Abstract
Maintaining appropriate levels of fear memory specificity is crucial for individual’s survival and mental health, whereas overgeneralized fear commonly occurs in neuropsychiatric disorders, including posttraumatic stress disorder and generalized anxiety disorder. However, the molecular mechanisms regulating fear memory specificity remain poorly understood. The medial prefrontal cortex (mPFC) is considered as a key brain region in fear memory regulation. Previous transcriptomic studies have identified that plexin-A1, a transmembrane receptor critical for axon development, was downregulated in the mPFC after fear memory training. In this study, we identified that learning-induced downregulation of the mRNA and protein levels of plexin-A1 specifically occurred in the inhibitory but not excitatory neurons in the infralimbic cortex (IL) of mPFC. Further studies of plexin-A1 by virus-mediated over-expression of functional mutants selectively in the IL inhibitory neurons revealed the critical roles of plexin-A1 for regulating memory specificity and anxiety. Moreover, our findings revealed that plexin-A1 regulated the distribution of glutamic acid decarboxylase 67, a GABA synthetase, which in turn modulated the activity of IL and its downstream brain regions. Collectively, our findings elucidate the molecular modifier of IL inhibitory neurons in regulating memory specificity and anxiety, and provide candidates for developing therapeutic strategies for the prevention or treatment of a series of fear generalization-related neuropsychiatric disorders.
Similar content being viewed by others
Introduction
The strength and specificity of memories are two fundamental features that determine the explicit property of prior experiences encoded in the brain. Over the past decades, while considerable advances have been obtained in uncovering the neural circuit and molecular mechanisms for regulating memory strength [1, 2], the mechanisms that regulate memory specificity remain underexplored. Yet, maintaining the balance between memory specificity and generalization is crucial for individual’s survival and mental health. While memory specificity refers to the ability to retrieve details of past experiences in order to discriminate between similar yet different conditions, memory generalization, on the contrary, allows individuals to generate learned response in a modified environment that partially resembles prior experiences. An appropriate level of memory generalization could protect individuals from potential dangers in an environment that partially resembles their previous aversive experiences, whereas over-generalization of fear is a core symptom of generalized anxiety disorder and posttraumatic stress disorder (PTSD) [3,4,5,6,7].
For contextual or episodic-like memories, the initial phase of memory consolidation, which stabilizes memory traces after acquisition, requires de novo gene expression in the hippocampus [8,9,10,11]. It is hypothesized that memories are stored in the cortex, and the ultimate storage of memory requires functional interactions between the hippocampus and the cortex [12]. Emerging evidences reveal that the medial prefrontal cortex (mPFC), which can be further divided into the prelimbic cortex (PL) and the infralimbic cortex (IL) in rodents, is critically implicated in memory processing [13,14,15,16,17]. The mPFC receives both direct and indirect projections from the hippocampus [18,19,20,21]. Recent studies have found that mPFC is activated by various contextual fear training paradigms [22, 23]. Blocking gene expression in the mPFC immediately after training resulted in impaired strength of contextual fear memory [23, 24]. Interestingly, inactivation of the mPFC, especially inactivation of the IL subregion during training enhanced generalization of fear memories, without affecting memory strength [25,26,27]. While recent studies have revealed numerous molecular and cellular mechanisms in the mPFC for the regulation of memory [15,16,17], it remains unclear how memory strength versus specificity are differentially mediated by distinct mechanisms in the mPFC.
Recent transcriptomic studies have identified differentially expressed genes (DEGs) in both hippocampus and mPFC that may subserve memory formation [28,29,30]. Unlike the hippocampus where DEGs after training were mostly upregulated, a large proportion of DEGs in the mPFC were downregulated [30]. Notably, the DEGs in the mPFC were enriched for regulation of axonogenesis, including plexin-A1 (Plxna1), a transmembrane receptor mediating semaphorin (SEMA) signaling [28, 30, 31]. PLXNA1 has been reported to induce growth cone collapse through cytoskeleton rearrangement and membrane endocytosis in cultured cells upon binding to its ligand SEMA3A [32]. In vivo, interaction of PLXNA1 with SEMA6D regulated the development of neurons in the heart and retinal ganglia during embryonic development [33,34,35]. However, the function of PLXNA1 in the adult brain, in particular its regulatory role in memory formation, remains unknown.
In the current study, we explored the spatiotemporal changes of Plxna1 expression in the mPFC of mice after contextual fear conditioning (CFC) and its regulatory role in memory formation. Our findings unexpectedly revealed the critical role of training-induced reduction of PLXNA1 in the IL inhibitory neurons for maintaining memory specificity, without affecting the strength of CFC memory. Functional alteration of PLXNA1 protein was found to modulate the distribution of GABA synthetase, glutamate decarboxylase 67 (GAD67), and modify the activities of the IL and its downstream brain regions. Our study therefore provides novel mechanistic insights for the molecular tuning factor of the IL inhibitory neurons that regulates memory specificity and the therapeutic targets for related neuropsychiatric disorders.
Materials and methods
Animals
Adult male C57BL/6J mice (8–12 weeks old) were obtained from the Institute of Experimental Animals of Sun Yat-sen University (Guangzhou, China). All mice were housed in groups of 4–5 in a specific pathogen free animal facility with 12 h light/dark cycle, temperature maintained at 23 ± 2 °C and humidity at 50–60%. Food and water were available ad libitum. All animal studies were approved by the Institutional Animal Care and Use Committee of Sun Yat-Sen University.
Viral vectors and stereotaxic viral injection
The AAVs expressing Plxna1 mutants were custom-made by OBiO (Shanghai, China). PCR-amplified fragments of Plxna1 cDNA encoding amino acid sequence 1218–1874 (Plxna1Δect, encoding PLXNA1 lacking the ectodomain) and 1266–1874 (Plxna1cyto, encoding the cytoplasmic domain of PLXNA1) were inserted into the pAAV-mDlx-HA-MCS backbone vector (HA: the hemagglutinin epitope tag; MCS: multiple cloning site) for constructing the pAAV-mDlx-HA-Plxna1ΔEct and the pAAV-mDlx-HA-Plxna1cyto plasmids, respectively [36]. The mDlx promoter used in the viral construct contains the mDlx enhancer derived from the distal-less homeobox 5 and 6 (Dlx5/6) genes in front of a minimal promoter to drive transgene expression restrictively in GABAergic interneurons, as reported by Dimidschstein et al. [37]. The plasmids were packaged into AAV serotype (AAV8). The AAVs were injected bilaterally into the IL subregion of mPFC. Detailed descriptions of stereotaxic injection are provided in the Supplementary Methods.
