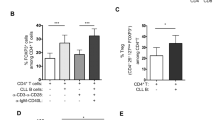
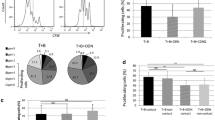
Abstract
Precursor B cell acute lymphoblastic leukemia (BCP-ALL) constitutes the leading cause of cancer-related death in children. While chromosomal alterations contribute to BCP-ALL pathogenesis, they are insufficient for leukemia development. Epidemiological data and evidence from a mouse model suggest that immune responses to infections may trigger the emergence of leukemia, but the mechanisms remain unclear. Here, we show that T helper (Th) cells from bone marrow of pediatric BCP-ALL patients can be attracted and activated by autologous BCP-ALL cells. Bone-marrow Th cells supportively interacted with BCP-ALL cells, inducing upregulation of important surface molecules and BCP-ALL cell proliferation. These Th cells displayed a Th1-like phenotype and produced high levels of IFN-γ. IFN-γ was responsible for the upregulation of CD38 in BCP-ALL cells, a molecule which we found to be associated with early relapse, and accountable for the production of IP-10, a chemokine involved in BCP-ALL migration and drug resistance. Thus, our data provide mechanistic support for an involvement of Th cell immune responses in the propagation of BCP-ALL and suggest that BCP-ALL cell-supportive Th cells may serve as therapeutic target.





Similar content being viewed by others
References
Pui CH, Robison LL, Look AT. Acute lymphoblastic leukaemia. Lancet. 2008;371:1030–43.
Ko RH, Ji L, Barnette P, Bostrom B, Hutchinson R, Raetz E. et al. Outcome of patients treated for relapsed or refractory acute lymphoblastic leukemia: a Therapeutic Advances in Childhood Leukemia Consortium study. J Clin Oncol. 2010;28:648–54.
Raetz EA, Bhatla T. Where do we stand in the treatment of relapsed acute lymphoblastic leukemia?. Hematol Am Soc Hematol Educ Program. 2012;2012:129–36.
Oeffinger KC, Mertens AC, Sklar CA, Kawashima T, Hudson MM, Meadows AT. et al. Chronic health conditions in adult survivors of childhood cancer. N Engl J Med. 2006;355:1572–82.
Ford AM, Bennett CA, Price CM, Bruin MC, Van Wering ER, Greaves M. Fetal origins of the TEL-AML1 fusion gene in identical twins with leukemia. Proc Natl Acad Sci USA. 1998;95:4584–8.
Mori H, Colman SM, Xiao Z, Ford AM, Healy LE, Donaldson C. et al. Chromosome translocations and covert leukemic clones are generated during normal fetal development. Proc Natl Acad Sci USA. 2002;99:8242–7.
Greaves M. Infection, immune responses and the aetiology of childhood leukaemia. Nat Rev Cancer. 2006;6:193–203.
Richardson RB. Promotional etiology for common childhood acute lymphoblastic leukemia: the infective lymphoid recovery hypothesis. Leuk Res. 2011;35:1425–31.
Greaves M. A causal mechanism for childhood acute lymphoblastic leukaemia. Nat Rev Cancer. 2018;8:471–84.
Kinlen L. Evidence for an infective cause of childhood leukaemia: comparison of a Scottish new town with nuclear reprocessing sites in Britain. Lancet. 1988;2:1323–7.
Martin-Lorenzo A, Hauer J, Vicente-Duenas C, Auer F, Gonzalez-Herrero I, Garcia-Ramirez I. et al. Infection exposure is a causal factor in B cell precursor acute lymphoblastic leukemia as a result of Pax5-inherited susceptibility. Cancer Discov. 2015;12:1328–43.
MacKenzie J, Greaves MF, Eden TO, Clayton RA, Perry J, Wilson KS, et al. The putative role of transforming viruses in childhood acute lymphoblastic leukemia. Haematologica. 2006;91:240–3.
Purizaca J, Meza I, Pelayo R. Early lymphoid development and microenvironmental cues in B cell acute lymphoblastic leukemia. Arch Med Res. 2012;43:89–101.
Boyerinas B, Zafrir M, Yesilkanal AE, Price TT, Hyjek EM, Sipkins DA. Adhesion to osteopontin in the bone marrow niche regulates lymphoblastic leukemia cell dormancy. Blood. 2013;121:4821–31.
Fei F, Joo EJ, Tarighat SS, Schiffer I, Paz H, Fabbri M, et al. B-cell precursor acute lymphoblastic leukemia and stromal cells communicate through Galectin-3. Oncotarget. 2015;6:11378–94.
