Abstract
Transdiagnostic approaches to psychiatry have significant potential in overcoming the limitations of conventional diagnostic paradigms. However, while frameworks such as the Research Domain Criteria have garnered significant enthusiasm among researchers and clinicians from a theoretical angle, examples of how such an approach might translate in practice to understand the biological mechanisms underlying complex patterns of behaviors in realistic and heterogeneous populations have been sparse. In a richly phenotyped clinical sample (n = 186) specifically designed to capture the complex nature of heterogeneity and comorbidity within- and between stress- and neurodevelopmental disorders, we use exploratory factor analysis on a wide range of clinical questionnaires to identify four stable functional domains that transcend diagnosis and relate to negative valence, cognition, social functioning and inhibition/arousal before replicating them in an independent dataset (n = 188). We then use connectopic mapping to map inter-individual variation in fine-grained topographical organization of functional connectivity in the striatum—a central hub in motor, cognitive, affective and reward-related brain circuits—and use multivariate machine learning (canonical correlation analysis) to show that these individualized topographic representations predict transdiagnostic functional domains out of sample (r = 0.20, p = 0.026). We propose that investigating psychiatric symptoms across disorders is a promising path to linking them to underlying biology, and can help bridge the gap between neuroscience and clinical psychiatry.
Similar content being viewed by others
Introduction
Psychiatric disorders are behaviorally and biologically complex, as is evidenced by our syndrome-level understanding of their clinical phenomenology. While a common framework is critical for providing patient care and evaluating the efficacy of therapeutic options, classification systems that are commonly used to define psychiatric disorders also constrain the way in which we are able to connect these disorders to underlying biology [1]. Moreover, these symptom-based diagnostic systems typically reflect a convergence of multiple distinct biological mechanisms, contributing to significant clinical heterogeneity within disorders and high levels of comorbidity between them [2, 3]. This comorbidity can also stem from a single biological origin driving various symptoms that are classified separately, which may help explain treatment being effective for different classified disorders. Our underlying neurobiology is not constrained to classification paradigms, which is exemplified by various brain regions, genes and neurobiological pathways being implicated across the psychiatric spectrum [4, 5]. This lack of distinct underlying biological features for psychiatric disorders has impaired clinical progress, for example in the search for reliable neuroimaging biomarkers for the presence or prognosis of mental disorders [6]. The MIND-Set (“Measuring Integrated Novel Dimensions in Neurodevelopmental and Stress-related Mental Disorders”) cohort was initiated with this in mind, employing concepts from transdiagnostic groundwork such as the Research Domain Criteria, to collect data across neurodevelopmental and stress-related disorders and investigate their clinical and biological (co)variation [7]. These transdiagnostic frameworks focus on domains of functioning across diagnoses that could prove valuable from a research perspective, as well as from the perspective of understanding patient functioning at the individual level [1].
Within such frameworks, neuroimaging studies have made significant progress in linking specific domains of functioning to brain networks and regions across disorders [6, 8, 9]. These studies suggest several key regions and circuits being relevant in the majority of psychiatric disorders, with related disorders (e.g. mood and anxiety disorders) showing strongest similarities with one another in terms of their association with distributed patterns of brain function and/or structure. The striatum, which has been implicated across the behavioral spectrum, is such a key locus of convergence for psychopathology across multiple disorders. As a central hub in a range of motor, cognitive, affective and reward-related brain circuits, the striatum receives a large array of lateral and medial cortical inputs, which are topographically organized [10, 11]. In addition, it receives afferents from the thalamus and dopaminergic input from the brain stem [12], while the striatum itself projects mainly to other basal ganglia. Via extensive connectivity with the (pre)frontal cortex, the striatum is critical in learning, adapting and executing goal-directed behavior, taking into account the complexity that leads to behavior such as emotion and cognition. Being involved in the entire spectrum of goal-directed behavior from sensory through motivational to cognitive and executive function, it is unsurprising that changes in striatal structure or function have been extensively reported across a wide range of psychiatric disorders [13,14,15,16,17,18]. In addition, inter-individual differences in striatal structure and function have been shown to reflect disease severity across psychotic disorders, depressive disorders, post-traumatic stress disorder and obsessive-compulsive disorder, regardless of which specific disorder was present [19]. These factors make the striatum a target of significant interest when investigating broad functional domains impaired across psychiatric disorders.