Contextual fear conditioning (CFC)
The animals were handled for 2–3 min/day for 4 days before training. CFC training was carried out in context A, which was a rectangular chamber (25 × 25 × 45 cm) with red-and-white-striped walls, illuminated with 90 lux red light. The floor was made of stainless steel rods connected to a shock delivery apparatus (Med Associates, St. Albans, Vermont, USA). The chamber was placed in an acoustically-insulated cabinet and cleaned with 75% alcohol between animals. During CFC training, the animals were allowed to explore context A for 3 min, after which three 0.75 mA, 2 s foot-shocks were delivered with 1 min inter-shock intervals, controlled by the Video Freeze Software (Med Associates, St. Albans, Vermont, USA). After the last shock, the animals stayed in the chamber for an additional minute before being returned to their home-cages. For the immediate-shock protocol, the animals were placed into the context A, received three 0.75 mA, 2 s foot-shocks with 3 s inter-shock intervals, and immediately retrieved from the context and returned to home-cage after the last shock. The next two days, the animals were tested sequentially in context A and a modified context (context B) for memory strength and specificity at an interval of 24 h, respectively (the order of the two tests was counter-balanced). For context B, the chamber was modified with a curved white wall insert and a white acrylic floor, and illuminated with 60 lux red light. Freezing behavior was videotaped and measured by an examiner blinded to the experimental groups. Freezing was defined as the absence of any visible movements other than those necessary for respiration. The percentage of time spent in freezing during the 3 min tests was measured as previously described [38, 39].
Open field test
The locomotor activities of the mice were recorded in an open-field arena (40 × 40 cm) with 90 lux red light illumination. The mice were gently placed onto the center of the floor and allowed to explore the arena for 5 min. The total distance traveled in the open field, as well as the distance traveled in the center zone (20 × 20 cm) were recorded and analyzed using the EthoVision XT software (Noldus, Wageningen, Netherlands) as previously described [40].
Immunofluorescence staining, RNAscope and image analyses
Mice were transcardially perfused with 4% paraformaldehyde in PBS, 2 days after the completion of behavioral tests. Cryosections of the brains were collected for staining. Detailed descriptions are included in the Supplementary Methods.
RNA extraction and qPCR
Mice were euthanized by rapid cervical dislocation after CFC training, at the timepoints specified in the Result section. The brains were sliced into 1 mm sections using a brain matrix (RWD Life Science, Shenzhen, China) in ice-cold dissection buffer. The mPFC was quickly punched out using a 15 G puncher and snap-frozen on dry ice. Total RNA was isolated using Trizol (Thermo Scientific, Waltham, MA) following the manufacturer’s protocol. 500 ng RNA from each sample was reverse-transcribed using the QuantiTect Reverse Transcription Kit (Qiagen, Venlo, Netherlands), followed by qPCR analysis as described previously [38, 40]. Detailed descriptions are provided in the Supplementary Methods.
Statistical analysis
Data were analyzed by GraphPad Prism v.7 (GraphPad Software, San Diego, California, USA), and presented as the mean ± s.e.m. The two-tailed unpaired Student’s t test was used to compare two groups, and one-way analysis of variance (ANOVA) or two-way ANOVA followed by Dunnett’s or Sidak’s post hoc tests were performed for multiple comparisons. The significance level was set to p < 0.05.
Results
CFC training reduces Plxna1 expression in the mPFC
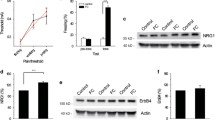
A previous transcriptomic study reported that inhibitory avoidance training in rats resulted in decreased expression of genes implicated in axonogenesis, including Plxna1 in the mPFC [30]. To explore the temporal course of the change and to examine whether the decrease also occurs after a different training paradigm and in a different species, we performed qPCR analyses of the relative mRNA levels of Plxna1 in the mPFC samples collected at 30 min, 1 h and 3 h after CFC training in mice, or at the corresponding time points for the naïve control animals (Fig. 1A). Mice that was trained with CFC reliably showed significantly enhanced freezing to the training context during long-term memory test at 24 h after training, suggesting that they were able to form an association between the training context and the foot-shocks (Supplementary Fig. 1A, B). Our data showed that the mRNA levels of Plxna1 remained unchanged at 30 min, but significantly decreased at both 1 h and 3 h after CFC training (Fig. 1B). The temporal profile of the Plxna1 mRNA changes was different from that of the c-fos mRNA, a well-characterized immediate-early gene product [41, 42], which showed increased expression at 30 min and 1 h after CFC training, but returned to baseline by 3 h in the same samples (Fig. 1C). Furthermore, Plxna1 mRNA level did not decreased at 1 h after training in the mPFC of mice that received immediate foot-shocks, which also failed to form long-term CFC memory (Supplementary Fig. 1), indicating that the reduction in Plxna1 mRNA level after CFC training was related to fear learning rather than foot-shock experience.
A Schematic of CFC training and sample collection. The animals in the CFC group were trained and sacrificed at 30 min, 1 h or 3 h after training. The animals in the naïve (N) control group were sacrificed in parallel at the corresponding time points. Since there was no difference in the Plxna1 mRNA level in the control animals collected at different time points, the qPCR results were pooled together as one N group. B, C Fold changes in Plxna1 and c-fos mRNA levels in the mPFC at 30 min, 1 h and 3 h post-CFC training, compared to those in the mPFC of naïve mice (n = 7–22 per group). D Schematic of the different subregions (PL and IL) and layers (deep and superficial) of the mPFC analyzed by immunofluorescence staining. E Representative images of immunofluorescence staining of PLXNA1 (green). Nuclei are labeled by DAPI (blue). Left panel: no primary antibody as the control staining, imaged by 20× objective; middle panel: PLXNA1 staining, imaged by 20× objective (scale bar: 125 µm); right panel: PLXNA1 staining under high magnification, imaged by 40× objective (scale bar: 40 µm). F Representative images of immunofluorescence staining of PLXNA1 in different layers of the PL and IL subregions of mPFC. Images from naïve and 1 h post-CFC groups were presented (scale bar: 40 µm). Fold changes in the fluorescence intensity of PLXNA1 staining in the overall PL and IL subregions of the mPFC (G), as well as in the superficial and deep layers of the PL (H) and IL (I) subregions (n = 8 per group). All data are presented as mean ± s.e.m. and analyzed by one-way ANOVA followed by Dunnett’s post hoc test (B and C) or two-way ANOVA followed by Sidak’s post hoc test (G–I). Tr training, Sac sacrifice, CFC contextual fear conditioning, mPFC medial prefrontal cortex, PL prelimbic cortex, IL infralimbic cortex.
We next examined the protein levels of PLXNA1 in the mPFC at 1 h after CFC training versus naïve controls using immunofluorescence staining to identify the spatial distribution of PLXNA1 protein (Fig. 1D, E). Analyses of the intensity of PLXNA1 immunofluorescence staining revealed that the level of PLXNA1 protein decreased in both the PL and IL subregions of mPFC at 1 h after CFC training, with relatively larger reduction in the deep layers than in the superficial layers (Fig. 1F–I).
Collectively, these findings revealed that the expression of both Plxna1 mRNA and protein in mice mPFC decreased as early as 1 h after CFC training.
