Abstract
This pre-registered (CRD42022322038) systematic review and meta-analysis investigated clinical and cognitive outcomes of external trigeminal nerve stimulation (eTNS) in neurological and psychiatric disorders. PubMed, OVID, Web of Science, Chinese National Knowledge Infrastructure, Wanfang, and VIP database for Chinese technical periodicals were searched (until 16/03/2022) to identify trials investigating cognitive and clinical outcomes of eTNS in neurological or psychiatric disorders. The Cochrane Risk of Bias 2.0 tool assessed randomized controlled trials (RCTs), while the Risk of Bias of Non-Randomized Studies (ROBINS-I) assessed single-arm trials. Fifty-five peer-reviewed articles based on 48 (27 RCTs; 21 single-arm) trials were included, of which 12 trials were meta-analyzed (N participants = 1048; of which ~3% ADHD, ~3% Epilepsy, ~94% Migraine; age range: 10–49 years). The meta-analyses showed that migraine pain intensity (K trials = 4, N = 485; SMD = 1.03, 95% CI[0.84–1.23]) and quality of life (K = 2, N = 304; SMD = 1.88, 95% CI[1.22–2.53]) significantly improved with eTNS combined with anti-migraine medication. Dimensional measures of depression improved with eTNS across 3 different disorders (K = 3, N = 111; SMD = 0.45, 95% CI[0.01–0.88]). eTNS was well-tolerated, with a good adverse event profile across disorders. eTNS is potentially clinically relevant in other disorders, but well-blinded, adequately powered RCTs must replicate findings and support optimal dosage guidance.
Similar content being viewed by others
Introduction
Neurological and psychiatric disorders are among the main leaders of disease burden worldwide [1, 2]. However, access to treatment remains a persistent and pervasive issue, which is mainly due to: cost of treatment and/or lack of resources to scale-up effective treatments; poor adherence due to side-effects and stigma of treatments; unclear evidence of longer-term efficacy of pharmacological treatment; and preference by many users for alternative non-drug treatments [1,2,3,4,5]. A key clinical need is therefore the development of cost-effective and practically accessible treatments with longer-term efficacy and a good tolerability profile.
One promising non-drug treatment that has a relatively good tolerability profile is external trigeminal nerve stimulation (eTNS), a non-invasive brain stimulation (NIBS) technique that applies an electric current through electrodes placed over the forehead to stimulate the supraorbital branch of the trigeminal nerve [6]. Through afferent projections from the trigeminal nerve to the nucleus of the solitary tract (NTS) and trigeminal nuclei [6,7,8], eTNS is thought to stimulate the brain stem, in particular the locus coeruleus (LC) and the reticular activation system (RAS) raphe nuclei, which are crucial for arousal and vigilance regulation [9, 10] and contribute to neurotransmitter release, particularly noradrenaline, dopamine, and serotonin [11,12,13]. Furthermore, LC and RAS project to limbic regions, which are important for emotion regulation (amygdala, limbic forebrain) [14]; and to the thalamo-cortical regions, which mediate cognitive control (prefrontal cortex; anterior cingulate their striato-thalamic connections) [15, 16], processing of sensory information (thalamus) [9, 17], and pain perception and analgesic responses (insula) [14, 18]. Therefore, eTNS has the potential to stimulate – from the bottom-up – several different fronto-cortico-thalamic and fronto-limbic pathways within the central nervous system, many of which are implicated in the symptoms, cognitive functions, and/or related behavioral features observed in several neurological and psychiatric disorders [6, 7, 19,20,21,22]. In addition, given that neurotransmitters are implicated in neurological and psychiatric disorders [23, 24], eTNS can potentially improve, for example, 1) inattention symptoms and arousal regulation in attention-deficit/hyperactivity disorder (ADHD) or seizure frequency in epilepsy via noradrenaline release [25,26,27], 2) symptoms in major depressive disorder (MDD) or migraine via enhanced noradrenaline and serotonin release [19, 28], or 3) cognitive decline and dopaminergic degeneration in Parkinson’s disease via modulating the LC-noradrenergic system [11, 29, 30]. While the evidence so far seems stronger for potential improvements of symptoms in neurological disorders, there is emerging evidence for improvement also of symptoms in psychiatric disorders. Further, eTNS is cheaper, easier to use, and more wearable compared to other transcranial NIBS techniques (e.g., transcranial magnetic or direct current stimulation, TMS or tDCS), which can be costly, require expert training, and are not readily portable [6, 31].
The evidence base for the clinical relevance of eTNS is promising, but limited. In line with the diffuse mechanism of action of bottom up stimulation of several brain networks, available systematic or narrative reviews indicate that continued use of eTNS may reduce symptoms in a variety of neurological and psychiatric disorders, such as epilepsy, migraine, major depressive, or post-traumatic stress disorders [6, 7, 32,33,34,35,36,37,38]. One meta-analysis [37] of to two randomized controlled trials (RCTs) in migraine [39, 40] found that, compared to sham, eTNS was significantly associated with improved pain ratings at 2 h (K = 2, SMD = 1.25; 95% CI [0.90–1.60] and 1 day (SMD = 0.53; 95% CI[0.14–0.92]) [37]. A second meta-analysis [34] with two RCTs in individuals with migraine [41, 42] reported significant eTNS-related improvement in the number of headache days (N = 2, SMD = −0.49; 95% CI [−0.80 to −0.19]. A third meta-analysis of up to four RCTs in migraine sufferers found significant improvement in favor of transcutaneous electrical nerve stimulation in terms of responder rates (K = 4, RR: 4.05; 95% CI[2.06 to 7.97]), monthly frequency of migraine attacks (K = 4, SMD: −0.48; 95% CI[−0.73 to − 0.23]), and painkiller intake (K = 2, SMD: −0.78; 95% CI[−1.14 to − 0.42]), but only one trial applied stimulation over the trigeminal nerve externally [41] – while the others applied stimulation percutaneously [43], to the vagus nerve [44], or to the occipital nerves [45] – which limits the interpretability of these findings. However, systematic reviews and meta-analyses mainly focus on individual (usually migraine or pain) rather than multiple disorders, and on clinical symptoms rather than also including cognitive or safety outcomes. Nor have systematic reviews and meta-analyses explored the possible optimal stimulation parameters needed to apply eTNS for a given disorder. These limitations are, however, due to the comparatively small number of published studies available for meta-analysis. However, in recent years, there has been a significant increase in the number of published trials. Thus, an up-to-date systematic review and meta-analysis of the published literature is both warranted and timely.
To our knowledge, this is the first systematic review and meta-analysis that aimed to critically appraise and quantify evidence investigating the effects of eTNS across a range of neurological and psychiatric disorders. This is well-justified given the diffuse effects of eTNS on several networks relevant to a variety of psychiatric and neurological disorders. Our primary outcomes were clinical and cognitive measures, with secondary measures of safety. Where possible, we also sought to identify the optimal disorder and/or outcome-specific stimulation parameters to address the current lack of dosage guidance in the available literature.