The striatum is extensively connected with almost the entire cortex and is involved in multiple behaviorally relevant circuits which can be probed, for example, through functional connectivity. However, connectivity in the striatum is topographically organized, such that nearby regions in the striatum are connected with nearby regions in cortex and therefore conventional functional connectivity approaches are unable to capture the complexity of functional connections within this complex system, nor reflect the topographic characteristics of striatal organization at the single-subject level. Through recent advancements, we can now investigate the topographic organization of striatal circuits through connectopic maps [20]. These maps, which are estimated at the single-subject level, represent slowly varying topographically organized connectivity patterns (‘connectopic gradients’) that reveal how connectivity to the rest of the brain is organized within a region of interest, even when multiple connectivity patterns are spatially overlapping but functionally distinct. The latter is important because in striatum we have shown that the dominant mode of connectivity change distinguishes between caudate nucleus, nucleus accumbens and putamen, while the second mode follows a rostral-caudal gradient across the three striatal substructures [21]. The high test-retest reliability of connectopic maps compared to conventional measures of function or functional connectivity further highlights their potential in obtaining the type of single-subject brain function that allows for strong translational work to the clinic [20]. We have also shown that topographic connectivity in the striatum is related to complex goal-directed behaviors at the individual level [21], and shows a strong correspondence with the spatial distribution of dopaminergic projections, demonstrating their potential for investigating striatal function [22]. Because connectopic maps characterize complex regions such as the striatum in a way that links to behavior, we hypothesize that individual differences in connectopic maps are predictive of psychiatric symptomatology across disorders.
In this study, we apply connectopic mapping to a richly phenotyped, transdiagnostic and highly comorbid cohort—the ‘Measuring Integrated Novel Dimensions in neurodevelopmental and stress-related mental disorders’ (MIND-Set) study—aiming to (i) dissect psychiatric symptom profiles within a heterogeneous and clinically representative sample, providing subjective quantitative measures across different domains of functioning (“behavior”) that cut across diagnoses and measurement instruments and (ii) show that these domains of functioning are linked to the topographic functional organization of the striatum at the level of the individual patient. To achieve these goals, we apply an innovative multivariate analytical strategy that combines penalized canonical correlation analysis with stability selection that provides the ability to learn brain-behavior associations whilst providing unbiased estimates of generalizability and strong statistical guarantees over false detection of associations.
Methods and materials
A detailed description of analytical procedures is provided in the supplement. In brief, data were collected as part of the MIND-Set study, an observational, cross-sectional study that includes adult patients with stress-related and/or neurodevelopmental disorders that were assessed at the outpatient unit of the department of psychiatry at the Radboud university medical center (Radboudumc) in Nijmegen, the Netherlands. For a more detailed description of the study design and set-up we refer to previous work [7]. Importantly, individuals in this sample have high levels of comorbidity, accurately reflecting the clinical reality. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. All procedures involving human subjects/patients were approved by the local medical ethical committee (METC Oost-Nederland). Written informed consent was obtained from all subjects.
Study participants
Discovery/neuroimaging sample: participants from the MIND-Set cohort were included who met criteria for at least one of the following psychiatric disorders: major depressive disorder, anxiety disorder, attention-deficit/hyperactivity disorder (ADHD) and autism spectrum disorder (ASD), and had completed behavioral and neuroimaging assessments (n = 203, before connectopic mapping; final sample n = 186). Diagnoses were confirmed using the Structured Clinical Interview for DSM-IV-TR (SCID-I/P) [23], the Diagnostic Interview for Adult ADHD (DIVA) [24] and/or the Nijmegen Interview for Diagnosing adult Autism spectrum disorders (NIDA) [25]. Replication sample: The replication sample was used to replicate the exploratory factor analysis (EFA) structure in an independent sample, n = 188 (101 men / 87 women; age 43.6 ± 14.3 years; MDD = 61, anxiety = 54, ADHD = 54, ASD = 38). This replication sample followed the same inclusion process and deep phenotyping as the main sample, with the exception of an MRI session.
Behavioral data and factor analysis
We used an extensive panel of questionnaires covering multiple symptom and functional domains (see Tables 1 and 2). To capture latent factors driving (sets of) behavior, we used exploratory factor analysis (EFA) to uncover four domains of functioning that transcend conventional diagnostic (DSM) boundaries. This also decreases our feature-to-sample ratio to prevent overfitting the data [26, 27]. The EFA was repeated on the independent replication sample.