CFC training decreases PLXNA1 expression in the IL inhibitory neurons
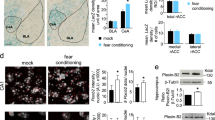
Next, we wondered in which neuronal subtype the reduction of mPFC PLXNA1 expression occurred after CFC training. Towards that end, we performed immunofluorescence co-staining of PLXNA1 with the excitatory neuronal marker calcium/calmodulin-dependent protein kinase II (CaMKII) or the inhibitory neuronal marker GAD67. As the mPFC deep layers showed a more robust decrease in the PLXNA1 level after training than the superficial layers (Fig. 1H, I), for this analysis, we focused on the mPFC deep layers (Fig. 2A).
A Schematic of the deep layers of the mPFC analyzed in the immunofluorescence staining study. B Representative images of immunofluorescence staining of PLXNA1 (left panel, green), GAD67 (middle panel, red), and PLXNA1 staining in GAD67-positive areas (right panel, green) in the IL deep layers, comparing naïve and 1 h post-CFC groups (scale bar: 30 µm). C Fold changes in the fluorescence intensity of PLXNA1 staining in GAD67-positive areas at 1 h post-CFC. D Fold changes in fluorescence intensity of GAD67 staining (n = 7–8 per group). E Representative images of immunofluorescence staining of PLXNA1 (left panel, green), CaMKII (middle panel, red), and PLXNA1 staining in CaMKII-positive areas (right panel, green) in IL deep layers, comparing naïve and 1 h post-CFC (scale bar: 30 µm). F Fold changes in the fluorescence intensity of PLXNA1 staining in CaMKII-positive areas of naïve and 1 h post-CFC groups. G Fold changes in fluorescence intensity of CaMKII staining of naïve and 1 h post-CFC groups (n = 7–8 per group). All data are presented as mean ± s.e.m. and analyzed by two-way ANOVA followed by Sidak’s post hoc test. CFC contextual fear conditioning, PL prelimbic cortex, IL infralimbic cortex.
Interestingly, we found that CFC training significantly reduced PLXNA1 expression in the GAD67-labeled inhibitory neurons of the IL deep layers (Fig. 2B, C). However, there was no significant change in the PLXNA1 level in the mPFC excitatory neurons labeled by CaMKII (Fig. 2E, F). The overall levels of CAMKII and GAD67 did not differ between naïve versus CFC (Fig. 2D, G). To confirm that PLXNA1 could indeed be expressed in the inhibitory neurons of IL deep layers, we performed RNAscope in situ hybridization of Plxna1 mRNA. As shown in the Supplementary Fig. 2, Plxna1 mRNA could be found localized in Gad67 mRNA-labeled inhibitory neurons in the IL deep layers.
Therefore, these studies revealed an unexpected finding that CFC training reduced PLXNA1 expression in the IL inhibitory neurons.
PLXNA1 in the IL inhibitory neurons regulates memory specificity, but not memory strength
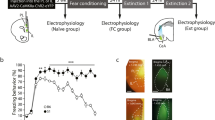
The ectodomain of PLXNA1 contains an autoinhibitory domain. Upon ligand binding, this domain is spatially segregated from the remainder of PLXNA1 to allow signaling transduction to remodel axonal cytoskeletons [36, 43]. Previous studies have demonstrated that Plxna1 lacking the ectodomain (PLXNA1ΔEct) is constitutively active [36, 43]. To investigate the role of PLXNA1 in contextual fear memory, we generated an AAV expressing HA-tagged PLXNA1ΔEct under the mDlx promoter to achieve cell-type specific expression in the inhibitory neurons [37]. AAV8-mDlx-HA-Plxna1ΔEct was bilaterally injected into the IL. The control groups were injected with AAV8 expressing either the HA-tag only or EGFP under the mDlx promoter (Fig. 3A). Group histology analysis showed that HA-expressing neurons were mostly distributed in the IL region (Supplementary Fig. 3A). Since no difference in the behavioral results was observed between these two types of controls, the data were combined for analysis. As shown in Supplementary Fig. 3B, AAV8-mDlx-EGFP expressed EGFP mainly in the GAD67-labeled inhibitory neurons, but not in CaMKII-labeled excitatory neurons. The animals were trained with CFC and 24 h later, tested for long-term memory strength in the training context, or for memory specificity in a modified context. The order of the two tests was counter-balanced (Fig. 3B). To our surprise, PLXNA1ΔEct overexpression in the IL inhibitory neurons (Supplementary Fig. 3C) did not cause any significant change in the freezing level during training or when the animals were tested in the training context during the long-term fear retrieval test, but resulted in enhanced freezing to the modified context, indicating an increase in fear generalization without affecting memory strength (Fig. 3C, Supplementary Fig. 4A). Fear generalization is often observed in patients with generalized anxiety disorder [44, 45]. We therefore examined anxiety-like behaviors of these animals using an open field test. Compared to the control animals, the mice with PLXNA1ΔEct overexpression in the IL inhibitory neurons tended to travel less in the center zone of the open field without changing the overall locomotion, indicating that the PLXNA1ΔEct group exhibited a trend towards enhanced anxiety-like behaviors after CFC (Fig. 3D–F). We did not find a significant correlation between the percentage of distance traveled in the center zone of the open field and the percentage of GAD67+ inhibitory neurons expressing PLXNA1ΔEct (R2 = 0.14, p = 0.28).
A Schematic of AAV infusion into the IL (left) and a representative image of the distribution of AAV-infected cells in the IL (labeled by HA staining) (scale bar: 400 µm). B Timeline of the CFC training (Tr) in context A (ctx A), fear memory strength test (F test) in context A, memory generalization test (G test) in context B (ctx B), and the open field test (OFT). The “F test” and “G test” were counterbalanced. C, G Quantification of the percentage of time spent in freezing during the F test and G test, comparing animals injected with the control AAV (Ctrl, AAV8-mDlx-HA or AAV8-mDlx-EGFP) and AAV8-mDlx-HA-PLXNA1ΔEct (C, n = 10–13 per group) or AAV8-mDlx-HA-PLXNA1cyto (G, n = 6–9 per group) into the IL. D, H Representative travel traces of the animals in the open field test. Inner squares bordered with dashed lines indicate the center zones. Quantification of total travel distance (E, I) and the distance in the center zone as a percentage of the total distance (F, J) in the open field, comparing the control and PLXNA1ΔEct groups (E, F, n = 11–14 per group), or the control and PLXNA1cyto groups (I, J, n = 10–11 per group). All data are presented as mean ± s.e.m. and analyzed by unpaired two-tailed student’s t test or two-way ANOVA followed by Sidak’s post hoc test.