Methods
Following a pre-registered protocol (PROSPERO ID: CRD42022322038; for changes, with justification, see Supplement) and the PRISMA 2020 guidelines, several databases were searched (up to 16th March 2022) with variations of “external Trigeminal Nerve Stimulation” as search keys, adapted for each database (for details, see Supplement; see also Supplementary PRISM 20202 Checklist). Four authors (SJW, AAC, WT, SX) independently i) screened all articles using our eligibility criteria (see Table 1), first at title/abstract level then at full-text level for articles that passed title/abstract screening; ii) extracted relevant data; and iii) assessed risk of bias of eligible report using the Risk of Bias (RoB) 2.0 tool [46] for RCTs and ROBINS-I [47] for non-randomized trials – with the overall risk of bias based on most severe RoB 2.0 or ROBINS-I level in any assessed domain. Disagreements were resolved consensually.
Data extraction
Means and standard deviations at all available time points were extracted from rating scales/subscales that directly measured neurological and/or psychiatric core symptoms and related clinical impairments (e.g., quality of life).
Statistical analysis
Standardized mean differences (SMDs) were calculated as mean baseline- to post-assessment (or follow-up) change in the intervention group minus the mean baseline- to post-assessment (or follow-up) change in the control group divided by the pooled baseline standard deviation with Hedges’ g adjustment [48]. Random effects meta-analyses estimated the effect of eTNS relative to control groups/conditions at all available assessment time points, with SMDs combined using the inverse variance method fitted via the DerSimonian-Laird method [49,50,51]. Analysis included at least two different trials reporting outcome measures of the same cognitive/clinical construct at similar time points (i.e., baseline, post-assessment, and follow-up). Between-SMD heterogeneity was tested using Q and the magnitude of true heterogeneity relative to random heterogeneity was estimated using the I2 statistic [49].
Pre-specified sensitivity analyses included analyses limited to trials (≥2) with 1) children/adolescents only; 2) adults only; 3) fixed stimulation intensity; 4) titrated stimulation intensity; 5) blinded outcome assessors; 6) unblinded outcome assessors; 7) the same control arms (e.g., sham control, treatment-as-usual, or waiting list control); and 8) longer-term outcomes measuring the same clinical or cognitive construct (grouped into outcomes measured at approximately 3, 6, or >9 months after the final TNS session). Finally, we ran Jacknife sensitivity analyses to identify influential single studies and the robustness of significant summary effect sizes estimates.
Where feasible (i.e., ≥10 studies per predictor) [52], separate meta-regressions tested associations between SMDs and either treatment period (in weeks), length of follow-up (in weeks), stimulation intensity, or mean age of participants.
Finally, Egger’s regression test was conducted on significant meta-analyses where heterogeneity was not significant or high. Analyses were conducted using metafor in R [50].
Results
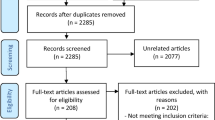
Of the 2738 potentially eligible reports, our systematic review included 48 separate trials (27 RCTs, 21 single-arm trials; reported in 55 published articles), of which 12 were meta-analyzed (for included studies, see Table 2; for exclusions, with reasons for exclusion, see Supplementary Fig. 1 [PRISMA 2020 Flowchart] and Supplementary Table 1).
Of the 27 RCTs assessed via RoB 2.0, most (N = 17) were rated with “High” risk of bias, mainly due to poor blinding (Domain 4), or having “some concerns” primarily driven by a lack of clear pre-specified outcomes (N = 25) (Domain 5) or inadequate randomization (N = 12) (Domain 1; e.g., poor balance between groups at baseline; see Supplementary Fig. 2). Twenty-seven articles were published from the 21 single-arm trials. All articles were assessed via ROBINS-I and were judged to have “Serious” risk of bias owing to lack of blinding (Supplementary Fig. 2).
Systematic review results
Attention-deficit/hyperactivity disorder (ADHD)
One open-label trial (N = 21, children/adolescents with ADHD) reported significant improvement at 4 and 8 weeks of nightly eTNS relative to baseline in investigator-rated parent reports of ADHD symptoms (primary outcome), with most effects observed at 4 weeks; as well as in secondary outcomes of: investigator-rated parent reported subscores of inattention and hyperactivity/impulsivity symptoms; clinical global impression improvement scale; parent-rated severity of ADHD and related impairments; several subscales of sleep measures, and several subscores of behavioral executive functioning (with the strongest being working memory); self-rated dimensional measures of depression; and incongruent reaction time on the attention network task, a measure of interference inhibition. Self-rated measures of anxiety were unchanged as well as all other cognitive measures [53].
In a double-blind, parallel-arm RCT (N = 59 children and adolescents with ADHD), a significant group by time interaction indicated that 4 weeks of nightly real relative to sham eTNS significantly reduced clinician-rated total parent-reported symptoms of ADHD (primary outcome), and secondary outcomes of clinician-rated parent reports of subscores of inattentive and hyperactivity/impulsivity symptoms, and clinical global impression. There was no significant change in secondary outcomes of: parent- or teacher-rated severity of ADHD and related impairments; parent-rated sleep measures, and behavioral executive functioning; parent- or child-rated irritability symptoms, and symptoms of anxiety; clinician-rated symptoms of depression and suicidality; and spatial working memory or attention network task performance. In both groups, 1 week after stimulation cessation, there was a significant increase in clinician-rated parent reported ADHD total symptoms [26]. Resting-state qEEG spectral power in right frontal (Delta, Theta, Beta, Gamma frequency bands) and frontal midline (Gamma frequency band) regions increased significantly immediately after real but not sham eTNS, with right frontal (Theta, Beta) and midline (Gamma) frequency band changes correlating with reduction in ADHD total and hyperactivity/impulsivity subscale ratings in the eTNS group. After trial completion, twenty sham participants received 4 weeks of nightly eTNS, which led to a significant group-by-time interaction, suggesting significantly improved parent-rated behavioral executive functioning immediately after stimulation in eTNS “responders” (N = 10, ADHD-RS Total Score <25%) compared to “non-responders” [54].
In summary, 4 weeks of eTNS has been shown to reduce core symptoms of ADHD, but in only one double-blind RCT [26]. There is limited evidence of improvement in other ADHD-related impairments, behavioral executive functioning, symptoms of depression and anxiety, and task-based measures of neuropsychological functioning. Longer-term effects are yet to be explored.