Connectopic mapping and statistical analysis
Imaging data was processed using standard pipelines with FSL 5.0.11 (FMRIB, Oxford, UK) and careful attention was given to addressing known confounds in resting FMRI, such as head motion (see supplementary data) [28, 29]. We applied ConGrads, a fully data-driven method based on manifold learning and spatial statistics, to the resting-state fMRI data to obtain highly individualized representations of striatal functioning (‘connectopic maps’) for each subject [20]. This method (i) derives voxel-wise connectivity fingerprints by correlating fMRI time-series within a region-of-interest (such as the striatum) to all fMRI time-series outside of the region-of-interest, then (ii) uses manifold learning to calculate the connectopic maps based on changes in connectivity pattern within the region-of-interest and finally (iii) spatial statistics to convert the connectopic maps to coefficients that can be tested within other analyses (fig. S1). The connectopic maps represent slowly varying topographic patterns of connectivity (‘gradients’) that map connectivity changes within a target region in relation to the rest of the brain at the individual subject level. Although we focused on the principal or dominant gradient, multiple overlapping topographic representations can exist simultaneously within a single region, so both principal and second gradients were estimated to be able to investigate other potential effects driving associations to behavior. All gradients were visually inspected, and subjects were excluded (n = 17) if a clear gradient could not be estimated, due to imaging artefacts or spatial correlation of individual gradients to the group maps was low, following the protocol by Isakoglou et al. [30]. For each subject, trend surface coefficients fit to the gradients from each hemisphere were used to provide a low-dimensional summary of the connectopy, following prior work [20, 21]. These were concatenated for each subject and entered into a penalized canonical correlation analysis (CCA) model [31]. The CCA was used to determine the association between the behavioral domains of functioning and striatal gradients, using feature stability selection [32] and out-of-sample testing. This procedure has been described in detail in earlier publications [31], and is detailed in the supplementary information. To establish model consistency, we performed the full analysis ten times using different (stratified) data splits. Finally, we calculated the (cross-)loadings of brain features to the different functional domains to understand the relative contribution of different behavioral and neurobiological features to the associations.
Results
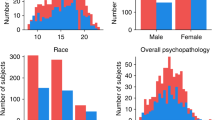
A total of 186 subjects were included in the final analysis (107 men, 79 women; age 37.9 ± 13.2 years). As per the study design, the sample contained high levels of comorbidity across symptoms and diagnoses: 102 subjects had a single diagnosis, 69 had two diagnoses, 13 had 3 diagnoses and 2 subjects had all included disorders (Table 1). Variation in comorbidity is presented in Fig. 1a.
A Diagram showing comorbidity within the data. *indicates comorbidity with the opposite disorder (MDD + ASD and AD + ADHD). B Normalized group-average subject loading onto the four functional domains in relation to diagnosis. C Normalized group-average subject loading onto functional domains in relation to degree of comorbidity. MDD major depressive disorder, AD anxiety disorder, ASD autism spectrum disorder, ADHD attention-deficit hyperactivity disorder.
Factor analysis and functional domains
Exploratory factor analysis on 31 scales and subscales across psychiatric symptomatology decomposed behavioral data into four factors (Kaiser–Meyer–Olkin test 0.85, see Table 2). The first factor relates to negative valence systems [1] and contains scores relating to depressive and anxiety symptoms, in addition to negative affect and repetitive negative thinking scores. The second factor includes symptoms related to cognitive function, including planning, organizing, working memory and mental capacity. The third factor relates to social functioning and includes social awareness, communication, and external awareness. The fourth factor includes symptoms relating to (dis)inhibition, antagonism and emotional lability. Broadly, we interpreted these four factors as representing four functional domains: (1) negative valence, (2) cognitive function, (3) social processes and (4) arousal/inhibition. These functional domains transcend diagnostic boundaries and each factor includes scales and subscales from questionnaires probing different underlying disorders. As expected, these factors were not independent but were correlated to one another. Factor analysis on the replication sample of 188 subjects showed almost identical grouping of behavioral data (Table 2; correlation of factor loadings r = 0.90, p < 0.001).
Regarding the distribution of factor coefficients within the different diagnostic classifications, we observed that while variance within groups was high, on average each diagnostic category showed a pattern that resembles its clinical manifestation (Fig. 1b). We also observed that as the degree of comorbidity increases (i.e. quantified by number of diagnoses for each individual), so did the comparative loading onto all four domains of functioning (Fig. 1c).