We further examined memory performance and anxiety-like behaviors in mice injected with AAV that expressed only the cytoplasmic domain of PLXNA1 protein (PLXNA1cyto) under the mDlx promoter (AAV8-mDlx-HA-Plxna1cyto). Since PLXNA1cyto lacked its membrane anchoring domain, we reasoned that it could lose the spatial property required for interacting with other signaling partners at the plasma membrane and therefore produce a dominant-negative effect for PLXNA1-mediated signaling. The animals with PLXNA1cyto overexpression in the IL inhibitory neurons (Supplementary Fig. 3A, D) again did not affect the memory strength as demonstrated by their unaltered freezing levels when tested in the same context at 24 h after CFC training or during training, but showed significantly reduced freezing to the modified context (Fig. 3G, Supplementary Fig. 4B). The PLXNA1cyto group also exhibited a trend towards decreased anxiety-like behaviors indicated by their increased overall locomotion in the open field as well as the distance traveled in the center zone (Fig. 3H–J). There was no significant correlation between the percentage of distance traveled in the center zone of the open field and the percentage of GAD67+ inhibitory neurons expressing PLXNA1cyto (R2 = 0.14, p = 0.27). Of note, as freezing to the same context used in CFC training during fear retrieval test was not affected, the decreased freezing level to the modified context by PLXNA1cyto overexpression observed in the memory generalization test could not be simply explained by increased locomotion (Fig. 3G, I).
Taken together, these findings suggest that enhanced PLXNA1 function in the IL inhibitory neurons facilitated contextual fear memory generalization and tended to enhance anxiety, whereas disruption of PLXNA1 function improved contextual fear memory specificity as well as tended to reduce anxiety.
PLXNA1 in the IL inhibitory neurons suppresses c-FOS expression in the IL and its downstream brain regions
As manipulation of PLXNA1 function in the IL inhibitory neurons affected contextual fear memory specificity, we wondered whether such behavioral changes were accompanied by changes in the neuronal activity of IL and its downstream regions. c-FOS is an immediate-early gene product whose expression is tightly regulated by neuronal activity. Thus, changes in the density of c-FOS-positive cells are often used as indicators of changes in brain region activities [41, 42]. We found that PLXNA1ΔEct overexpression significantly decreased c-FOS expression, whereas PLXNA1cyto overexpression enhanced c-FOS expression in the IL region (Fig. 4A–D). We further examined c-FOS expression in the downstream brain regions of IL (Fig. 4E) [46]. Our data showed that PLXNA1ΔEct overexpression in the IL inhibitory neurons reduced c-FOS expression in the anterior cingulate cortex (ACC), caudate putamen (CPu) and dorsal raphe (DR) (Fig. 4F), whereas PLXNA1cyto overexpression increased c-FOS expression in the PL, ACC, CPu, agranular insular cortex (AI), claustrum (CLA), diagonal band (DB), nucleus accumbens (NAc), bed nucleus of the stria terminalis (BNST), paraventricular thalamic nucelus (PVT), dorsomedial hypothalamic nucleus (DMH), posterior hypothalamic area (PH) and entorhinal cortex (EC) (Fig. 4G).
A Schematic of AAV infusion into the IL. B Representative images of immunofluorescence staining of c-FOS in the IL deep layers (scale bar: 250 µm). C, D Fold changes in the density of c-FOS positive cells in the IL, comparing the control (Ctrl) and PLXNA1ΔEct groups (C, n = 8 per group), or the control and PLXNA1cyto groups (D, n = 7–9 per group). E Atlas of the brain regions analyzed (highlighted in orange) in the c-FOS immunofluorescence staining study. Fold changes in the density of c-FOS positive cells in the downstream brain regions of IL, comparing the Ctrl and PLXNA1ΔEct groups (F, n = 7–8 per group), or the control and PLXNA1cyto groups (G, n = 6–8 per group). All data are presented as mean ± s.e.m. and analyzed by unpaired two-tail student’s t test.
These data therefore suggest that PLXNA1 in the IL inhibitory neurons coordinately regulated the activity of IL and its downstream projecting regions.
PLXNA1 in the inhibitory neurons increases the number of GAD67+ puncta in the IL
PLXNA1 has been implicated in regulating axonogenesis during developmental stages [31]. However, its functional role in the adult brain remains unclear. Our findings now revealed that PLXNA1 in the IL inhibitory neurons suppressed the overall activity of IL. Because GAD67 has been reported to localize in the axonal processes and regulate local GABA synthesis [47], we wondered whether PLXNA1 could regulate the expression or distribution of GAD67 in the IL inhibitory neurons. We found that the number of GAD67+ puncta surrounding the targeted neurons was significantly decreased in the IL deep layers after CFC training, though the overall intensity of GAD67 staining was not changed (Fig. 2D, Supplementary Fig. 5). PLXNA1ΔEct and PLXNA1cyto overexpression in the IL inhibitory neurons significantly increased and decreased, respectively, the number of GAD67+ puncta surrounding the targeted neurons (Fig. 5A, B, D, E). However, the overall intensity of GAD67 staining was not significantly different between the groups (Fig. 5C, F). These results indicated that PLXNA1 regulated presynaptic distribution of GAD67 protein in the IL inhibitory neurons.
A, D Representative images of immunofluorescence staining of GAD67 + (red) and DAPI (blue) in the IL inhibitory neurons (scale bar: 20 µm). B, E Fold changes in the density of GAD67-positive puncta in the IL deep layers, comparing the control (Ctrl) and PLXNA1ΔEct groups (B, n = 7 per group), or the control and PLXNA1cyto groups (E, n = 6–8 per group). C, F Fold changes in the overall fluorescence intensity of GAD67 staining in the IL deep layers, comparing the Ctrl and PLXNA1ΔEct groups (C, n = 8 per group), or the control and PLXNA1cyto groups (F, n = 7–8 per group). All data are presented as mean ± s.e.m. and analyzed by unpaired two-tailed student’s t test.
Discussion
In the present study, we identified that both the mRNA and protein levels of Plxna1 decreased in the mPFC after CFC. Memory consolidation requires changes in transcription and translation [10, 48]. Previous studies have explored extensively how changes in gene expression in the hippocampus and the amygdala contribute to the formation of long-term synaptic plasticity and memory [8,9,10,11]. Growing evidences have suggested the mPFC as a cortical region critically implicated in memory processing [15, 49]. Inhibition of gene expression in the mPFC immediately after learning impaired long-term memory formation [23, 24]. Inspired by these findings, a few recent studies applied transcriptomic analysis of the mPFC to uncover the differential expression profile of learning-associated genes [28,29,30]. Unlike hippocampus where most DEGs are upregulated, a substantial proportion of the DEGs in the mPFC are downregulated by training [28, 30]. In addition, the identified DEGs as well as the enriched gene ontology and pathways in the mPFC were largely distinct from those identified in the hippocampus, suggesting that mPFC recruits a unique set of molecular architecture to subserve memory formation [30]. A gene set that regulates axon development was identified as one of the top biological processes enriched in the mPFC early after learning [28, 30]. Plxna1 is one of the downregulated genes in the mPFC that is also engaged in axonogenesis during development [30, 31]. Here, using qPCR and immunofluorescence staining, we confirmed that Plxna1 expression decreased in the mPFC after learning. Interestingly, such decrease started around 1 h and persisted to at least 3 h post-training, and occurred in the inhibitory but not excitatory neurons in the deep layers of the IL subregion of mPFC. More importantly, our findings indicated that PLXNA1 played an essential role in memory processing in the adult mPFC. These findings support the notion that similar to excitatory neurons, inhibitory neurons can also undergo experience-dependent transcriptional and translational changes to regulate memory processing [50, 51].