Depression
Two double-blind parallel-arm RCTs in 24 [55] or 40 [56] adults with major depressive disorder (MDD) reported a significant group-by-time interaction, indicating significantly reduced self-rated dimensional measures of symptoms of depression immediately and one-month after 10 nights of real compared to sham eTNS (primary outcome measure) [55], with a significant improvement in response (>50% fewer depression symptoms) but not remission rates (<8 HDRS-17 score) [56]. A significant group-by-time interaction showed that Montreal Cognitive Assessment (MoCA) scores were significantly improved one-month after real compared to sham eTNS in one trial [56], but MoCA and self-reported quality of life were unchanged in the other (secondary outcomes) [55].
Two open-label trials reported significant improvements from baseline in symptoms of depression [57, 58] and anxiety [58], immediately and one-month after 10 days of eTNS [58]. MoCA remained unchanged [57]. Two other open-label trials found significant improvement from baseline at 2, 4, 6, and 8 weeks of daily eTNS in clinician- or self-rated symptoms of depression [19, 59, 60], and at 8 weeks of eTNS in clinician-rated clinical global impression [59, 60] and self-rated quality of life [19, 59, 60].
In summary, eTNS has been shown to significantly improve depression symptoms at first end-point and one-month after stimulation cessation (with two double-blinded RCTs), with limited evidence of improvement in symptoms of anxiety, quality of life or MoCA.
Epilepsy
One double-blind, parallel-arm RCT (N = 42, with drug resistant, partial-onset epilepsy) reported significant improvements immediately after 18 weeks of daily 120 Hz versus 2 Hz eTNS based on change scores (baseline-endpoint) in ratings of symptoms of depression (secondary outcome), but not seizure frequency or response rates (i.e., >50% reduction in seizures relative to baseline), and time to the fourth seizure (primary outcomes) [61]. After trial completion, thirty-five participants (19 from eTNS group; 16 from active control group) received 12 months of daily unblinded eTNS. Seizure frequency was significantly reduced at 6 months versus baseline and at 12 months versus 6 months, but only in participants who previously received eTNS in the blinded RCT [62].
One parallel-arm, unblinded RCT in adults with drug-resistant epilepsy (N = 40 with temporal or frontal epilepsy unsuitable for surgery) reported significant improvement at 6 and 12 but not at 3 months of daily eTNS compared to treatment-as-usual (TAU) in primary outcomes of response rates (≥50% reduction in seizure frequency), and in secondary outcomes of changes from baseline (i.e., baseline—endpoint) in seizure frequency and quality of life scores. At 12 months, response rates and changes from baseline in seizure frequency were significantly improved in participants with temporal relative to frontal epilepsy. There was no group difference in changes from baseline in ratings of symptoms of depression and anxiety symptoms or task-based measures of logical memory, visual memory, auditory learning, cognitive flexibility, working memory, and naming [63]. Another parallel-arm, unblinded RCT (N = 30 with drug-resistant epilepsy) compared 6 months of 120 Hz versus 20 Hz eTNS. Change scores (baseline—3 or 6 months of eTNS) showed a significant improvement in favor of 120 Hz versus 20 Hz eTNS in seizure severity and rates of depression (i.e., HRSD score ≥20) at 3 and 6 months, and seizure frequency and symptoms of depression at 6 months, but not 3 months [64].
One open-label trial reported significant improvement from baseline in daily seizures at 3 but not 6 or 12 months of eTNS [65,66,67,68], or self-rated quality of life and symptoms of depression, but not sleep measures, immediately after 18 weeks of eTNS (M = 42, adults with drug-resistant epilepsy) [69]. Another open label trial observed but did not test a 35% response rate in seizure frequency at 6 and 12 months and 14% at 48 months of eTNS [70].
In summary, eTNS has been shown to reduce seizure frequency, symptoms of depression, and quality of life, in people with epilepsy, but the evidence is based on mostly unblinded studies with only one unblinded RCT showing no effects on seizure frequency [61].
Trigeminal neuralgia
One double-blind, parallel-arm RCT (N = 52 adults with trigeminal neuralgia) combined real or sham eTNS with carbamazepine over 6-weeks. The mean dose of carbamazepine prescribed to participants was significantly reduced immediately, 6-weeks, 12-weeks, but not 3-months, after real stimulation compared to sham, while pain intensity and its effect on everyday functioning remained unchanged. No group-by-time interaction was conducted [71]. One parallel-arm, unblinded head-to-head trial (N = 31 adults with trigeminal neuralgia) reported improvement in pain intensity with constant or theta-burst eTNS, for 3 weeks, but without statistical analyses [72].
In summary, evidence supporting the clinical relevance of eTNS in trigeminal neuralgia is limited with only one double-blind RCT, but there are initial indicators of improvement regarding medication intake and pain intensity.
Migraine
Four parallel-arm (three unblinded, one double-blinded) RCTs applied 3 months of daily eTNS. One double-blind RCT (N = 67 adults with migraine) reported significant group difference favoring 60 versus 1 Hz eTNS in all primary outcomes (i.e., responders [>50% reduction from baseline in monthly migraines], and baseline—endpoint change in migraine days) and secondary outcomes (i.e., baseline—endpoint change in migraine attacks, headache days, and anti-migraine drug intake), but migraine severity was unchanged [41, 73]. In one RCT (N = 124 adults with migraine), change scores (i.e., baseline—post-assessment immediately after stimulation) based on measures of headache severity, duration, frequency, response rates (i.e., >25% reduction in headache frequency and duration), and quality of life showed significant improvement 8 weeks after twice-weekly daytime eTNS plus flunarizine relative to flurazine alone [74]. In another RCT (N = 76 adults with migraine, measures of headache pain, frequency, duration, and response rates (i.e., >25% reduction in headache frequency and duration) were significantly reduced immediately after 12 weeks of twice-weekly daytime eTNS plus nimodipine versus nimodipine alone; however, baseline scores were not included in the analysis and the difference might hence be due to chance [75]. Finally, one head-to-head RCT (N = 90 adults with migraine) found significant improvement versus baseline in migraine/headache symptoms and anti-migraine drug-use, but not accompanying symptoms, immediately after eTNS or mastoid electrical stimulation versus baseline only, while change scores (baseline—endpoint) of the impact of headaches daily functioning significantly improved with eTNS versus mastoid electrical stimulation. Symptoms accompanying migraine were unaffected [76].
One double-blinded, parallel-arm RCT (N = 45 adults with chronic type tension headache) reported significantly improved headache pain and its impact on quality of life immediately after 8 weeks of daily eTNS plus physiotherapy versus analgesic medication or physiotherapy alone, but no group by time interaction was tested [77].
