with radiotherapy to reduce the irradiation exposure necessary for an adequate antitumor effect [40, 41]; the outcomes observed with such combination regimens have been positive. However, HDT agents which are able to transition into the CNS, such as thiotepa, are likely to provide more clinical benefit in this regard. Furthermore, the observation that children tend to tolerate HDT better than adults [42], and the improved outcomes for pediatric primary malignant CNS tumors with the use of HDT and autologous HSCT reported from the ‘Head Start’ trials [31, 43, 44], has led to the administration of HDT plus autologous HSCT as a potential curative option for patients with high-risk disease. Regimens incorporating two alkylating agents are commonly used as HDT prior to HSCT in other malignancies, and have been reported to provide enhanced clinical benefit compared with a single drug [45, 46]. Our results confirm the safety and efficacy data observed in the prior phase I study [8], and thiotepa in conjunction with melphalan appears to be a useful addition to the treatment armamentarium for HDT prior to autologous HSCT in Japanese patients with pediatric solid CNS/non-CNS tumors. Notably, the penetrative ability of thiotepa into the CNS [8, 10] is a key facet supporting its use for this indication.
There are some study limitations that should be considered when evaluating these data. The open-label design and small size of the study restrict the conclusions that can be drawn, and the enrollment of only Asian patients may preclude the extrapolation of the results to the global patient population. Finally, further studies will be necessary to evaluate outcomes over longer durations, as we did not examine long-term survival beyond the first 100 days post-HSCT.
In conclusion, we have confirmed the safety of IV thiotepa HDT, in conjunction with melphalan, prior to autologous HSCT for patients with pediatric solid or brain tumors, with no new toxicity concerns. We consider that thiotepa is a suitable HDT agent for this patient population.
Data availability
The research data underlying this study are subject to restrictions and cannot be shared.
Change history
08 December 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41409-022-01890-5
References
Steliarova-Foucher E, Colombet M, Ries LAG, Moreno F, Dolya A, Bray F, et al. International incidence of childhood cancer, 2001-10: a population-based registry study. Lancet Oncol. 2017;18:719–31. https://doi.org/10.1016/S1470-2045(17)30186-9
Japanese Society of Pediatric Hematology/Oncology. Practical guidelines for pediatric cancer [In Japanese]. 2016. http://27.34.141.111/journal_en/guideline.html
Katanoda K, Shibata A, Matsuda T, Hori M, Nakata K, Narita Y, et al. Childhood, adolescent and young adult cancer incidence in Japan in 2009-2011. Jpn J Clin Oncol. 2017;47:762–71. https://doi.org/10.1093/jjco/hyx070
GBD Childhood Cancer Collaborators. The global burden of childhood and adolescent cancer in 2017: an analysis of the Global Burden of Disease Study 2017. Lancet Oncol. 2019;20:1211–25. https://doi.org/10.1016/S1470-2045(19)30339-0
Japanese Society of Pediatric Hematology/Oncology. Number of registered solid tumors by institution (2015), aggregation results of disease registration. 2015. http://27.34.141.112/disease_record.html
Barrett D, Fish JD, Grupp SA. Autologous and allogeneic cellular therapies for high-risk pediatric solid tumors. Pediatr Clin N Am. 2010;57:47–66. https://doi.org/10.1016/j.pcl.2010.01.001
Japanese Data Center for Hematopoietic Cell Transplantation/Japan Society for Hematopoietic Cell Transplantation. Hematopoietic cell transplantation in Japan, National Survey Report. 2019. http://www.jdchct.or.jp/data/report/2019/
Kondo E, Ikeda T, Goto H, Nishikori M, Maeda N, Matsumoto K, et al. Pharmacokinetics of thiotepa in high-dose regimens for autologous hematopoietic stem cell transplant in Japanese patients with pediatric tumors or adult lymphoma. Cancer Chemother Pharmacol. 2019;84:849–60. https://doi.org/10.1007/s00280-019-03914-2
Maanen MJ, Smeets CJ, Beijnen JH. Chemistry, pharmacology and pharmacokinetics of N,N’,N” -triethylenethiophosphoramide (ThioTEPA). Cancer Treat Rev. 2000;26:257–68. https://doi.org/10.1053/ctrv.2000.0170
Heideman RL, Cole DE, Balis F, Sato J, Reaman GH, Packer RJ, et al. Phase I and pharmacokinetic evaluation of thiotepa in the cerebrospinal fluid and plasma of pediatric patients: Evidence for dose-dependent plasma clearance of thiotepa. Cancer Res. 1989;49:736–41.