In addition, nonsense- or microRNA-mediated RNA degradation, as well as degradation of proteins through proteasome and lysosome pathways have been reported to occur after learning, which play important roles in rapid turnover of immediate-early gene products (such as Arc and c-Fos), and spine remodeling [52,53,54,55,56]. The decrease in Plxna1 expression in the IL inhibitory neurons after learning is likely due to a shift in the balance between synthesis and degradation induced by changes in the activity of IL inhibitory neurons, which awaits further examination in future studies.
Previous studies of learning-associated mechanisms in the hippocampus and amygdala have proposed that there are two groups of molecules engaged, memory enhancers (e.g., CREB and C/EBP) and brakes (e.g., protein phosphatase-1, calcineurin and elongation initiation factor-2), whose expression or activity are tightly regulated during memory consolidation and work in concert to maintain memory strength [10, 57, 58]. In the current study, we found surprisingly that the reduction of PLXNA1 in the IL inhibitory neurons after learning was required for maintaining memory specificity but not memory strength. These findings have advanced our understanding of the molecular mechanisms of learning-induced changes of inhibitory neuron, and suggest distinct mechanisms that regulate memory strength versus specificity during the early phase of memory consolidation.
Our findings that PLXNA1ΔEct expression in the inhibitory neurons decreased IL activity and enhanced fear generalization are consistent with previous reports that pre-training lesion or inactivation of the mPFC or the IL subregion of mPFC during fear conditioning resulted in generalized fear without affecting memory strength [26, 27, 59]. However, accumulating evidences also suggest that memory consolidation is not simply controlled by switching on or off any brain region, but rather, different neuronal subtypes that contribute to the regulation of micro-circuit and macro-circuit activities. For example, Bero et al. [28] reported that optogenetic silencing of mPFC excitatory neurons during CFC impaired both recent and remote long-term memory. Interestingly, optogenetic silencing of somatostatin-positive inhibitory neurons within mPFC during fear conditioning also impaired formation of long-term memory [60], suggesting that both excitatory and somatostatin-positive inhibitory neurons of mPFC can regulate memory strength. mPFC inhibitory neurons have also been implicated in regulating memory specificity. Yan et al. [61] reported that in a fear discrimination training paradigm, safety signal increased the activity of parvalbumin-positive inhibitory neurons to suppress fear generalization. Our data now provide additional evidences supporting the importance of IL inhibitory neurons in regulating memory specificity and suggest that the reduction of PLXNA1 after learning-induced redistribution of GAD67 in the IL inhibitory neurons. Collectively, previous and our studies reveal the complex nature of different subtypes of mPFC neurons to memory formation, and our findings emphasize the importance to examine cell-type specific molecular changes and their roles in regulating memory strength and specificity.
Previous studies have revealed that some of the molecular mechanisms that regulate neural development can be reused in the adult brain to regulate neuroplasticity [62]. PLXNA1 is a receptor for multiple semaphorins, including SEMA3A and SEMA6D [31,32,33,34,35]. Activation of PLXNA1 induces axon repulsion and collapse of axonal growth cone during early development [63, 64]. PLXNA1 forms complex with neuropilin-1 or neuronal cell adhesion molecule on the plasma membrane [33, 36, 63, 64]. The cytoplasmic domain of PLXNA1 has tyrosine kinase activity and Rho-GAP activity, which initiate signaling pathways to trigger cytoskeleton rearrangement and membrane endocytosis [65,66,67]. Considered as an important player in axonogenesis during development, the function of PLXNA1 in the adult brain however was unclear. Here, we used both gain-of-function and loss-of-function mutants of PLXNA1 to show that PLXNA1 regulated the number of GAD67 puncta in the IL inhibitory neurons. As one of the major GABA synthetases, the clustering of GAD67 at axon terminals regulates local GABA synthesis at the synapses [47]. Therefore, our data suggest that PLXNA1 may regulate the availability of GABA at the synapses, which in turn changes the excitation / inhibition balance in the IL. In line with this notion, manipulation of PLXNA1 in the inhibitory neurons resulted in bidirectionally changes in c-FOS expression in the IL.
Furthermore, growing evidences suggest that memory is regulated by a network of distributed yet interconnected brain regions [68, 69]. Here, we found that changes in IL activity by PLXNA1 led to changes in multiple downstream brain regions. We noticed that the brain regions affected by PLXNA1ΔEct overexpression in the IL inhibitory neurons were fewer than those affected by PLXNA1cyto expression. A likely explanation is that the c-FOS analyses were conducted on brain samples collected 2 days after behavioral tests, when many brain regions expressed minimal c-FOS. Thus, PLXNA1ΔEct overexpression in the IL inhibitory neurons failed to further decrease c-FOS expression in these regions.
Our results showed that after CFC, PLXNA1 expression significantly decreased in the IL deep layers, but only marginally decreased in the IL superficial layers. Studies have found that distinct layers of IL have different connectivity. While excitatory projection neurons in the superficial layers project mostly to other cortical regions, those in the deep layers target mainly subcortical regions [70]. Activities of these projection neurons are modulated by inhibitory neurons scattered in multiple layers. Interestingly, studies have revealed that the inhibitory neurons preferentially regulate the IL projection neurons that target subcortical regions than those targeting other cortical regions [70]. Therefore, the preferential downregulation of PLXNA1 expression in the deep layers of IL by CFC might lead to alteration in the inhibitory control of subcortical-targeting projection neurons, which in turn regulate memory generalization. It is worth mentioning that the deep layers of IL, as compared to the superficial layers, receive more pronounced inputs from the basolateral amygdala (BLA), which conveys aversive information, and from the ventral hippocampus (vHC) that provides contextual information [71, 72]. It would be of interest to examine whether the converging inputs from BLA and vHC during CFC training contribute to the downregulation of PLXNA1 in IL deep layers.