Fortney JE, Zhao W, Wenger SL, Gibson LF. Bone marrow stromal cells regulate caspase 3 activity in leukemic cells during chemotherapy. Leuk Res. 2001;25:901–7.
Duan CW, Shi J, Chen J, Wang B, Yu YH, Qin X, et al. Leukemia propagating cells rebuild an evolving niche in response to therapy. Cancer Cell. 2014;25:778–93.
Iwamoto S, Mihara K, Downing JR, Pui CH, Campana D. Mesenchymal cells regulate the response of acute lymphoblastic leukemia cells to asparaginase. J Clin Invest. 2007;117:1049–57.
Mudry RE, Fortney JE, York T, Hall BM, Gibson LF. Stromal cells regulate survival of B-lineage leukemic cells during chemotherapy. Blood. 2000;96:1926–32.
Feuerer M, Beckhove P, Bai L, Solomayer EF, Bastert G, Diel IJ. et al. Therapy of human tumors in NOD/SCID mice with patient-derived reactivated memory T-cells from bone-marrow. Nat Med. 2001;7:452–8.
Dhodapkar MV, Krasovsky J, Osman K, Geller MD. Vigorous premalignancy-specific effector T-cell response in the bone-marrow of patients with monoclonal gammopathy. J Exp Med. 2003;198:1753–7.
Chen JJ, Huang JC, Shirtliff M, Briscoe E, Ali S, Cesani F, et al. CD4 lymphocytes in the blood of HIV(+) individuals migrate rapidly to lymph nodes and bone-marrow: support for homing theory of CD4 cell depletion. J Leukoc Biol. 2002;72:271–8.
Herndler-Brandstetter D, Landgraf K, Jenewein B, Tzankov A, Brunauer R, Brunner S, et al. Human bone-marrow hosts polyfunctional memory CD4+and CD8+T-cells with close contact to IL-15-producing cells. J Immunol. 2011;186:6965–71.
Okhrimenko A, Grun JR, Westendorf K, Fang Z, Reinke S, von Roth P, et al. Human memory T-cells from the bone-marrow are resting and maintain long-lasting systemic memory. Proc Natl Acad Sci USA. 2014;111:9229–34.
Tokoyoda K, Zehentmeier S, Hegazy AN, Albrecht I, Grun JR, Lohning M, et al. Professional memory CD4+T lymphocytes preferentially reside and rest in the bone-marrow. Immunity. 2009;30:721–30.
Mosmann TR, Cherwinski H, Bond MW, Giedlin MA, Coffman RL. Two types of murine helper T-cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986;136:2348–57.
Szabo SJ, Kim ST, Costa GL, Zhang X, Fathman CG, Glimcher LH. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell. 2000;100:655–69.
Zheng W, Flavell RA. The transcription factor GATA-3 is necessary and sufficient for Th2 cytokine gene expression in CD4 T-cells. Cell. 1997;89:587–96.
Yamashita M, Ukai-Tadenuma M, Miyamoto T, Sugaya K, Hosokawa H, Hasegawa A, et al. Essential role of GATA3 for the maintenance of type 2 helper T (Th2) cytokine production and chromatin remodeling at the Th2 cytokine gene loci. J Biol Chem. 2004;279:26983–90.
Pai SY, Truitt ML, Ho IC. GATA-3 deficiency abrogates the development and maintenance of T helper type 2 cells. Proc Natl Acad Sci USA. 2004;101:1993–8.
Liston A, Gray DH. Homeostatic control of regulatory T-cell diversity. Nat Rev Immunol. 2014;14:154–65.
Fluckiger AC, Rossi JF, Bussel A, Bryon P, Banchereau J, Defrance T. Responsiveness of chronic lymphocytic leukemia B cells activated via surface Igs or CD40 to B cell tropic factors. Blood. 1992;80:3173–81.
Buske C, Gogowski G, Schreiber K, Rave-Frank M, Hiddemann W, Wormann B. Stimulation of B-chronic lymphocytic leukemia cells by murine fibroblasts, IL-4, anti-CD40 antibodies, and the soluble CD40 ligand. Exp Hematol. 1997;25:329–37.
Umetsu DT, Esserman L, Donlon TA, DeKruyff RH, Levy R. Induction of proliferation of human follicular (B type) lymphoma cells by cognate interaction with CD4+T-cell clones. J Immunol. 1990;144:2550–7.