Shay H, Zarafonetis C, Smith N, Woldow I, Sun DC. Treatment of leukemia with triethylene thiophosphoramide (thio-TEPA); preliminary results in experimental and clinical leukemia. AMA Arch Intern Med. 1953;92:628–45. https://doi.org/10.1001/archinte.1953.00240230028004
Shay H, Sun DC. Clinical studies of triethylenethiophosphoramide in the treatment of inoperable cancer. Cancer. 1955;8:498–511. https://doi.org/10.1002/1097-0142(1955)8:3<498::aid-cncr2820080310>3.0.co;2-m
TEPADINA 15 mg powder for concentrate for solution for infusion (summary of product characteristics). ADIENNE S.r.l. S.U, Caponago, Italy; 2015. https://www.ema.europa.eu/en/documents/product-information/tepadina-epar-product-information_en.pdf
Kondo E. Primary central nervous system lymphoma: advances in treatment strategies [In Japanese]. Rinsho Ketsueki 2020;61:510–9. https://doi.org/10.11406/rinketsu.61.510
Suzuki R, Takahashi Y, Inoue M, Kanamori H, Hashii Y, Sakamaki H, et al. Thiotepa for autologous hematopoietic stem cell transplantation for solid tumors. Ann Oncol. 2015;26:vii140. https://doi.org/10.1093/annonc/mdv472.156
Nishiwaki S, Ando Y. Application of the new process for unapproved drug use: dilemma of universal health care coverage in Japan. J Glob Onco.l 2019;5:1–3. https://doi.org/10.1200/JGO.19.00313
Thiotepa 100 mg (RETHIO IV infusion) [Japanese package insert] Osaka, Japan: Sumitomo Pharma Co., Ltd.; 2022. https://pins.japic.or.jp/pdf/newPINS/00068017.pdf
Nishikori M, Masaki Y, Fujii N, Ikeda T, Takahara-Matsubara M, Sugimoto S, et al. An expanded-access clinical study of thiotepa (DSP-1958) high-dose chemotherapy before autologous hematopoietic stem cell transplantation in patients with malignant lymphoma. Int J Hematol. 2022;115:391–98. https://doi.org/10.1007/s12185-021-03263-y
Barton CD, Waugh LK, Nielsen MJ, Paulus S. Febrile neutropenia in children treated for malignancy. J Infect. 2015;71:S27–35. https://doi.org/10.1016/j.jinf.2015.04.026
Haeusler GM, Sung L, Ammann RA, Phillips B. Management of fever and neutropenia in paediatric cancer patients: Room for improvement? Curr Opin Infect Dis. 2015;28:532–8. https://doi.org/10.1097/QCO.0000000000000208
Bhuta R, Nieder M, Jubelirer T, Ladas EJ. The gut microbiome and pediatric cancer: current research and gaps in knowledge. J Natl Cancer Inst Monogr. 2019;2019:169–73. https://doi.org/10.1093/jncimonographs/lgz026
Secombe KR, Coller JK, Gibson RJ, Wardill HR, Bowen JM. The bidirectional interaction of the gut microbiome and the innate immune system: implications for chemotherapy-induced gastrointestinal toxicity. Int J Cancer. 2019;144:2365–76. https://doi.org/10.1002/ijc.31836
Valteau-Couanet D, Le Deley MC, Bergeron C, Ducassou S, Michon J, Rubie H, et al. Long-term results of the combination of the N7 induction chemotherapy and the busulfan-melphalan high dose chemotherapy. Pediatr Blood Cancer 2014;61:977–81. https://doi.org/10.1002/pbc.24713
Epperla N, Li A, Logan B, Fretham C, Chhabra S, Aljurf M, et al. Incidence, risk factors for and outcomes of transplant-associated thrombotic microangiopathy. Br J Haematol. 2020;189:1171–81. https://doi.org/10.1111/bjh.16457
Caselli D, Rosati A, Faraci M, Podda M, Ripaldi M, Longoni D, et al. Risk of seizures in children receiving busulphan-containing regimens for stem cell transplantation. Biol Blood Marrow Transplant 2014;20:282–5. https://doi.org/10.1016/j.bbmt.2013.10.028
Vassal G, Deroussent A, Hartmann O, Challine D, Benhamou E, Valteau-Couanet D, et al. Dose-dependent neurotoxicity of high-dose busulfan in children: a clinical and pharmacological study. Cancer Res. 1990;50:6203–7.