In addition to IL, PLXNA1 expression also significantly decreased in the PL deep layers, which showed a trend to decreased expression in both excitatory and inhibitory neurons. It is possible that the significant reduction of PLXNA1 in this region was due to the combined partial reduction of PLXNA1 in both excitatory and inhibitory neurons. In addition, to analyze overall PLXNA1 level, we used images taken by a 20× objective, which covered a relatively large field and thus minimized variability due to sub-area differences in PLXNA1 distribution. However, in order to recognize PLXNA1 staining in different neuronal subtypes, we used images taken by a 63× objective. Although data from multiple images per animal were averaged as a single data point, it still covered a relatively smaller field of view, which might contribute to the inconsistency in the data. In the classical model, the PL subregion of mPFC promotes fear memory expression, whereas the IL subregion facilitates fear extinction, though arguments against a clear functional division between PL and IL exist [17, 49, 73,74,75,76]. It remains to be determined how PLXNA1 in the PL affects memory strength or specificity.
Overgeneralized fear is a core symptom for anxiety disorders and PTSD [3,4,5,6,7]. Functional brain imaging studies have found increased ventral mPFC activity when human subjects received visual images deviating from the previous presented ones, indicating a role of ventral mPFC in signal discrimination [77, 78]. The ventral mPFC activity was found to be reduced in generalized anxiety disorder and PTSD patients, and such reduction was associated with inability to constrain fear and anxiety in these patients [77, 79,80,81]. It will be of great interest to examine whether abnormal changes of the PLXNA1 expression and function in the inhibitory neurons of ventral mPFC contribute to the pathological development of generalized anxiety disorder and PTSD, and to evaluate the therapeutic potential of targeting PLXNA1 for disease intervention. The lack of investigation of female mice was a limitation of the current study. Female mice were excluded from the original design of this study due to the concern of the fluctuation in the estrogen levels during the estrous cycle of female mice that may affect fear generalization as reported in the previous studies [82,83,84] to generate greater individual variability. Since women are more likely to develop PTSD and anxiety disorders than men [85, 86], it will be of importance to examine in the future work whether similar mechanisms affect memory generalization in female mice. However, it is worth noting that men respond differently to psychotherapy than women [87]. Therefore, it would still be of value to identify a potential target for limiting fear generalization using male mice.
In conclusion, our study has identified the novel role of PLXNA1 in the IL inhibitory neurons to modulate memory specificity. Our results showed that contextual fear conditioning reduced PLXNA1 expression in the IL inhibitory, but not excitatory neurons, which in turn was important for maintaining memory specificity and suppressing anxiety, without affecting memory strength. Furthermore, PLXNA1 functioned in the IL inhibitory neurons to regulate the number of GAD67 puncta, which in turn modulated the activity of IL and its downstream brain regions. These findings provide novel insights on learning-induced molecular changes that modify mPFC inhibitory neurons and neural network in regulating memory specificity, and suggest novel targets for therapeutic interventions of generalized anxiety disorder and PTSD.
References
Herry C, Johansen JP. Encoding of fear learning and memory in distributed neuronal circuits. Nat Neurosci. 2014;17:1644–54.
Asok A, Leroy F, Rayman JB, Kandel ER. Molecular mechanisms of the memory trace. Trends Neurosci. 2019;42:14–22.
Steiner M, Allgulander C, Ravindran A, Kosar H, Burt T, Austin C. Gender differences in clinical presentation and response to sertraline treatment of generalized anxiety disorder. Hum Psychopharmacol. 2005;20:3–13.
Mahan AL, Ressler KJ. Fear conditioning, synaptic plasticity and the amygdala: implications for posttraumatic stress disorder. Trends Neurosci. 2012;35:24–35.
Gotlib IH, Joormann J. Cognition and depression: current status and future directions. Annu Rev Clin Psychol. 2010;6:285–312.
Yehuda R, Hoge CW, McFarlane AC, Vermetten E, Lanius RA, Nievergelt CM, et al. Post-traumatic stress disorder. Nat Rev Dis Prim. 2015;1:15057.
Shin LM, Liberzon I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology. 2010;35:169–91.
Dudai Y, Karni A, Born J. The consolidation and transformation of memory. Neuron. 2015;88:20–32.
Goelet P, Castellucci VF, Schacher S, Kandel ER. The long and the short of long-term memory-a molecular framework. Nature. 1986;322:419–22.
Costa-Mattioli M, Sossin WS, Klann E, Sonenberg N. Translational control of long-lasting synaptic plasticity and memory. Neuron. 2009;61:10–26.
Alberini CM. Transcription factors in long-term memory and synaptic plasticity. Physiological Rev. 2009;89:121–45.
Dudai Y. The neurobiology of consolidations, or, how stable is the engram? Annu Rev Psychol. 2004;55:51–86.
Uylings HB, Groenewegen HJ, Kolb B. Do rats have a prefrontal cortex? Behav Brain Res. 2003;146:3–17.
Szczepanski SM, Knight RT. Insights into human behavior from lesions to the prefrontal cortex. Neuron. 2014;83:1002–18.
Euston DR, Gruber AJ, McNaughton BL. The role of medial prefrontal cortex in memory and decision making. Neuron. 2012;76:1057–70.
Gilmartin MR, Balderston NL, Helmstetter FJ. Prefrontal cortical regulation of fear learning. Trends Neurosci. 2014;37:455–64.
Giustino TF, Maren S. The role of the medial prefrontal cortex in the conditioning and extinction of fear. Front Behav Neurosci. 2015;9:298.
Hoover WB, Vertes RP. Anatomical analysis of afferent projections to the medial prefrontal cortex in the rat. Brain Struct Funct. 2007;212:149–79.
Ye X, Kapeller-Libermann D, Travaglia A, Inda MC, Alberini CM. Direct dorsal hippocampal-prelimbic cortex connections strengthen fear memories. Nat Neurosci. 2017;20:52–61.
Barker GR, Banks PJ, Scott H, Ralph GS, Mitrophanous KA, Wong LF, et al. Separate elements of episodic memory subserved by distinct hippocampal-prefrontal connections. Nat Neurosci. 2017;20:242–50.
Eichenbaum H. Prefrontal-hippocampal interactions in episodic memory. Nat Rev Neurosci. 2017;18:547–58.
Heroux NA, Osborne BF, Miller LA, Kawan M, Buban KN, Rosen JB, et al. Differential expression of the immediate early genes c-Fos, Arc, Egr-1, and Npas4 during long-term memory formation in the context preexposure facilitation effect (CPFE). Neurobiol Learn Mem. 2018;147:128–38.
Zhang Y, Fukushima H, Kida S. Induction and requirement of gene expression in the anterior cingulate cortex and medial prefrontal cortex for the consolidation of inhibitory avoidance memory. Mol Brain. 2011;4:4.
Rizzo V, Touzani K, Raveendra BL, Swarnkar S, Lora J, Kadakkuzha BM, et al. Encoding of contextual fear memory requires de novo proteins in the prelimbic cortex. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017;2:158–69.
Spalding KN. The role of the medial prefrontal cortex in the generalization of conditioned fear. Neuropsychology. 2018;32:1–17.
Xu W, Sudhof TC. A neural circuit for memory specificity and generalization. Science. 2013;339:1290–5.