Pangault C, Ame-Thomas P, Ruminy P, Rossille D, Caron G, Baia M, et al. Follicular lymphoma cell niche: identification of a preeminent IL-4-dependent T(FH)-B cell axis. Leukemia. 2010;24:2080–9.
Johnson PW, Watt SM, Betts DR, Davies D, Jordan S, Norton AJ, et al. Isolated follicular lymphoma cells are resistant to apoptosis and can be grown in vitro in the CD40/stromal cell system. Blood. 1993;82:1848–57.
Wang D, Floisand Y, Myklebust CV, Burgler S, Parente-Ribes A, Hofgaard PO, et al. Autologous bone marrow Th cells can support multiple myeloma cell proliferation in vitro and in xenografted mice. Leukemia. 2017;31:2114–21.
Os A, Burgler S, Ribes AP, Funderud A, Wang D, Thompson KM, et al. Chronic lymphocytic leukemia cells are activated and proliferate in response to specific T helper cells. Cell Rep. 2013;4:566–77.
Burgler S, Gimeno A, Parente-Ribes A, Wang D, Os A, Devereux S, et al. Chronic lymphocytic leukemia cells express CD38 in response to Th1-cell-derived IFN-gamma by a T-bet-dependent mechanism. J Immunol. 2015;194:827–35.
Renard N, Lafage-Pochitaloff M, Durand I, Duvert V, Coignet L, Banchereau J, et al. Demonstration of functional CD40 in B-lineage acute lymphoblastic leukemia cells in response to T-cell CD40 ligand. Blood. 1996;87:5162–70.
Cardoso AA, Schultze JL, Boussiotis VA, Freeman GJ, Seamon MJ, Laszlo S, et al. Pre-B acute lymphoblastic leukemia cells may induce T-cell anergy to alloantigen. Blood. 1996;88:41–8.
Reid GS, Terrett L, Alessandri AJ, Grubb S, Stork L, Seibel N, et al. Altered patterns of T-cell cytokine production induced by relapsed pre-B ALL cells. Leuk Res. 2003;27:1135–42.
Wu S, Gessner R, von Stackelberg A, Kirchner R, Henze G, Seeger K. Cytokine/cytokine receptor gene expression in childhood acute lymphoblastic leukemia: correlation of expression and clinical outcome at first disease recurrence. Cancer. 2005;103:1054–63.
Nakase K, Kita K, Miwa H, Nishii K, Shikami M, Tanaka I, et al. Clinical and prognostic significance of cytokine receptor expression in adult acute lymphoblastic leukemia: interleukin-2 receptor alpha-chain predicts a poor prognosis. Leukemia. 2007;21:326–32.
Burgler S. Role of CD38 expression in diagnosis and pathogenesis of chronic lymphocytic leukemia and its potential as therapeutic target. Crit Rev Immunol. 2015;35:417–32.
Liu M, Guo S, Hibbert JM, Jain V, Singh N, Wilson NO, et al. CXCL10/IP-10 in infectious diseases pathogenesis and potential therapeutic implications. Cytokine Growth Factor Rev. 2011;22:121–30.
Lee Y, Chittezhath M, Andre V, Zhao H, Poidinger M, Biondi A, et al. Protumoral role of monocytes in human B-cell precursor acute lymphoblastic leukemia: involvement of the chemokine CXCL10. Blood. 2012;119:227–37.
Gomez AM, Martinez C, Gonzalez M, Luque A, Melen GJ, Martinez J, et al. Chemokines and relapses in childhood acute lymphoblastic leukemia: a role in migration and in resistance to antileukemic drugs. Blood Cells Mol Dis. 2015;55:220–7.
Ghia P, Transidico P, Veiga JP, Schaniel C, Sallusto F, Matsushima K, et al. Chemoattractants MDC and TARC are secreted by malignant B cell precursors following CD40 ligation and support the migration of leukemia-specific T-cells. Blood. 2001;98:533–40.
McHeyzer-Williams LJ, Malherbe LP, McHeyzer-Williams MG. Helper T-cell-regulated B cell immunity. Curr Top Microbiol Immunol. 2006;311:59–83.
Taylor GM, Hussain A, Verhage V, Thompson PD, Fergusson WD, Watkins G, et al. Strong association of the HLA-DP6 supertype with childhood leukaemia is due to a single allele, DPB1*0601. Leukemia. 2009;23:863–9.
Thompson P, Urayama K, Zheng J, Yang P, Ford M, Buffler P. et al. Differences in meiotic recombination rates in childhood acute lymphoblastic leukemia at an MHC class II hotspot close to disease associated haplotypes. PLoS ONE. 2014;9:e100480
Einav U, Tabach Y, Getz G, Yitzhaky A, Ozbek U, Amariglio N, et al. Gene expression analysis reveals a strong signature of an interferon-induced pathway in childhood lymphoblastic leukemia as well as in breast and ovarian cancer. Oncogene. 2005;24:6367–75.