Four parallel-arm unblinded RCTs applied daily eTNS for one-month. One (N = 154 adults with migraine) reported significantly reduced migraine frequency, pain intensity, and anti-migraine rescue medication, and a higher number of responders (≧50% reduction of migraine frequency) immediately after eTNS plus flurazine versus flurazine or eTNS alone. Flurazine alone significantly improved change scores in migraine intensity only when compared to eTNS alone [42]. A second RCT (N = 60 adults with migraine) found that – compared to rizatriptan benzoate alone or eTNS alone – eTNS plus rizatriptan benzoate led to significantly improved change scores (i.e., baseline—post-assessment) in headache frequency and pain intensity at 30-days but not 7 or 14 days after stimulation [78]. A third RCT (N = 180 adults with migraine) reported significantly improved change scores (i.e., baseline—immediately after stimulation) in headache pain severity, quality of life, and sleep quality immediately after eTNS plus nimesulide relative to nimesulide alone, with a significant higher number of recurrence of headaches at 3 months in the nimesulide alone versus eTNS plus nimesulide group (34% versus 20%) [79]. The fourth RCT (N = 74 adults with migraine) analyzed change scores (i.e., baseline—10, 20, or 30-days of stimulation), and found that twice-daily eTNS plus rizatriptan benzoate relative to rizatriptan benzoate alone significantly improved headache frequency at 10 days and pain at 10-, 20-days, and 30-days [80].
Three parallel-arm unblinded RCTs applied eTNS over several days. Significant improvements were reported in: migraine symptoms immediately after one-week of eTNS versus TAU [81] (N = 118 adults with migraine); time without headaches (but not pain or everyday functioning) immediately after 10 days of twice daily eTNS plus flunarizine hydrochloride versus [82] (N = 60 adults with migraine); and headache frequency and duration 1 month after 10 days of eTNS but not TAU compared to baseline and in change scores (baseline—endpoint) in pain intensity 1 month after eTNS versus TAU [83] (N = 91 with migraine or other primary headaches). In the latter, a subsample (N not reported) of high self-reported pain ratings immediately after eTNS showed a significantly greater reduction in primary headache pain 30-days after eTNS versus TAU [83].
One head-to-head unblinded RCT (N = 120 adults with chronic headache) compared two brands of eTNS devices, eTNS with the MK-MT11 device (Maikang Medical Instrument Company, Beijing) versus the Cefaly device (STX-Med Sprl, Belgium) and found that change scores (baseline—post-treatment) in the number of headache attacks and headache pain intensity did not significantly differ between the two eTNS devices [84].
Three double-blind, parallel-arm RCTs in adults with migraine [39, 40, 85] administered single session eTNS. A significant group-by-time interaction suggested significant improvements relative to baseline immediately and 100-minutes after eTNS but not sham stimulation (N = 78) [40]. eTNS versus sham stimulation significantly predicted improvements in freedom from pain and pain relief immediately and 24 h after stimulation and in migraine-associated symptoms immediately after stimulation, but there was no change in anti-migraine rescue medication intake (N = 538) [85]. Finally, a significant group difference in baseline—endpoint change in migraine pain and the proportion of pain-free participants was found immediately after 100 Hz versus 3 Hz eTNS, with the migraine pain reduction only persisting 2 and 24 h after eTNS (N = 106) [39].
One crossover, unblinded RCT reported but did not test reduced pain from baseline immediately after single-session eTNS over the trigeminal or occipital and trigeminal nerves, with greater reduction in the latter (N = 10) [86]. One parallel-arm unblinded RCT (N = 108 adults with migraine) found significantly improved headache pain severity, and quality of life, immediately after eTNS versus non-specified nerve stimulation [87]. Stimulation duration was not reported.
Across 11 open-label trials (N Mean 36, range 17–100), eTNS led to significant improvements compared to baseline, which are summarized as follows. Six trials found a significant migraine/headache reduction at 8 weeks (but not 4 weeks) of eTNS [88] and/or immediately after 1 [89, 90] and/or 3 months of eTNS [22, 88, 91, 92]. Three out of four trials reported a significant reduction in anti-migraine medication intake immediately after 1 month [89] or 3 months of eTNS [91, 93], while one trial found no effect on medication intake [92]. Five out of seven trials found that pain intensity was reduced i) immediately after 60 min of eTNS and again 60 min later [94] or ii) 1 [89] or 3 months of eTNS [91, 93], but two trials found no effect on pain intensity [88, 92]. Finally, one trial found that migraine duration was reduced after 3 months of eTNS [91]. In addition to these 11 trials, a further four found – but did not statistically analyze – reductions from baseline in migraine pain, migraine symptoms, vertigo, or headache severity immediately after 20 min [95] and again 24 h later in [96] or 1 month of eTNS [97]; migraine frequency over 4 months of eTNS [98]; and anti-migraine medication intake after 20 min and again 24 h later [96] or over 4 months of eTNS [98].
In summary, there is evidence, consistent across studies, of reduced migraine frequency and/or symptoms (namely pain) with continued use of eTNS versus sham. There is some evidence that these improvements persist after stimulation or that eTNS can also improve the impact of migraine on quality of life. There was a lack, however, of well-blinded control arms with a few exceptions that showed clinical improvements [39,40,41, 85].
Insomnia
An open-label pilot study (N = 13 adults with insomnia) showed significantly improved self-reported sleep quality, insomnia severity, sleepiness in daily life, but not polysomnographic measures of sleep, immediately after 4 weeks of eTNS relative to baseline [99].
Tolerability, adverse events
Overall, eTNS was well-tolerated with no severe adverse events reported across any of the studies. The most commonly reported mild adverse events included mild and transitory itching, skin redness, pain, or paresthesia, usually reported in a minority of participants in any given trial. Several studies reported participants dropping out due to discomfort, but these were equivalent across eTNS or comparator arms (see Tables 3 and 4; Supplementary Fig. 3).
Meta-analyses results
Twelve RCTs were included in our meta-analyses, making a total of 1,048 participants [26, 40,41,42, 63, 64, 74, 76, 78,79,80, 87]. These studies applied eTNS alone [26, 40, 41, 76, 78, 87], or eTNS plus TAU [63] or eTNS plus another medication that was part of the trial, such as anti-migraine medication [42, 74, 79] or breathing exercises [100]. These interventions (eTNS alone or plus another intervention) were compared to sham eTNS [26, 40, 41], or another form of stimulation [64, 76], TAU [63], or a medication treatment [42, 74, 78]. We were unable to include several outcomes from six RCTs in our meta-analyses (see Supplements for excluded outcomes, with reasons). Unfortunately, there were insufficient trials to analyze other outcomes measuring neuropsychological processes or neurophysiology (e.g., heart rate variability).