Connectopic mapping and canonical correlation analysis
At the group-level, striatal connectopic maps showed a best fit using up to a third-order polynomial. The principal connectopic maps showed striatal topography similar to previous work [21], where it follows structural boundaries and gradually changes from putamen, to nucleus accumbens, to caudate (Fig. 2a). Projection maps for the striatal gradient onto the rest of the brain were also in line with previous work (Fig. 2b).
Functional domain scores and trend surface coefficients were entered into a penalized canonical correlation analysis. Stability selection revealed the first and third-order trend surface coefficients in the x-direction of the left striatum as stable brain features. We found a clear and consistent correlation between functional domain scores and the connectivity gradients in the striatum (average in-sample correlation r = 0.25 ± 0.04, out-of-sample correlation r = 0.20 ± 0.02, p = 0.026). This interaction explained variance ((R2), out-of-sample) across three of the four behavioral domains (negative valence 2.5%, cognitive 2.6%, social 0.2%, arousal/inhibition 3.6%), comparable in size to previous CCA-derived brain-behavioral associations [31]. While these results were driven by left striatum organization within the model, no differences in gradients between left and right striatum were observed.
Additional tests were performed to determine whether our results could be explained by other effects (see Supplementary information). Firstly, we showed that the reported association is specific to this particular topographic representation by repeating the full analysis using the second functional striatal gradient. Secondly, we showed that functional effects we report cannot be explained by underlying structural differences by repeating the analysis using classical volumetric measures of the striatum and finally, we showed that striatal topographic organization is not associated with the conventional diagnostic labels by repeating the CCA using the diagnostic labels instead of the functional domain scores. None of these other data modalities revealed any significant interactions between behavior and striatum structure or function. We also explored how degree of comorbidity was related to changes in the connectopic map by comparing patients without comorbidity to those with high comorbidity, but while we observed that the difference maps between these groups still followed a similar gradient to the separate and group maps, we found no significant differences.
Discussion
In this work, we showed how complex behavioral data in a sample with high psychiatric comorbidity can be represented in stable and reproducible domains of functioning. These functional domains are cross-diagnostic in that each of these domains incorporate parts from different questionnaires, and reproducible in that we were able to derive effectively identical latent structure across two independent samples. These domains of functioning are also similar to broad research areas in cognitive neuroscience in both healthy and clinical populations [1]. We found one domain strongly tied to negative emotion and repetitive negative thinking (negative valence), one tied to organization and broad cognition, a domain related to social perception and functioning, and a domain related to arousal and (dis)inhibition. We observed that at the group level classified disorders showed a typical pattern of functional impairment across all four functional domains. The functional domains also showed a sensible grouping of symptoms that are known to be present within psychiatric patients, such as depressive symptoms co-occurring with repetitive negative thinking and problems in self-conceptualization. By grouping related symptomatology across disorders and across measurement instruments, these domains could be used to identify specific targets of treatment. As an example, repetitive negative thinking can be present in both depression and anxiety disorders and are grouped within the negative valence system. Treatments designed to affect negative valence systems dysfunction, such as cognitive behavioral therapy, might be effective for both disorders because they target this underlying functional domain.
We established that these functional domains are reflected in underlying brain function, as represented by the functional organization of the striatum across disorders. While the strength of the observed relation is in line with other studies using similar (CCA-)analysis methods, our findings in light of the broad scope of behavioral information and highly comorbid sample used in this work highlight the value of transdiagnostic frameworks in understanding the biological underpinnings of psychiatric disorders and symptomatology. In addition to the earlier-mentioned relevance of the striatum in stress- and neurodevelopmental disorders, there is a large body of literature that supports our findings in linking striatum function and connectivity to the functional domains we observed. The ventral striatum is the key node within reward-circuitry, and crucial in the context of arousal and (dis)inhibition [11]. Within the frontostriatal circuit the striatum plays an important role in regulating behavior in response to salient stimuli. Differences in frontostriatal circuitry have been at the forefront of neuroimaging research into disorders marked by strong disinhibition such as ADHD, oppositional defiant disorder, conduct disorder and addiction [33,34,35]. The ventral striatum and the nucleus accumbens are also well established as important areas for negative valence and affect [36], and our findings are in line with other studies that have found striatal connectivity changes in (remitted) depression and depressive symptoms [37, 38]. With respect to the cognitive functional domain, the striatum and its dopaminergic modulation regulate those parts of cognition related to goal-directed behavior, such as working memory and attention switching [39]. In fact, striatal markers based on neonatal imaging are predictive of cognitive ability years later [40]. Changes in striatal morphology are also a distinguishing feature of neurodegenerative disorders marked by strong cognitive decline such as Alzheimer’s and Parkinson’s disease [41], disorders with high psychiatric comorbidity.
