Abstract
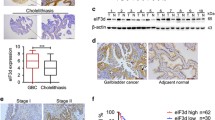
Orthodenticle homeobox (OTX1) is reported to be involved in numerous cancers, but the expression level and molecular function of OTX1 in gallbladder cancer (GBC) remain unknown. Here, we found the elevated level of OTX1 associated with poor prognosis in human gallbladder cancer. In vitro and in vivo studies of human gallbladder cancer cell lines demonstrated that overexpression of OTX1 promoted cell proliferation, whereas the downregulation inhibited it. Additionally, we found a tight correlation between the serum level of taurodeoxycholic acid (TDCA) and OTX1 expression. TDCA-induced activation of YAP1 by phosphorylation inhibition contributed to the transcriptional activation of OTX1. Mechanistically, we identified that OTX1 activated AKT signaling pathway by transactivating the expression of IFITM3 and thus promoted the proliferation of GBC cells. Taken together, our results showed that TDCA-YAP1-dependent expression of OTX1 regulated IFITM3 and affected GBC proliferation via the AKT signaling pathway. Our experiments also suggested that OTX1 is a novel therapeutic target for GBC.








Similar content being viewed by others

Data availability
The primary RNA-Seq data generated for this study will be available in a public, open-access repository with an accession number at the time of publication. The public datasets analyzed during the current study are available in the repositories listed below: The Cancer Genome Atlas: TCGA-LUSC; TCGA-LUAD; TCGA-COAD; TCGA-UCEC; TCGA-STAD; TCGA-BRCA; TCGA-PRAD; TCGA-BLCA; TCGA-LIHC; TCGA-CHOL; TCGA-ESCA; TCGA-PAAD; TCGA-KIRP; TCGA-KIRC; TCGA-THCA; TCGA-KICH; TCGA-HNSC; TCGA-ACC; TCGA-LGG; TCGA-MESO; TCGA-SARC. Gene Expression Omnibus: GSE62335; GSE76633; GSE100363; GSE139682.
References
Hundal R, Shaffer EA. Gallbladder cancer: epidemiology and outcome. Clin Epidemiol. 2014;6:99–109.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30.
Hickman L, Contreras C. Gallbladder cancer: diagnosis, surgical management, and adjuvant therapies. Surg Clin North Am. 2019;99:337–55.
Roa I, Araya JC, Villaseca M, De Aretxabala X, Riedemann P, Endoh K, et al. Preneoplastic lesions and gallbladder cancer: an estimate of the period required for progression. Gastroenterology. 1996;111:232–6.
Jia W, Xie G, Jia W. Bile acid-microbiota crosstalk in gastrointestinal inflammation and carcinogenesis. Nat Rev Gastroenterol Hepatol. 2018;15:111–28.
Wang S, Kuang J, Zhang H, Chen W, Zheng X, Wang J, et al. Bile acid-microbiome interaction promotes gastric carcinogenesis. Adv Sci. 2022;9:e2200263.
Liu J, Wei Y, Jia W, Can C, Wang R, Yang X, et al. Chenodeoxycholic acid suppresses AML progression through promoting lipid peroxidation via ROS/p38 MAPK/DGAT1 pathway and inhibiting M2 macrophage polarization. Redox Biol. 2022;56:102452.
Sharma N, Yadav M, Tripathi G, Mathew B, Bindal V, Falari S, et al. Bile multi-omics analysis classifies lipid species and microbial peptides predictive of carcinoma of gallbladder. Hepatology. 2022;76:920–35.
Klein WH, Li X. Function and evolution of Otx proteins. Biochem Biophys Res Commun. 1999;258:229–33.
Kastury K, Druck T, Huebner K, Barletta C, Acampora D, Simeone A, et al. Chromosome locations of human EMX and OTX genes. Genomics. 1994;22:41–45.
Larsen KB, Lutterodt MC, Mollgard K, Moller M. Expression of the homeobox genes OTX2 and OTX1 in the early developing human brain. J Histochem Cytochem. 2010;58:669–78.
Sakurai Y, Kurokawa D, Kiyonari H, Kajikawa E, Suda Y, Aizawa S. Otx2 and Otx1 protect diencephalon and mesencephalon from caudalization into metencephalon during early brain regionalization. Dev Biol. 2010;347:392–403.
Pagani IS, Terrinoni A, Marenghi L, Zucchi I, Chiaravalli AM, Serra V, et al. The mammary gland and the homeobox gene Otx1. Breast J. 2010;16:S53–56.
Yu K, Cai XY, Li Q, Yang ZB, Xiong W, Shen T, et al. OTX1 promotes colorectal cancer progression through epithelial-mesenchymal transition. Biochem Biophys Res Commun. 2014;444:1–5.
Terrinoni A, Pagani IS, Zucchi I, Chiaravalli AM, Serra V, Rovera F, et al. OTX1 expression in breast cancer is regulated by p53. Oncogene. 2011;30:3096–103.