Migraine pain intensity
We found no significant improvement in favor of eTNS applied alone versus a comparator (i.e., sham, medication control, or active control) when all trials were analyzed (K = 6, SMD = 0.63, 95% CI[−0.26–1.51]), nor in sensitivity analyses limited to trials with sham control, medication control, or with blinded outcome assessors. Between effect size heterogeneity was high and statistically significant (see Table 5, Fig. 1; all I2 roughly 95, and significant Qs all p < 0.001). However, a post hoc analysis showed a significant, large improvement in favor of eTNS when combined with an anti-migraine medication versus medication alone, which was associated with low and non-significant heterogeneity (K = 4, SMD = 1.03, 95% CI[0.84–1.23]; I2 = 0, Q p = 0.78). Jacknife sensitivity analysis (i.e., repeating the analysis with a different trial excluded each time) showed that the significant improvement in pain intensity with all trials included was robust, with no change in effect direction or significance level, with effect size ranging from moderate to large, while heterogeneity remained low and non-significant (see Table 6).
Effects have been groups according to whether eTNS was applied alone (A) or when combined with anti-migraine medication (B). The summary effect size and its precision (95% confidence interval) are indicated by the diamond, with the dotted line indicating the dispersion of the true effect (i.e., 95% prediction interval). All post-assessment time points were <24 h after the last eTNS session, with the exception of An et al. (2020), which was conducted during treatment period at week 8 of stimulation. A positive effect indicates an effect in favor of the active eTNS intervention. Legend. FLZ Flurazine, NM Nimesulide, RBZ Rizatriptan.
Quality of Life
We found a significant, large improvement in quality of life that was in favor of eTNS plus anti-migraine compared to medication alone (K = 2, SMD = 1.88, 95% CI[1.22.5–2.53]). However, heterogeneity was high and statistically significant (see Table 5, Fig. 2).
The summary effect size and its precision (95% confidence interval) are indicated by the diamond, with the dotted line indicating the dispersion of the true effect (i.e., 95% prediction interval). All post-assessment time points were <24 h after the last eTNS session, with the exception of An et al. (2020), which was conducted during treatment period at week 8 of stimulation. A positive effect indicates an effect in favor of the active eTNS intervention. Legend. FLZ Flurazine, NM Nimesulide.
Depression
We found a significant, moderate improvement in dimensional measures of depression symptoms in patients with epilepsy with (K = 1) and without depression (K = 2) or with ADHD (K = 1) that favored eTNS relative to sham (ADHD), TAU (epilepsy without depression) or 20 Hz eTNS (epilepsy with depression) (K = 3, SMD = 0.45, 95% CI[0.01–0.88]), which was no longer significant when limited to trials with only adult samples (K = 2). Heterogeneity was low and statistically non-significant (see Table 5, Fig. 3). Jacknife sensitivity analysis revealed that this effect was no longer significant when we removed three studies [26, 63, 64]. Heterogeneity remained low and statistically non-significant (see Table 6).
The summary effect size and its precision (95% confidence interval) are indicated by the diamond, with the dotted line indicating the dispersion of the true effect (i.e., 95% prediction interval). All post-assessment time points were <24 h after the last eTNS session. A positive effect indicates an effect in favor of the active eTNS intervention.
Other outcome measures
We found no significant effect on monthly anti-migraine drug use, monthly migraine attacks, and migraine days. With the exception of the latter, all analyses were associated with high and statistically significant heterogeneity (see Table 5, Figs. 4–6).
The summary effect size and its precision (95% confidence interval) are indicated by the diamond, with the dotted line indicating the dispersion of the true effect (i.e., 95% prediction interval). All post-assessment time points were <24 h after the last eTNS session. A positive effect indicates an effect in favor of the active eTNS intervention. Legend. FLZ Flurazine, PMS Mastoid Stimulation.
The summary effect size and its precision (95% confidence interval) are indicated by the diamond, with the dotted line indicating the dispersion of the true effect (i.e., 95% prediction interval). All post-assessment time points were <24 h after the last eTNS session. A positive effect indicates an effect in favor of the active eTNS intervention. Legend. FLZ Flurazine, PMS Mastoid Stimulation.
The summary effect size and its precision (95% confidence interval) are indicated by the diamond, with the dotted line indicating the dispersion of the true effect (i.e., 95% prediction interval). All post-assessment time points were <24 h after the last eTNS session. A positive effect indicates an effect in favor of the active eTNS intervention. Legend. PMS Mastoid Stimulation.
Meta-regression
Only the analysis of migraine pain intensity outcomes had the required number of trials (i.e., 10 or more per predictor) to conduct a meta-regression analysis. The overall effect size estimates were not significantly predicted by follow-up length, duration of treatment, or mean age (see Supplementary Table 2).
Publication bias
Egger’s regression test was non-significant for the analysis of dimensional measures of depression (p = 0.13). Egger’s regression test could not be conducted for the analyses on migraine pain intensity, migraine days, and quality of life because the significant heterogeneity would have confounded any interpretation of funnel plot asymmetry.
Discussion
This is the first systematic review and meta-analysis investigating the effects of eTNS across neurological and psychiatric disorders. In the meta-analysis, while we found no significant meta-analytic effect across 6 trials of 4–12 weeks of eTNS alone on migraine pain intensity, anti-migraine medication use, migraine days, and monthly migraine attacks, we found that eTNS combined with anti-migraine medication across 4 trials significantly improved migraine pain intensity and quality of life. Furthermore, we found that 4–12 months of eTNS applied alone improved dimensional measures of depression, but were likely limited to cases of depression with a clinical diagnosis of depression. Our findings, therefore, provide encouraging initial evidence supporting eTNS in combination with anti-migraine medication in reducing pain migraine intensity with, additionally, possible clinical utility in improving quality of life and – if applied alone – on depression symptoms in individuals with a clinical diagnosis of depression.
Our meta-analytic finding of improved migraine pain intensity corroborates and extends evidence from previous systematic reviews and meta-analyses that indicate analgesic effects of eTNS in migraine [33, 34, 36, 101,102,103,104]. Although the mechanisms of action are unknown, afferent projections from the brainstem via trigeminal nerve stimulation to structures involved in pain regulation or perception, such as the insula, thalamus and ACC, may be one means through which eTNS can modulate migraine pain perception [14, 17, 18]. Further, the fact that reduced migraine intensity was found only when eTNS was combined with anti-migraine medication, suggests that eTNS as an adjunct to medication treatment may elicit a synergistic effect on migraine symptoms. However, this analysis was based on four studies only, all of which had poorly blinded outcome assessors so that we cannot rule out that effect size estimates were inflated due to outcome assessor bias. Therefore, further RCTs with well-blinded outcomes and rigorous control conditions are needed to achieve a more accurate estimate of the eTNS effect.
We also found some indication of improved quality of life in participants with migraine and epilepsy across two studies. Given that both studies [74, 79] were also included in the analysis of migraine pain intensity, this might indicate that eTNS effects on migraine may transfer beyond symptom-specific outcomes to functional outcomes. However, this interpretation should be treated in the context of significant heterogeneity. At the very least, our findings point to the importance of measuring more functionally related outcomes, rather than focusing only on symptom outcomes in order not to miss important transfer effects of eTNS.