Taken together, known associations between striatal function and several functional domains in both clinical and healthy populations corroborate our findings. Furthermore, several studies have found striatal structure and/or function to be a marker for general symptoms, regardless of diagnosis [19]. The ventral striatum is also increasingly recognized as a target for deep brain stimulation across disorders, again highlighting its transdiagnostic importance [42]. The nature of its impact on systems that are core to dysfunction in the context of psychiatric disorders explains limited disorder-specificity, while also underscoring the relevance of investigating functions over classifications. With regards to the underlying biology, we observed brain-behavioral interactions in the dominant functional gradient, where earlier work found interactions to behavior in higher-level gradients [21, 22]. We consider that our results underline the complexity of the functional organization of the striatum, where different overlapping gradients are associated with different biological mechanisms. This underscores the necessity of investigating spatially overlapping patterns of connectivity in order to fully understand striatal involvement. The ability to use connectomics to uncover these complex patterns on the single-subject level holds promise for clinical applications, as similar techniques are already being used to guide clinical decision making where high individualized accuracy is critical such as neurosurgical planning [43, 44]. By design, understanding the biology driving patterns of symptoms within a single individual has a much more narrow gap to the clinical reality compared to group-level biological differences driving broad classified disorders.
Several limitations to the current work need to be addressed. Firstly, the MIND-Set sample is highly comorbid by design, so replication and generalization studies in less complex or healthy samples are warranted to demonstrate transferability of the functional domains. Secondly, while substantial for a highly heterogeneous cohort, our sample size is moderate. Although balanced by careful assessment procedures including rigorous out-of-sample validation and stability selection [32], repeating the analysis in larger samples with a broader scope of biological targets could further uncover related circuitry. This would also allow for further analyses on subsets of patients with specific patterns of symptomatology to disentangle how the changes in striatal function relate to changes within functional domains, and how this relates to the degree of comorbidity. Finally, due to the heterogenous nature of our sample, we cannot completely rule out the effects of medication use, illness duration and age of onset, as these are directly tied to the pattern of symptomatology and/or disorders present.
In conclusion, we showed how psychiatric symptomatology can be deconstructed into functional domains that are reflected in underlying neurobiology. We believe that this transdiagnostic approach, which enables investigating domains of functioning that still carry the signature of the classified disorder while also incorporating individual variation that transcends the label, has great potential in overcoming current limitations in clinical and computational psychiatry. With a stronger biological footing and individualized nature, the functional domains could prove valuable in predicting clinical outcome. Through transdiagnostic research and understanding how disruptions in neural circuitry give rise to non-specific psychiatric symptoms or shared symptoms across different disorders, we will be able to pave the way for personalized treatment targeting circuits, regardless of which classified disorder is present.
References
Cuthbert BN, Insel TR. Toward the future of psychiatric diagnosis: the seven pillars of RDoC. BMC Med. 2013. https://doi.org/10.1186/1741-7015-11-126.
van Loo HM, Romeijn JW, de Jonge P, Schoevers RA. Psychiatric comorbidity and causal disease models. Prev Med (Balt). 2013;57:748–52.
Lynch CJ, Gunning FM, Liston C. Causes and consequences of diagnostic heterogeneity in depression: paths to discovering novel biological depression subtypes. Biol Psychiatry 2020;88:83–94.
Geschwind DH, Flint J. Genetics and genomics of psychiatric disease. Science (80-). 2015;349:1489–94.
Gandal MJ, Haney JR, Parikshak NN, Leppa V, Ramaswami G, Hartl C, et al. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science. 2018;359:693–7.
Goodkind M, Eickhoff SB, Oathes DJ, Jiang Y, Chang A, Jones-Hagata LB, et al. Identification of a common neurobiological substrate for mental illness. JAMA Psychiatry. 2015;72:305–15.
van Eijndhoven PFP, Collard RM, Vrijsen JN, Geurts DGM, Arias-Vasquez A, Schellekens AFA et al. Measuring integrated novel dimensions in neurodevelopmental and stress-related mental disorders (MIND-Set): a cross-sectional comorbidity study from an RDoC perspective. medRxiv. 2021. https://doi.org/10.1101/2021.06.05.21256695.