Bayer H, Bertoglio LJ. Infralimbic cortex controls fear memory generalization and susceptibility to extinction during consolidation. Sci Rep. 2020;10:15827.
Bero AW, Meng J, Cho S, Shen AH, Canter RG, Ericsson M, et al. Early remodeling of the neocortex upon episodic memory encoding. Proc Natl Acad Sci USA. 2014;111:11852–7.
Chen MB, Jiang X, Quake SR, Sudhof TC. Persistent transcriptional programmes are associated with remote memory. Nature. 2020;587:437–42.
Katzman A, Khodadadi-Jamayran A, Kapeller-Libermann D, Ye X, Tsirigos A, Heguy A, et al. Distinct transcriptomic profiles in the dorsal hippocampus and prelimbic cortex are transiently regulated following episodic learning. J Neurosci. 2021;41:2601–14.
Waimey KE, Cheng HJ. Axon pruning and synaptic development: how are they per-plexin? Neuroscientist. 2006;12:398–409.
Takahashi T, Fournier A, Nakamura F, Wang L-H, Murakami Y, Kalb RG, et al. Plexin-neuropilin-1 complexes form functional semaphorin-3A receptors. Cell. 1999;99:59–69.
Kuwajima T, Yoshida Y, Takegahara N, Petros TJ, Kumanogoh A, Jessell TM, et al. Optic chiasm presentation of Semaphorin6D in the context of Plexin-A1 and Nr-CAM promotes retinal axon midline crossing. Neuron. 2012;74:676–90.
Peng Y, Song L, Li D, Kesterson R, Wang J, Wang L, et al. Sema6D acts downstream of bone morphogenetic protein signalling to promote atrioventricular cushion development in mice. Cardiovasc. Res. 2016;112:532–42.
Toyofuku T, Zhang H, Kumanogoh A, Takegahara N, Suto F, Kamei J, et al. Dual roles of Sema6D in cardiac morphogenesis through region-specific association of its receptor, Plexin-A1, with off-track and vascular endothelial growth factor receptor type 2. Genes Dev. 2004;18:435–47.
Takahashi T, Strittmatter SM. Plexina1 autoinhibition by the plexin sema domain. Neuron. 2001;29:429–39.
Dimidschstein J, Chen Q, Tremblay R, Rogers SL, Saldi GA, Guo L, et al. A viral strategy for targeting and manipulating interneurons across vertebrate species. Nat Neurosci. 2016;19:1743–49.
Lin WJ, Jiang C, Sadahiro M, Bozdagi O, Vulchanova L, Alberini CM, et al. VGF and Its C-Terminal Peptide TLQP-62 Regulate Memory Formation in Hippocampus via a BDNF-TrkB-Dependent Mechanism. J Neurosci. 2015;35:10343–56.
Lin WJ, Zhao Y, Li Z, Zheng S, Zou JL, Warren NA, et al. An increase in VGF expression through a rapid, transcription-independent, autofeedback mechanism improves cognitive function. Transl. Psychiatry. 2021;11:383.
Jiang C, Lin WJ, Labonte B, Tamminga CA, Turecki G, Nestler EJ, et al. VGF and its C-terminal peptide TLQP-62 in ventromedial prefrontal cortex regulate depression-related behaviors and the response to ketamine. Neuropsychopharmacology. 2019;44:971–81.
Gallo FT, Katche C, Morici JF, Medina JH, Weisstaub NV. Immediate early genes, memory and psychiatric disorders: focus on c-Fos, Egr1 and Arc. Front Behav Neurosci. 2018;12:79.
Sheng M, McFadden G, Greenberg ME. Membrane depolarization and calcium induce c-fos transcription via phosphorylation of transcription factor CREB. Neuron. 1990;4:571–82.
Togashi H, Schmidt EF, Strittmatter SM. RanBPM contributes to Semaphorin3A signaling through plexin-A receptors. J Neurosci. 2006;26:4961–9.
Dymond S, Dunsmoor JE, Vervliet B, Roche B, Hermans D. Fear generalization in humans: systematic review and implications for anxiety disorder research. Behav Ther. 2015;46:561–82.
Jasnow AM, Lynch JF 3rd, Gilman TL, Riccio DC. Perspectives on fear generalization and its implications for emotional disorders. J Neurosci Res. 2017;95:821–35.
Vertes RP. Differential projections of the infralimbic and prelimbic cortex in the rat. Synapse. 2004;51:32–58.
Kanaani J, Kolibachuk J, Martinez H, Baekkeskov S. Two distinct mechanisms target GAD67 to vesicular pathways and presynaptic clusters. J Cell Biol. 2010;190:911–25.
Alberini CM, Kandel ER. The regulation of transcription in memory consolidation. Cold Spring Harb Perspect Biol. 2014;7:a021741.
Rozeske RR, Valerio S, Chaudun F, Herry C. Prefrontal neuronal circuits of contextual fear conditioning. Genes Brain Behav. 2015;14:22–36.
Sharma V, Sood R, Khlaifia A, Eslamizade MJ, Hung TY, Lou D, et al. eIF2α controls memory consolidation via excitatory and somatostatin neurons. Nature. 2020;586:412–16.
Shrestha P, Shan Z, Mamcarz M, Ruiz KSA, Zerihoun AT, Juan CY, et al. Amygdala inhibitory neurons as loci for translation in emotional memories. Nature. 2020;586:407–11.
Lee YS, Lee JA, Kaang BK. Regulation of mRNA stability by ARE-binding proteins in synaptic plasticity and memory. Neurobiol Learn Mem. 2015;124:28–33.
Jaffrey SR, Wilkinson MF. Nonsense-mediated RNA decay in the brain: emerging modulator of neural development and disease. Nat Rev Neurosci. 2018;19:715–28.
Kiebler MA, Bassell GJ. Neuronal RNA granules: movers and makers. Neuron. 2006;51:685–90.
Jarome TJ, Helmstetter FJ. The ubiquitin-proteasome system as a critical regulator of synaptic plasticity and long-term memory formation. Neurobiol Learn Mem. 2013;105:107–16.
Fleming A, Rubinsztein DC. Autophagy in neuronal development and plasticity. Trends Neurosci. 2020;43:767–79.
Stern SA, Alberini CM. Mechanisms of memory enhancement. Wiley Interdiscip Rev Syst Biol Med. 2013;5:37–53.
Mansuy IM, Shenolikar S. Protein serine/threonine phosphatases in neuronal plasticity and disorders of learning and memory. Trends Neurosci. 2006;29:679–86.
Antoniadis EA, McDonald RJ. Fornix, medial prefrontal cortex, nucleus accumbens, and mediodorsal thalamic nucleus: roles in a fear-based context discrimination task. Neurobiol Learn Mem. 2006;85:71–85.
Cummings KA, Clem RL. Prefrontal somatostatin interneurons encode fear memory. Nat Neurosci. 2020;23:61–74.