The finding that eTNS significantly improved dimensional measures of depression across three studies may suggest a possible transdiagnostic effect of eTNS on mood regulation. However, only one study included participants with epilepsy with co-occurring clinical depression [64], while the other two studies recruited participants with epilepsy [63] or ADHD [26] but provided dimensional measures of depressive symptoms. That eTNS may modulate mood chimes with evidence from anatomical studies of projections from the trigeminal nerve via the brainstem to key regions known to regulate mood, such as LC, raphe nuclei, nucleus tractus solitarius (NTS), medullary reticular activating system (RAS), and thalamic and fronto-limbic structures [84,85,86]. However, we cannot rule out that the effect was driven by the comparatively large and only significant effect reported by Zhang et al. (2018) [64]; as this was the only study to include participants with co-morbid depression, our findings in fact indicate most pronounced effects on clinical depression. Future studies should explore dimensionality effects further by including measures of mood in non-mood disorders.
Unfortunately, several eligible studies could not be included in the meta-analysis because we were unable to retrieve the relevant data, despite our efforts to gather them from study authors. It is therefore worth reflecting on whether inclusion of these studies would have altered the conclusions from our findings. For example, BDI significantly improved in favor of 120 Hz eTNS relative to 2 Hz eTNS in participants with drug-resistant epilepsy [61] or relative to sham in participants with MDD [56], thus supporting our finding of improvements in dimensional measures of depressive symptoms. In another study, quality of life remained unchanged in one study in participants with MDD [55]. Although at odds with our meta-analytic finding of improved quality of life in people with epilepsy and migraine, the lack of effects on quality of life in individuals with MDD is consistent with our tentative finding that quality of life may be improved in participants with migraine who also showed symptom improvement (thereby suggesting a transfer of improvement to quality of life) and/or in trials that combined eTNS with anti-migraine medication (thereby suggesting a synergistic effect of eTNS). However, this interpretation is speculative and would require further investigation.
Studies on three disorders (ADHD, epilepsy, and trigeminal neuralgia) were not included in our meta-analyses due to insufficient trials or unavailable data. In ADHD, one well-conducted double-blinded RCT found a significant improvement in ADHD symptoms with 4 weeks of eTNS versus sham based on blinded parent-ratings on a standard outcome measure (ADHD-Rating Scale) completed by a clinician [26], yet the increase in ADHD symptoms at the one-week follow up after eTNS discontinuation might indicate short-lived improvement. Further, qEEG data showed a positive correlation between reduced ADHD total and hyperactivity/impulsivity subscale ratings in the eTNS group only with right frontal (Theta, Beta) and midline (Gamma) frequency band changes [54], thus illuminating a possible mechanism of action and the specificity of eTNS effects. In epilepsy, daily eTNS use of up to one-year can significantly improve seizure frequency, dimensional measures of depression symptoms, and quality of life, but longer-term effects have not been studied, and most trials are unblinded or single-arm trials and therefore vulnerable to outcome assessor bias. In trigeminal neuralgia, there is evidence of reduced medication intake and inconsistent evidence of reduced pain intensity, but this is based on only two trials that failed to test or report a significant group-by-time interaction [71, 72]. Therefore, while on balance our meta-analytic findings favor the use of eTNS combined with anti-migraine medication in migraine, there is encouraging albeit emerging evidence of wider applications in ADHD and epilepsy particularly, that need replication and further exploration with well-blinded RCTs with longer-term follow-ups. We are aware of two ongoing and prospectively registered trials in 7–12 year old children with ADHD in the USA (N = 280, NCT05374187) and our study in the UK in 8–18-year-old children/adolescents with ADHD (N = 150, ISRCTN82129325) that aim to replicate and expand these findings in much larger samples (with a comprehensive set of clinical, neurocognitive and neuroimaging outcomes and longer follow-ups of 6 months).
Finally, our findings demonstrate that eTNS is well-tolerated with a good adverse-event profile. No study reported any severe adverse event that could be attributable to eTNS. Further, while mild or moderate adverse events were associated with eTNS in the majority of studies, these were limited to mild skin irritation, redness, or discomfort under the electrodes, headaches, and/or fatigue during stimulation, all of which were well-tolerated by a majority of participants, were transient and resolved on their own. Several studies found no significant effects on vital signs (e.g., blood pressure, heart rate variability), which might have been affected due to brainstem stimulation. Four studies [39, 40, 97, 98] reported dropouts from a minority of participants due to pain, but this may be related to a high current intensity (i.e., 16 mA) applied in 3 of them [39, 97, 98]. Allowing participants to adjust the current intensity that is comfortable for them may reduce dropouts [7, 19, 26, 53]. However, researchers opting to titrate stimulation in this manner should bear in mind that the efficacy of other forms of non-invasive brain stimulation (e.g., TMS, tDCS, vague nerve stimulation) partly rely on current intensity [105, 106] - yet this has not been directly investigated in eTNS.
As with any systematic review, ours was limited by limitations in the included studies. First, of the 26 RCTs, only 10 employed well-blinded outcome assessments. Future studies should strive to conceal knowledge of group assignment in order to minimize the risk of outcome assessor bias that can potentially inflate efficacy of eTNS. Second, despite increasing homogeneity by clustering outcomes that measured the same clinical or neuropsychological constructs across studies in the same or similar populations, several of our meta-analyses were associated with significant heterogeneity, which can limit the interpretability of our findings. Future studies should include outcomes that are both appropriate and facilitate comparisons across studies and evidence syntheses. Third, due to low power in most of our analyses, related to the paucity of data gathered in individual studies, we were unable to explore eTNS parameters that may lead to optimal and reliable outcomes. In the context of a field-wide lack of dosage guidance, we urge future studies to explore stimulation parameters that may optimize eTNS effects [31, 107, 108]. Forth, in many studies, key stimulation parameters were not reported sufficiently, with missing information regarding repetition frequency, pulse width, duty cycle, time of stimulation (night or day), and waveform. Failing to transparently report this information undermines the reproducibility of past findings, but may also introduce other methodological issues. For instance, while eTNS waveform is invariably never reported, it may differ across devices even if all the major parameters are equal (repetition frequency, pulse width, intensity), which could explain differences in subjective experiences of eTNS depending on the device used (I. Cook, 2021 via personal communication). To our knowledge, there are no formal guidelines for best-practice reporting of eTNS stimulation parameters, but we strongly recommend that future researchers consult standards set out for similar devices and work collaboratively to establish reporting guidelines [105]. Fifth, several studies did not measure tolerability or adverse-events, and only a minority measured vital signs (e.g., heart rate, blood pressure etc). We strongly encourage that future studies monitor potential adverse events and broaden outcome measures to capture potential downsides to eTNS. This is especially important given that – although our findings support the view that it is relatively safe –no available guidance on the optimal dose and safe administration of eTNS exists. Sixth, the interpretation of our meta-analytic findings is limited by lack of trials that compared eTNS with an active treatment control. We therefore consider it premature to compare the relative superiority or inferiority of eTNS with another treatment. Instead, future studies should first establish if eTNS is efficacious in providing clinical, cognitive, or brain function improvement compared to a well-blinded, rigorous no-treatment control arm, ideally one that is as comparable to eTNS as possible (e.g., sham or very low frequency eTNS control). Finally, we were unable to include data from several eligible studies. In the spirit of the Open Science movement, authors of future studies should make their data available on request or – more preferably – ensure that it is stored on a publicly accessible platform.