Li H, Miao Q, Xu CW, Huang JH, Zhou YF, Wu MJ. OTX1 contributes to hepatocellular carcinoma progression by regulation of ERK/MAPK pathway. J Korean Med Sci. 2016;31:1215–23.
Qin SC, Zhao Z, Sheng JX, Xu XH, Yao J, Lu JJ, et al. Downregulation of OTX1 attenuates gastric cancer cell proliferation, migration and invasion. Oncol Rep. 2018;40:1907–16.
Kuhn T, Stepien M, Lopez-Nogueroles M, Damms-Machado A, Sookthai D, Johnson T, et al. Prediagnostic plasma bile acid levels and colon cancer risk: a prospective study. J Natl Cancer Inst. 2020;112:516–24.
Ma C, Han M, Heinrich B, Fu Q, Zhang Q, Sandhu M, et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells. Science 2018;360:876.
Anakk S, Bhosale M, Schmidt VA, Johnson RL, Finegold MJ, Moore DD. Bile acids activate YAP to promote liver carcinogenesis. Cell Rep. 2013;5:1060–9.
Lee CK, Jeong SH, Jang C, Bae H, Kim YH, Park I, et al. Tumor metastasis to lymph nodes requires YAP-dependent metabolic adaptation. Science. 2019;363:644–9.
de Boer JF, Bloks VW, Verkade E, Heiner-Fokkema MR, Kuipers F. New insights in the multiple roles of bile acids and their signaling pathways in metabolic control. Curr Opin Lipidol. 2018;29:194–202.
Liu R, Li X, Hylemon PB, Zhou H. Conjugated bile acids promote invasive growth of esophageal adenocarcinoma cells and cancer stem cell expansion via Sphingosine 1-Phosphate Receptor 2-Mediated Yes-associated protein activation. Am J Pathol. 2018;188:2042–58.
Garcia P, Rosa L, Vargas S, Weber H, Espinoza JA, Suarez F, et al. Hippo-YAP1 is a prognosis marker and potentially targetable pathway in advanced gallbladder cancer. Cancers 2020;12:778.
Li M, Lu J, Zhang F, Li H, Zhang B, Wu X, et al. Yes-associated protein 1 (YAP1) promotes human gallbladder tumor growth via activation of the AXL/MAPK pathway. Cancer Lett. 2014;355:201–9.
Liu-Chittenden Y, Huang B, Shim JS, Chen Q, Lee SJ, Anders RA, et al. Genetic and pharmacological disruption of the TEAD-YAP complex suppresses the oncogenic activity of YAP. Genes Dev. 2012;26:1300–5.
Martin D, Degese MS, Vitale-Cross L, Iglesias-Bartolome R, Valera JLC, Wang Z, et al. Assembly and activation of the Hippo signalome by FAT1 tumor suppressor. Nat Commun. 2018;9:2372.
Szulzewsky F, Holland EC, Vasioukhin V. YAP1 and its fusion proteins in cancer initiation, progression and therapeutic resistance. Dev Biol. 2021;475:205–21.
Bo X, Wang J, Wang C, Nan L, Gao Z, Xin Y, et al. High infiltration of mast cells is associated with improved response to adjuvant chemotherapy in gallbladder cancer. Cancer Sci. 2020;111:817–25.
Dent P, Fang Y, Gupta S, Studer E, Mitchell C, Spiegel S, et al. Conjugated bile acids promote ERK1/2 and AKT activation via a pertussis toxin-sensitive mechanism in murine and human hepatocytes. Hepatology. 2005;42:1291–9.
Zhou L, Li H, Zhang D, Chen L, Dong H, Yuan Y, et al. OTX1 promotes tumorigenesis and progression of cervical cancer by regulating the Wnt signaling pathway. Oncol Rep. 2022;48:204.
Chai J, Xu T, Yang Y, Yuan Y, Xu J, Liu J, et al. Overexpression of OTX1 promotes tumorigenesis in patients with esophageal squamous cell carcinoma. Pathol Res Pract. 2022;232:153841.
Jiang L, Zuo Z, Lin J, Yang C. Orthodenticle homeobox OTX1 is a potential prognostic biomarker for bladder cancer. Bioengineered. 2021;12:6559–71.
Funabashi M, Grove TL, Wang M, Varma Y, McFadden ME, Brown LC, et al. A metabolic pathway for bile acid dehydroxylation by the gut microbiome. Nature. 2020;582:566–70.
Schramm C. Bile acids, the microbiome, immunity, and liver tumors. N Engl J Med. 2018;379:888–90.
Lin R, Zhan M, Yang L, Wang H, Shen H, Huang S, et al. Deoxycholic acid modulates the progression of gallbladder cancer through N(6)-methyladenosine-dependent microRNA maturation. Oncogene. 2020;39:4983–5000.
Bai X, Wei H, Liu W, Coker OO, Gou H, Liu C, et al. Cigarette smoke promotes colorectal cancer through modulation of gut microbiota and related metabolites. Gut 2022;71:2439–50.