Yan R, Wang T, Zhou Q. Elevated dopamine signaling from ventral tegmental area to prefrontal cortical parvalbumin neurons drives conditioned inhibition. Proc Natl Acad Sci USA. 2019;116:13077–86.
Carew TJ, Menzel R, Shatz CJ. Mechanistic relationships between development and learning: report of the Dahlem Workshop on Mechanistic Relationships between Development and Learning, Berlin, January 19-25, 1997. New York: John Wiley & Sons; 1998.
Rohm B, Ottemeyer A, Lohrum M, Püschel AW. Plexin/neuropilin complexes mediate repulsion by the axonal guidance signal semaphorin 3A. Mechanisms Dev. 2000;93:95–104.
Wu KY, He M, Hou QQ, Sheng AL, Yuan L, Liu F, et al. Semaphorin 3A activates the guanosine triphosphatase Rab5 to promote growth cone collapse and organize callosal axon projections. Sci Signal. 2014;7:ra81.
Wang H, Hota PK, Tong Y, Li B, Shen L, Nedyalkova L, et al. Structural basis of Rnd1 binding to plexin Rho GTPase binding domains (RBDs). J Biol Chem. 2011;286:26093–106.
Rohm B, Rahim B, Kleiber B, Hovatta I, Puschel AW. The semaphorin 3A receptor may directly regulate the activity of small GTPases. FEBS Lett. 2000;486:68–72.
Turner LJ, Nicholls S, Hall A. The activity of the plexin-A1 receptor is regulated by Rac. J Biol Chem. 2004;279:33199–205.
Nadel L, Hardt O. Update on memory systems and processes. Neuropsychopharmacology. 2011;36:251–73.
Vetere G, Kenney JW, Tran LM, Xia F, Steadman PE, Parkinson J, et al. Chemogenetic Interrogation of a Brain-wide Fear Memory Network in Mice. Neuron 2017;94:363–74 e4.
Anastasiades PG, Carter AG. Circuit organization of the rodent medial prefrontal cortex. Trends Neurosci. 2021;44:550–63.
Cheriyan J, Kaushik MK, Ferreira AN, Sheets PL. Specific targeting of the basolateral amygdala to projectionally defined pyramidal neurons in prelimbic and infralimbic cortex. eNeuro. 2016;3:e0002–16.2016 1-13.
Liu X, Carter AG. Ventral hippocampal inputs preferentially drive corticocortical neurons in the infralimbic prefrontal cortex. J Neurosci. 2018;38:7351–63.
Peters J, Kalivas PW, Quirk GJ. Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem. 2009;16:279–88.
Sotres-Bayon F, Quirk GJ. Prefrontal control of fear: more than just extinction. Curr Opin Neurobiol. 2010;20:231–5.
Riga D, Matos MR, Glas A, Smit AB, Spijker S, Van den Oever MC. Optogenetic dissection of medial prefrontal cortex circuitry. Front Syst Neurosci. 2014;8:230.
Courtin J, Bienvenu TC, Einarsson EO, Herry C. Medial prefrontal cortex neuronal circuits in fear behavior. Neuroscience. 2013;240:219–42.
Greenberg T, Carlson JM, Cha J, Hajcak G, Mujica-Parodi LR. Ventromedial prefrontal cortex reactivity is altered in generalized anxiety disorder during fear generalization. Depress Anxiety. 2013;30:242–50.
Lissek S. Toward an account of clinical anxiety predicated on basic, neurally mapped mechanisms of Pavlovian fear-learning: the case for conditioned overgeneralization. Depress Anxiety. 2012;29:257–63.
Kim MJ, Loucks RA, Palmer AL, Brown AC, Solomon KM, Marchante AN, et al. The structural and functional connectivity of the amygdala: from normal emotion to pathological anxiety. Behav Brain Res. 2011;223:403–10.
Pitman RK, Rasmusson AM, Koenen KC, Shin LM, Orr SP, Gilbertson MW, et al. Biological studies of post-traumatic stress disorder. Nat Rev Neurosci. 2012;13:769–87.
Rougemont-Bucking A, Linnman C, Zeffiro TA, Zeidan MA, Lebron-Milad K, Rodriguez-Romaguera J, et al. Altered processing of contextual information during fear extinction in PTSD: an fMRI study. CNS Neurosci Ther. 2011;17:227–36.
Lynch J 3rd, Cullen PK, Jasnow AM, Riccio DC. Sex differences in the generalization of fear as a function of retention intervals. Learn Mem. 2013;20:628–32.
Lynch JF 3rd, Dejanovic D, Winiecki P, Mulvany J, Ortiz S, Riccio DC, et al. Activation of ERbeta modulates fear generalization through an effect on memory retrieval. Horm Behav. 2014;66:421–9.
Lynch JF 3rd, Winiecki P, Vanderhoof T, Riccio DC, Jasnow AM. Hippocampal cytosolic estrogen receptors regulate fear generalization in females. Neurobiol Learn Mem. 2016;130:83–92.
Tolin DF, Foa EB. Sex differences in trauma and posttraumatic stress disorder: a quantitative review of 25 years of research. Psychol Bull. 2006;132:959–92.
McLean CP, Asnaani A, Litz BT, Hofmann SG. Gender differences in anxiety disorders: prevalence, course of illness, comorbidity and burden of illness. J Psychiatr Res. 2011;45:1027–35.
Wade D, Varker T, Kartal D, Hetrick S, O’Donnell M, Forbes D. Gender difference in outcomes following trauma-focused interventions for posttraumatic stress disorder: Systematic review and meta-analysis. Psychol Trauma. 2016;8:356–64.
Acknowledgements
We thank the animal facility and the core facility of the Zhongshan School of Medicine, Sun Yat-sen University for technical assistance. We thank all members of the Ye lab and Lin lab for valuable discussions and technical assistance during the execution of this projection. Supported in part by grants from National Natural Science Foundation of China (No. 81873797 to XY, No. 81972967 to WJL), Natural Science Foundation of Guangdong Province (No. 2019A1515011483 to XY, No. 2019A1515011754 to WJL), the Fundamental Research Funds for the Central Universities (No. 19ykzd40 to XY), Guangdong Project (No. 2019QN01Y202 to XY), Science and Technology Program of Guangzhou (No. 202007030001 to XY and WJL). The authors report no biomedical financial interests or potential conflicts of interest.
Author information
Authors and Affiliations
Contributions
XY and WJL designed the research project. XC, YZ, SZ, PZ and XY performed the experiments. XC, YZ, SZ, PZ, JLZ and WW analyzed the data. XY and WJL wrote the paper with input from XC, YZ, SZ, PZ and WW.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Cheng, X., Zhao, Y., Zheng, S. et al. Plexin-A1 expression in the inhibitory neurons of infralimbic cortex regulates the specificity of fear memory in male mice. Neuropsychopharmacol. 47, 1220–1230 (2022). https://doi.org/10.1038/s41386-021-01177-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-021-01177-1
- Springer Nature Switzerland AG