Conclusion
Compared to other NIBS techniques (i.e. tDCS, tRNS, tACS, TMS), eTNS has a diffuse “bottom-up” mechanism of action that activates (via the brainstem) many different fronto-cortico-thalamic, fronto-cerebellar and fronto-limbic regions and pathways as well as different neurotransmitters that are affected in many diverse disorders [6, 7, 19,20,21], suggesting potential transdiagnostic effects. Collectively, the studies included in our systematic review suggest that eTNS is a well-tolerated and safe technique for use in ADHD, depression, trigeminal neuralgia, migraine, and insomnia. Our meta-analysis found that eTNS can improve migraine pain intensity and quality of life when combined with anti-migraine medication or dimensional measures of depression when applied alone, albeit likely limited to individuals with a clinical diagnosis of depression. Our review shows evidence for potential improvement of other disorders such as ADHD and epilepsy, which will need to be corroborated by further RCTs. Conclusive meta-analytic evidence was precluded by heterogeneous stimulation protocols and outcome measures. Future studies should ideally be adequately powered, include well-blinded no-treatment controls, with homogeneous protocols to test both clinical and cognitive outcomes to address the current lack of dosage guidance regarding the optimal stimulation parameters (e.g., stimulation characteristics, number of sessions, timing).
Data availability
All data is available upon reasonable request.
References
GBD 2016 Neurology Collaborators, Daryani A, De la Cruz-Góngora V, De la Hoz FP, Deribe K, Dharmaratne SD, et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:459–80.
GBD 2019 Mental Disorders Collaborators. Global, regional, and national burden of 12 mental disorders in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Psychiatry. 2022;9:137–50.
Feigin VL, Nichols E, Alam T, Bannick MS, Beghi E, Blake N, et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:459–80.
Feigin VL, Vos T, Nichols E, Owolabi MO, Carroll WM, Dichgans M, et al. The global burden of neurological disorders: translating evidence into policy. Lancet Neurol. 2020;19:255–65.
Kang S, Eum S, Chang Y, Koyanagi A, Jacob L, Smith L, et al. Burden of neurological diseases in Asia from 1990 to 2019: a systematic analysis using the Global Burden of Disease Study data. BMJ Open. 2022;12:e059548.
Adair D, Truong D, Esmaeilpour Z, Gebodh N, Borges H, Ho L, et al. Electrical stimulation of cranial nerves in cognition and disease. Brain Stimul. 2020;13:717–50.
Cook IA, Espinoza R, Leuchter AF. Neuromodulation for depression. Neurosurg Clin N Am. 2014;25:103–16.
van Boekholdt L, Kerstens S, Khatoun A, Asamoah B, Mc Laughlin M. tDCS peripheral nerve stimulation: a neglected mode of action? Mol Psychiatry. 2021;26:456–61.
Iidaka T. Fluctuations in arousal correlate with neural activity in the human thalamus. Cereb Cortex Commun. 2021;2:tgab055.
Maldonato M. The ascending reticular activating system. In: Bassis S, Esposito A, Morabito FC, editors. Recent advances of neural network models and applications. Cham: Springer International Publishing, 2014. p. 333–44. (Smart Innovation, Systems and Technologies).
Poe GR, Foote S, Eschenko O, Johansen JP, Bouret S, Aston-Jones G, et al. Locus coeruleus: a new look at the blue spot. Nat Rev Neurosci. 2020;21:644–59.
Tramonti Fantozzi MP, Artoni F, Di Galante M, Briscese L, De Cicco V, Bruschini L, et al. Effect of the trigeminal nerve stimulation on auditory event-related potentials. Cereb Cortex Commun. 2021;2:tgab012.
De Cicco V, Tramonti Fantozzi MP, Cataldo E, Barresi M, Bruschini L, Faraguna U, et al. Trigeminal, visceral and vestibular inputs may improve cognitive functions by acting through the locus coeruleus and the ascending reticular activating system: a new hypothesis. Front Neuroanat. 2018;11:130.
Alper J, Seifert AC, Verma G, Huang KH, Jacob Y, Al Qadi A, et al. Leveraging high-resolution 7-tesla MRI to derive quantitative metrics for the trigeminal nerve and subnuclei of limbic structures in trigeminal neuralgia. J Headache Pain. 2021;22:112.
Kolling N, Wittmann M, Behrens T, Boorman E, Mars R, Rushworth M. Anterior cingulate cortex and the value of the environment, search, persistence, and model updating. Nat Neurosci. 2016;19:1280–5.
Shenhav A, Cohen JD, Botvinick MM. Dorsal anterior cingulate cortex and the value of control. Nat Neurosci. 2016;19:1286–91.
Danyluk H, Andrews J, Kesarwani R, Seres P, Broad R, Wheatley BM, et al. The thalamus in trigeminal neuralgia: structural and metabolic abnormalities, and influence on surgical response. BMC Neurol. 2021;21:290.
Uddin LQ, Nomi JS, Hebert-Seropian B, Ghaziri J, Boucher O. Structure and function of the human insula. J Clin Neurophysiol Publ Am Electroencephalogr Soc. 2017;34:300–6.
Cook IA, Abrams M, Leuchter AF. Trigeminal nerve stimulation for comorbid posttraumatic stress disorder and major depressive disorder. Neuromodulation Technol Neural Interface. 2016;19:299–305.
Kamel HAM, Toland J. Trigeminal nerve anatomy. Am J Roentgenol. 2001;176:247–51.
Mercante B, Deriu F, Rangon CM. Auricular neuromodulation: the emerging concept beyond the stimulation of vagus and trigeminal nerves. Med Basel Switz. 2018;5:10.
Magis D, D’Ostilio K, Thibaut A, De Pasqua V, Gerard P, Hustinx R, et al. Cerebral metabolism before and after external trigeminal nerve stimulation in episodic migraine. Cephalalgia. 2017;37:881–91.