McTeague LM, Huemer J, Carreon DM, Jiang Y, Eickhoff SB, Etkin A. Identification of common neural circuit disruptions in cognitive control across psychiatric disorders. Am J Psychiatry. 2017;174:676–85.
Arnsten AFT, Rubia K. Neurobiological circuits regulating attention, cognitive control, motivation, and emotion: disruptions in neurodevelopmental psychiatric disorders. J Am Acad Child Adolesc Psychiatry. 2012;51:356–67.
Alexander GE, DeLong MR, Strick PL. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci. 1986;9:357–81.
Haber SN, Knutson B. The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology. 2010;35:4–26.
Haber SN. Corticostriatal circuitry. Dialogues Clin Neurosci. 2016;18:7–21.
McCutcheon RA, Abi-Dargham A, Howes OD. Schizophrenia, dopamine and the striatum: from biology to symptoms. Trends Neurosci. 2019;42:205–20.
Der-Avakian A, Markou A. The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci. 2012;35:68–77.
Luking KR, Repovs G, Belden AC, Gaffrey MS, Botteron KN, Luby JL, et al. Functional connectivity of the amygdala in early-childhood-onset depression. J Am Acad Child Adolesc Psychiatry. 2011;50:1–22.
Lago T, Davis A, Grillon C, Ernst M. Striatum on the anxiety map: small detours into adolescence. Brain Res. 2017;1654:177–84.
Kim HJ, Lee JH, Yun K, Kim JH. Alterations in striatal circuits underlying addiction-like behaviors. Mol Cells. 2017;40:379–85.
Shepherd GMG. Corticostriatal connectivity and its role in disease. Nat Rev Neurosci 2013;14:278–91.
Gong Q, Scarpazza C, Dai J, He M, Xu X, Shi Y, et al. A transdiagnostic neuroanatomical signature of psychiatric illness. Neuropsychopharmacology. 2019;44:869–75.
Haak KV, Marquand AF, Beckmann CF. Connectopic mapping with resting-state fMRI. Neuroimage 2018;170:83–94.
Marquand AF, Haak KV, Beckmann CF Functional corticostriatal connection topographies predict goal-directed behaviour in humans. Nat Hum Behav. 2017. https://doi.org/10.1038/s41562-017-0146.
Oldehinkel M, Llera A, Faber M, Huertas I, Buitelaar JK, Bloem BR, et al. Mapping dopaminergic projections in the human brain with resting-state fMRI. Elife. 2022. https://doi.org/10.7554/ELIFE.71846.
First M, Gibbon M, Spitzer R, Williams J, Benjamin J. Structured clinical interview for DSM-IV-TR axis I disorders, reseach version, patient edition (I/P). New York: Biometrics Research, New York State Psychiatric Institute; 1997.
Pettersson R, Söderström S, Nilsson KW. Diagnosing ADHD in adults: an examination of the discriminative validity of neuropsychological tests and diagnostic assessment instruments. J Atten Disord. 2018;22:1019–31.
Vuijk R, Deen M, Arntz A, Geurts HM. First psychometric properties of the dutch interview for diagnostic assessment of autism spectrum disorder in adult males without intellectual disability. J Autism Dev Disord. 2021. https://doi.org/10.1007/S10803-021-05225-Z.
Dinga R, Schmaal L, Marquand AF. A closer look at depression biotypes: correspondence relating to Grosenick et al. (2019). Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:554–5.
Mihalik A, Adams RA, Huys Q. Canonical correlation analysis for identifying biotypes of depression. Biol psychiatry Cogn Neurosci neuroimaging. 2020;5:478–80.
Jenkinson M, Beckmann CF, Behrens TEJ, Woolrich MW, Smith SM. FSL. Neuroimage. 2012;62:782–90.
Pruim RHR, Mennes M, van Rooij D, Llera A, Buitelaar JK, Beckmann CF. ICA-AROMA: a robust ICA-based strategy for removing motion artifacts from fMRI data. Neuroimage. 2015;112:267–77.
Isakoglou C, Haak KV, Wolfers T, Floris DL, Llera A, Oldehinkel M et al. Fine-grained topographic organization within somatosensory cortex during resting-state and emotional face-matching task and its association with ASD traits. bioRxiv. 2022. https://doi.org/10.1101/2022.04.26.489525.