Wolff SN, Herzig RH, Fay JW, LeMaistre CF, Brown RA, Frei-Lahr D, et al. High-dose N,N’,N”-triethylenethiophosphoramide (thiotepa) with autologous bone marrow transplantation: Phase I studies. Semin Oncol. 1990;17:2–6.
Weaver CH, Bensinger WI, Appelbaum FR, Lilleby K, Sandmaier B, Brunvand M, et al. Phase I study of high-dose busulfan, melphalan and thiotepa with autologous stem cell support in patients with refractory malignancies. Bone Marrow Transplant. 1994;14:813–9.
Hara J, Osugi Y, Ohta H, Matsuda Y, Nakanishi K, Takai K, et al. Double-conditioning regimens consisting of thiotepa, melphalan and busulfan with stem cell rescue for the treatment of pediatric solid tumors. Bone Marrow Transplant. 1998;22:7–12. https://doi.org/10.1038/sj.bmt.1701283
Okada K, Yamasaki K, Nitani C, Fujisaki H, Osugi Y, Hara J. Double-conditioning regimen consisting of high-dose thiotepa and melphalan with autologous stem cell rescue for high-risk pediatric solid tumors: a second report. Pediatr Blood Cancer. 2019;66:e27953 https://doi.org/10.1002/pbc.27953
Mason WP, Grovas A, Halpern S, Dunkel IJ, Garvin J, Heller G, et al. Intensive chemotherapy and bone marrow rescue for young children with newly diagnosed malignant brain tumors. J Clin Oncol. 1998;16:210–21. https://doi.org/10.1200/JCO.1998.16.1.210
Altshuler C, Haley K, Dhall G, Vasquez L, Gardner SL, Stanek J, et al. Decreased morbidity and mortality of autologous hematopoietic transplants for children with malignant central nervous system tumors: the ‘Head Start’ trials, 1991-2009. Bone Marrow Transplant. 2016;51:945–8. https://doi.org/10.1038/bmt.2016.45
Cacchione A, LeMaitre A, Couanet DV, Benhamou E, Amoroso L, Simonnard N, et al. Risk factors for hepatic veno-occlusive disease: A retrospective unicentric study in 116 children autografted after a high-dose BU-thiotepa regimen. Bone Marrow Transplant. 2008;42:449–54. https://doi.org/10.1038/bmt.2008.186
Desai AV, Heneghan MB, Li Y, Bunin NJ, Grupp SA, Bagatell R, et al. Toxicities of busulfan/melphalan versus carboplatin/etoposide/melphalan for high-dose chemotherapy with stem cell rescue for high-risk neuroblastoma. Bone Marrow Transplant. 2016;51:1204–10. https://doi.org/10.1038/bmt.2016.84
Elborai Y, Hafez H, Moussa EA, Hammad M, Hussein H, Lehmann L, et al. Comparison of toxicity following different conditioning regimens (busulfan/melphalan and carboplatin/etoposide/melphalan) for advanced stage neuroblastoma: Experience of two transplant centers. Pediatr Transplant. 2016;20:284–9. https://doi.org/10.1111/petr.12638
Grill J, Kalifa C, Doz F, Schoepfer C, Sainte-Rose C, Couanet D, et al. A high-dose busulfan-thiotepa combination followed by autologous bone marrow transplantation in childhood recurrent ependymoma. A phase-II study. Pediatr Neurosurg. 1996;25:7–12. https://doi.org/10.1159/000121089
DeNunzio NJ, Yock TI. Modern radiotherapy for pediatric brain tumors. Cancers. 2020;12:1533. https://doi.org/10.3390/cancers12061533.