Hong J, Behar J, Wands J, Resnick M, Wang LJ, Delellis RA, et al. Bile acid reflux contributes to development of esophageal adenocarcinoma via activation of phosphatidylinositol-specific phospholipase Cgamma2 and NADPH oxidase NOX5-S. Cancer Res. 2010;70:1247–55.
Ballout F, Lu H, Chen L, Sriramajayam K, Que J, Meng Z, et al. APE1 redox function is required for activation of Yes-associated protein 1 under reflux conditions in Barrett’s-associated esophageal adenocarcinomas. J Exp Clin Cancer Res. 2022;41:264.
Pepe-Mooney BJ, Dill MT, Alemany A, Ordovas-Montanes J, Matsushita Y, Rao A, et al. Single-cell analysis of the liver epithelium reveals dynamic heterogeneity and an essential role for YAP in homeostasis and regeneration. Cell Stem Cell. 2019;25:23–38.e28.
Yu B, Su J, Shi Q, Liu Q, Ma J, Ru G, et al. KMT5A-methylated SNIP1 promotes triple-negative breast cancer metastasis by activating YAP signaling. Nat Commun. 2022;13:2192.
Chen Q, Wang H, Li Z, Li F, Liang L, Zou Y, et al. Circular RNA ACTN4 promotes intrahepatic cholangiocarcinoma progression by recruiting YBX1 to initiate FZD7 transcription. J Hepatol. 2022;76:135–47.
Yang L, Chen J, Liang J, Zhang Y, Wang Q, Ren X, et al. Modeling hepatoblastoma development with human fetal liver organoids reveals YAP1 activation is sufficient for tumorigenesis. Protein Cell. 2022;13:683–8.
Chen X, Li Y, Luo J, Hou N. Molecular mechanism of Hippo-YAP1/TAZ pathway in heart development, disease, and regeneration. Front Physiol. 2020;11:389.
Perra A, Kowalik MA, Ghiso E, Ledda-Columbano GM, Di Tommaso L, Angioni MM, et al. YAP activation is an early event and a potential therapeutic target in liver cancer development. J Hepatol. 2014;61:1088–96.
Hou Y, Wang S, Gao M, Chang J, Sun J, Qin L, et al. Interferon-Induced Transmembrane Protein 3 Expression upregulation is involved in progression of hepatocellular carcinoma. Biomed Res Int. 2021;2021:5612138.
Coomer CA, Rahman K, Compton AA. CD225 proteins: a family portrait of fusion regulators. Trends Genet. 2021;37:406–10.
Shi G, Schwartz O, Compton AA. More than meets the I: the diverse antiviral and cellular functions of interferon-induced transmembrane proteins. Retrovirology. 2017;14:53.
Zhao X, Li J, Winkler CA, An P, Guo JT. IFITM genes, variants, and their roles in the control and pathogenesis of viral infections. Front Microbiol. 2018;9:3228.
Chu PY, Huang WC, Tung SL, Tsai CY, Chen CJ, Liu YC, et al. IFITM3 promotes malignant progression, cancer stemness and chemoresistance of gastric cancer by targeting MET/AKT/FOXO3/c-MYC axis. Cell Biosci. 2022;12:124.
Liu X, Chen L, Fan Y, Hong Y, Yang X, Li Y, et al. IFITM3 promotes bone metastasis of prostate cancer cells by mediating activation of the TGF-beta signaling pathway. Cell Death Dis. 2019;10:517.
Li D, Peng Z, Tang H, Wei P, Kong X, Yan D, et al. KLF4-mediated negative regulation of IFITM3 expression plays a critical role in colon cancer pathogenesis. Clin Cancer Res. 2011;17:3558–68.
Yang M, Gao H, Chen P, Jia J, Wu S. Knockdown of interferon-induced transmembrane protein 3 expression suppresses breast cancer cell growth and colony formation and affects the cell cycle. Oncol Rep. 2013;30:171–8.
Min J, Hu J, Luo C, Zhu J, Zhao J, Zhu Z, et al. IFITM3 upregulates c-myc expression to promote hepatocellular carcinoma proliferation via the ERK1/2 signalling pathway. Biosci Trends. 2020;13:523–9.
Acknowledgements
We are grateful to our laboratory members and professors for their insightful comments. This work was supported by the National Natural Science Foundation of China (Grant number: 82072668).
Author information
Authors and Affiliations
Contributions
WW, HW, and CCZ conceived the project. RFY, LHY, MML, and FYW designed the experiments. RFY, LHY, MML, and FYW performed most of the experiments. YHS, RX, and JJL assisted with the animal experiments and data collection. YYZ, YL, and YJ provided technical suggestions. RFY and FYW wrote the first version of the manuscript. WW and HW revised the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, R., Yang, L., Long, M. et al. Taurodeoxycholic acid-YAP1 upregulates OTX1 in promoting gallbladder cancer malignancy through IFITM3-dependent AKT activation. Oncogene 42, 1466–1477 (2023). https://doi.org/10.1038/s41388-023-02660-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-023-02660-3
- Springer Nature Limited