Zaki M, Douglas R, Patten N, Bachinsky M, Lamb R, Nowell P, et al. Disruption of the IFN-gamma cytokine network in chronic lymphocytic leukemia contributes to resistance of leukemic B cells to apoptosis. Leuk Res. 2000;24:611–21.
Buschle M, Campana D, Carding SR, Richard C, Hoffbrand AV, Brenner MK. Interferon gamma inhibits apoptotic cell death in B cell chronic lymphocytic leukemia. J Exp Med. 1993;177:213–8.
Rakoff-Nahoum S. Why cancer and inflammation? Yale J Biol Med. 2006;79:123–30.
Baldridge MT, King KY, Boles NC, Weksberg DC, Goodell MA. Quiescent haematopoietic stem cells are activated by IFN-gamma in response to chronic infection. Nature. 2010;465:793–7.
Hersher R. Companies wager high on CD38-targeting drugs for blood cancer. Nat Med. 2012;18:1446.
Deckert J, Wetzel MC, Bartle LM, Skaletskaya A, Goldmacher VS, Vallee F, et al. SAR650984, a novel humanized CD38-targeting antibody, demonstrates potent antitumor activity in models of multiple myeloma and other CD38+hematologic malignancies. Clin Cancer Res. 2014;20:4574–83.
van der Veer MS, de Weers M, van Kessel B, Bakker JM, Wittebol S, Parren PW, et al. The therapeutic human CD38 antibody daratumumab improves the anti-myeloma effect of newly emerging multi-drug therapies. BloodCancer J. 2011;1:e41.
Nijhof IS, Groen RW, Noort WA, van Kessel B, de Jong-Korlaar R, Bakker J. et al. Preclinical evidence for the therapeutic potential of CD38-targeted immuno-chemotherapy in multiple myeloma patients refractory to lenalidomide and bortezomib. Clin Cancer Res. 2014;21:2802–10.
Lokhorst HM, Plesner T, Laubach JP, Nahi H, Gimsing P, Hansson M. et al. Targeting CD38 with daratumumab monotherapy in multiple myeloma. N Engl J Med. 2015;373:1207–19.
Vaisitti T, Audrito V, Serra S, Buonincontri R, Sociali G, Mannino E, et al. The enzymatic activities of CD38 enhance CLL growth and trafficking: implications for therapeutic targeting. Leukemia. 2015;29:356–68.
Chillemi A, Zaccarello G, Quarona V, Lazzaretti M, Martella E, Giuliani N, et al. CD38 and bone-marrow microenvironment. Front Biosci. 2014;19:152–62.
Kreis C, Lupatsch JE, Niggli F, Egger M, Kuehni CE, Spycher BD, et al. Space-time clustering of childhood leukemia: evidence of an association with ETV6-RUNX1 (TEL-AML1) fusion. PLoS ONE 2017;12:e0170020.
Hogan LE, Meyer JA, Yang J, Wang J, Wong N, Yang W, et al. Integrated genomic analysis of relapsed childhood acute lymphoblastic leukemia reveals therapeutic strategies. Blood. 2011;118:5218–26.
Acknowledgements
We thank the pediatric patients and donors who provided the clinical samples. This work was supported by the Forschungskredit of the University of Zürich, The Children’s Research Center (CRC) of the Children’s Hospital Zurich, the Stiftung für Krebsbekämpfung and the Krebsliga Schweiz/Swiss Cancer League (grant number KLS 3189-02-2013), the Krebsliga Schweiz/Swiss Cancer League (grant number KLS 3189-02-2013) and the Charles Meyer Cancer Research Initiative.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Traxel, S., Schadt, L., Eyer, T. et al. Bone marrow T helper cells with a Th1 phenotype induce activation and proliferation of leukemic cells in precursor B acute lymphoblastic leukemia patients. Oncogene 38, 2420–2431 (2019). https://doi.org/10.1038/s41388-018-0594-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0594-4
- Springer Nature Limited
This article is cited by
-
In Utero Development and Immunosurveillance of B Cell Acute Lymphoblastic Leukemia
Current Treatment Options in Oncology (2022)
-
Support of BCP-ALL-cells by autologous bone marrow Th-cells involves induction of AID expression but not widespread AID off-target mutagenesis
Cancer Immunology, Immunotherapy (2021)