Kline CN, Mueller S. Neurocognitive outcomes in children with brain tumors. Semin Neurol. 2020;40:315–21. https://doi.org/10.1055/s-0040-1708867
Mulhern RK, Merchant TE, Gajjar A, Reddick WE, Kun LE. Late neurocognitive sequelae in survivors of brain tumours in childhood. Lancet Oncol. 2004;5:399–408. https://doi.org/10.1016/S1470-2045(04)01507-4
Okada K, Soejima T, Sakamoto H, Hirato J, Hara J. Phase II study of reduced-dose craniospinal irradiation and combination chemotherapy for children with newly diagnosed medulloblastoma: a report from the Japanese Pediatric Brain Tumor Consortium. Pediatr Blood Cancer. 2020;67:e28572. https://doi.org/10.1002/pbc.28572
Yamasaki K, Okada K, Soejima T, Sakamoto H, Hara J. Strategy to minimize radiation burden in infants and high-risk medulloblastoma using intrathecal methotrexate and high-dose chemotherapy: a prospective registry study in Japan. Pediatr Blood Cancer. 2020;67:e28012. https://doi.org/10.1002/pbc.28012
Yesilipek MA. Hematopoetic stem cell transplantation in children. Turk Pediatr Ars. 2014;49:91–8. https://doi.org/10.5152/tpa.2014.2010
Gardner SL, Asgharzadeh S, Green A, Horn B, McCowage G, Finlay J. Intensive induction chemotherapy followed by high dose chemotherapy with autologous hematopoietic progenitor cell rescue in young children newly diagnosed with central nervous system atypical teratoid rhabdoid tumors. Pediatr Blood Cancer. 2008;51:235–40. https://doi.org/10.1002/pbc.21578
Zaky W, Dhall G, Ji L, Haley K, Allen J, Atlas M, et al. Intensive induction chemotherapy followed by myeloablative chemotherapy with autologous hematopoietic progenitor cell rescue for young children newly-diagnosed with central nervous system atypical teratoid/rhabdoid tumors: The Head Start III experience. Pediatr Blood Cancer. 2014;61:95–101. https://doi.org/10.1002/pbc.24648
Bashir Q, Thall PF, Milton DR, Fox PS, Kawedia JD, Kebriaei P, et al. Conditioning with busulfan plus melphalan versus melphalan alone before autologous haemopoietic cell transplantation for multiple myeloma: an open-label, randomised, phase 3 trial. Lancet Haematol. 2019;6:e266–75. https://doi.org/10.1016/S2352-3026(19)30023-7
Kebriaei P, Madden T, Kazerooni R, Wang X, Thall PF, Ledesma C, et al. Intravenous busulfan plus melphalan is a highly effective, well-tolerated preparative regimen for autologous stem cell transplantation in patients with advanced lymphoid malignancies. Biol Blood Marrow Transplant. 2011;17:412–20. https://doi.org/10.1016/j.bbmt.2010.07.016
Mosteller RD. Simplified calculation of body-surface area. N Engl J Med. 1987;317:1098. https://doi.org/10.1056/NEJM198710223171717
Du Bois D, Du, Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition. 1989;5:303–11.
Acknowledgements
The authors would like to express our sincere gratitude to all patients and their families who participated in this study. We thank Sally-Anne Mitchell, Ph.D. (McCANN HEALTH CMC, Japan) for providing medical writing support, which was funded by Sumitomo Pharma Co., Ltd., in compliance with Good Publication Practice guidelines (https://www.ismpp.org/gpp-2022).
Author information
Authors and Affiliations
Contributions
All authors have contributed significantly to the manuscript and meet all relevant authorship requirements. JH and MM substantially contributed to the conception and design of the study; other authors were involved in the acquisition, analysis, or interpretation of data. All authors took part in manuscript drafting or critical revision for important intellectual content, are in agreement with the content of the manuscript, and provided final approval of the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
This study was funded by Sumitomo Pharma Co., Ltd (formerly Sumitomo Dainippon Pharma Co., Ltd.). JH is an advisory board member of OHARA Pharmaceuticals and received consultancy fees from OHARA Pharmaceuticals. KM and NM declared no conflict of interest. MT and SS are employees of Sumitomo Pharma. HG received lecture fees from Amgen Astellas BioPharma.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The information to Supplementary Table 2 was given by mistake as Supplementary Table 1 in the sentence: The remaining two patients did not meet the narrow definition of engraftment used in this study (neutrophil count ≥500/mm3 for 3 consecutive days), but both patients did achieve a neutrophil count ≥500/mm3 on multiple non-consecutive days after autologous HSCT (Supplementary Table S2). In addition supplementary table 2 was missing from this article and has now been uploaded.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hara, J., Matsumoto, K., Maeda, N. et al. High-dose thiotepa, in conjunction with melphalan, followed by autologous hematopoietic stem cell transplantation in patients with pediatric solid tumors, including brain tumors. Bone Marrow Transplant 58, 123–128 (2023). https://doi.org/10.1038/s41409-022-01820-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01820-5
- Springer Nature Limited