Ing A, Sämann PG, Chu C, Tay N, Biondo F, Robert G, et al. Identification of neurobehavioural symptom groups based on shared brain mechanisms. Nat Hum Behav. 2019;3:1306–18.
Meinshausen N, Bühlmann P. Stability selection. J R Stat Soc Ser B Stat Methodol. 2010;72:417–73.
Cubillo A, Halari R, Smith A, Taylor E, Rubia K. A review of fronto-striatal and fronto-cortical brain abnormalities in children and adults with Attention Deficit Hyperactivity Disorder (ADHD) and new evidence for dysfunction in adults with ADHD during motivation and attention. Cortex 2012;48:194–215.
Noordermeer SDS, Luman M, Oosterlaan J. A systematic review anmeta-analysis of neuroimaging in Oppositional Defiant Disorder (ODD) and Conduct Disorder (CD) taking Attention-Deficit Hyperactivity Disorder (ADHD) into account. Neuropsychol Rev 2016;26:44–72.
Feil J, Sheppard D, Fitzgerald PB, Yücel M, Lubman DI, Bradshaw JL. Addiction, compulsive drug seeking, and the role of frontostriatal mechanisms in regulating inhibitory control. Neurosci Biobehav Rev 2010;35:248–75.
Becker MPI, Simon D, Miltner WHR, Straube T. Altered activation of the ventral striatum under performance-related observation in social anxiety disorder. Psychol Med. 2017;47:2502–12.
Wang L, Li F, Mitchell PB, Wang CY, Si TM. Striatal resting-state connectivity abnormalities associated with different clinical stages of major depressive disorder. J Clin Psychiatry. 2020. https://doi.org/10.4088/JCP.19m12790.
DelDonno SR, Jenkins LM, Crane NA, Nusslock R, Ryan KA, Shankman SA, et al. Affective traits and history of depression are related to ventral striatum connectivity. J Affect Disord. 2017;221:72–80.
Cools R. Dopaminergic control of the striatum for high-level cognition. Curr Opin Neurobiol 2011;21:402–7.
Ullman H, Spencer-Smith M, Thompson DK, Doyle LW, Inder TE, Anderson PJ, et al. Neonatal MRI is associated with future cognition and academic achievement in preterm children. Brain. 2015;138:3251–62.
Looi JCL, Walterfang M. Striatal morphology as a biomarker in neurodegenerative disease. Mol Psychiatry 2013;18:417–24.
Sullivan CRP, Olsen S, Widge AS. Deep brain stimulation for psychiatric disorders: from focal brain targets to cognitive networks. Neuroimage. 2021. https://doi.org/10.1016/J.NEUROIMAGE.2020.117515.
Hart MG, Price SJ, Suckling J. Connectome analysis for pre-operative brain mapping in neurosurgery. Br J Neurosurg. 2016;30:506–17.
Hacker CD, Roland JL, Kim AH, Shimony JS, Leuthardt EC. Resting-state network mapping in neurosurgical practice: A review. Neurosurg Focus. 2019;47:1–9.
Acknowledgements
AFM gratefully acknowledges support from the European Research Council (ERC, grant ‘MENTALPRECISION’ 10100118). The authors would like to thank professor Aart Schene for his continuous support in setting up the MIND-Set cohort. Without him, this work would not have been possible. Unfortunately professor Schene passed away too early to see the benefit of his continued support.
Author information
Authors and Affiliations
Contributions
PCRM: formulating research question, data analysis, writing article; PFPvE: formulating research question, designing study, writing article; JvO: carrying out study, writing article; MO: writing article; FAD: carrying out study, writing article; JDK: carrying out study, writing article; RMC: study design, writing article; JNV: study design, writing article; KVH: writing article; CFB: writing article; IT: formulating research question, study design, writing article; AFM: formulating research question, data analysis, writing article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mulders, P.C.R., van Eijndhoven, P.F.P., van Oort, J. et al. Striatal connectopic maps link to functional domains across psychiatric disorders. Transl Psychiatry 12, 513 (2022). https://doi.org/10.1038/s41398-022-02273-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-022-02273-6
- Springer Nature Limited
This article is cited by
-
A phylogenetically-conserved axis of thalamocortical connectivity in the human brain
Nature Communications (2023)