Hansen JY, Shafiei G, Markello RD, Smart K, Cox SML, Nørgaard M, et al. Mapping neurotransmitter systems to the structural and functional organization of the human neocortex. Nat Neurosci. 2022;25:1569–81.
Ng J, Papandreou A, Heales S, Kurian M. Monoamine neurotransmitter disorders - clinical advances and future perspectives. Nat Rev Neurol. 2015;11:567–84.
Englot DJ, D’Haese PF, Konrad PE, Jacobs ML, Gore JC, Abou-Khalil BW, et al. Functional connectivity disturbances of the ascending reticular activating system in temporal lobe epilepsy. J Neurol Neurosurg Psychiatry. 2017;88:925.
McGough JJ, Sturm A, Cowen J, Tung K, Salgari GC, Leuchter AF, et al. Double-blind, sham-controlled, pilot study of trigeminal nerve stimulation for attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2019;58:403–11.e3.
D’Andrea G, D’Arrigo A, Dalle Carbonare M, Leon A. Pathogenesis of migraine: role of neuromodulators. Headache. 2012;52:1155–63.
Wang F, Wang J, Cao Y, Xu Z. Serotonin–norepinephrine reuptake inhibitors for the prevention of migraine and vestibular migraine: a systematic review and meta-analysis. Reg Anesth Pain Med. 2020;45:323–30.
Zhou C, Guo T, Bai X, Wu J, Gao T, Guan X, et al. Locus coeruleus degeneration is associated with disorganized functional topology in Parkinson’s disease. NeuroImage Clin. 2021;32:102873.
Paredes-Rodriguez E, Vegas-Suarez S, Morera-Herreras T, De Deurwaerdere P, Miguelez C. The noradrenergic system in Parkinson’s disease. Front Pharmacol. 2020;11. https://www.frontiersin.org/articles/10.3389/fphar.2020.00435/full.
Westwood SJ, Radua J, Rubia K. Noninvasive brain stimulation in children and adults with attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. J Psychiatry Neurosci JPN. 2021;46:E14–33.
Clark O, Mahjoub A, Osman N, Surmava AM, Jan S, Lagman-Bartolome AM. Non-invasive neuromodulation in the acute treatment of migraine: a systematic review and meta-analysis of randomized controlled trials. Neurol Sci J Ital Neurol Soc Ital Soc Clin Neurophysiol. 2022;43:153–65.
Garcia-Isidoro S, Castellanos-Sanchez VO, Iglesias-Lopez E, Perpiña-Martinez S. Invasive and non-invasive electrical neuromodulation in trigeminal nerve neuralgia: a systematic review and meta-analysis. Curr Neuropharmacol. 2021;19:320–33.
Moisset X, Pereira B, Ciampi de Andrade D, Fontaine D, Lantéri-Minet M, Mawet J. Neuromodulation techniques for acute and preventive migraine treatment: a systematic review and meta-analysis of randomized controlled trials. J Headache Pain. 2020;21:142.
Shiozawa P, Silva ME, Carvalho TC, Cordeiro Q, Brunoni AR, Fregni F. Transcutaneous vagus and trigeminal nerve stimulation for neuropsychiatric disorders: a systematic review. Arq Neuropsiquiatr. 2014;72:542–7.
Stanak M, Wolf S, Jagoš H, Zebenholzer K. The impact of external trigeminal nerve stimulator (e-TNS) on prevention and acute treatment of episodic and chronic migraine: a systematic review. J Neurol Sci. 2020;412. https://www.jns-journal.com/article/S0022-510X(20)30061-7/fulltext.
VanderPluym JH, Halker Singh RB, Urtecho M, Morrow AS, Nayfeh T, Torres Roldan VD, et al. Acute treatments for episodic migraine in adults: a systematic review and meta-analysis. JAMA. 2021;325:2357–69.
Lauritsen CG, Silberstein SD. Rationale for electrical parameter determination in external trigeminal nerve stimulation (eTNS) for migraine: a narrative review. Cephalalgia Int J Headache. 2019;39:750–60.
Chou DE, Shnayderman Yugrakh M, Winegarner D, Rowe V, Kuruvilla D, Schoenen J. Acute migraine therapy with external trigeminal neurostimulation (ACME): a randomized controlled trial. Cephalalgia. 2019;39:3–14.
Hokenek NM, Erdogan MO, Hokenek UD, Algin A, Tekyol D, Seyhan AU. Treatment of migraine attacks by transcutaneous electrical nerve stimulation in emergency department: a randomize controlled trial. Am J Emerg Med. 2021;39:80–5.
Schoenen J, Vandersmissen B, Jeangette S, Herroelen L, Vandenheede M, Gerard P, et al. Migraine prevention with a supraorbital transcutaneous stimulator: A randomized controlled trial. Neurology 2013;80:697–704.
Jiang L, Yuan DL, Li M, Liu C, Liu Q, Zhang Y, et al. Combination of flunarizine and transcutaneous supraorbital neurostimulation improves migraine prophylaxis. Acta Neurol Scand. 2018;139:276–83.
Li H, Xu QR. Effect of percutaneous electrical nerve stimulation for the treatment of migraine. Medicine. 2017;96:e8108.
Silberstein SD, Calhoun AH, Lipton RB, Grosberg BM, Cady RK, Dorlas S, et al. Chronic migraine headache prevention with noninvasive vagus nerve stimulation: the EVENT study. Neurology. 2016;87:529–38.
Liu Y, Dong Z, Wang R, Ao R, Han X, Tang W, et al. Migraine prevention using different frequencies of transcutaneous occipital nerve stimulation: a randomized controlled trial. J Pain. 2017;18:1006–15.
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898.
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919.
Morris SB. Estimating effect sizes from pretest-posttest-control group designs. Organ Res Methods. 2008;11:364–86.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions version 6.2 (updated February 2021). Cochrane. 2021. www.training.cochrane.org/handbook.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36:1–48.
DerSimonian R, Kacker R. Random-effects model for meta-analysis of clinical trials: an update. Contemp Clin Trials. 2007;28:105–14.
Borenstein M, Higgins JPT. Meta-analysis and subgroups. Prev Sci J Soc Prev Res. 2013;14:134–43.
McGough JJ, Loo SK, Sturm A, Cowen J, Leuchter AF, Cook IA. An eight-week, open-trial, pilot feasibility study of trigeminal nerve stimulation in youth with attention-deficit/hyperactivity disorder. Brain Stimul. 2015;8:299–304.
Loo SK, Salgari GC, Ellis A, Cowen J, Dillon A, McGough JJ. Trigeminal nerve stimulation for attention-deficit/hyperactivity disorder: cognitive and electrophysiological predictors of treatment response. J Am Acad Child Adolesc Psychiatry. 2020. http://ovidsp.ovid.com/ovidweb.cgi?T=JS&PAGE=reference&D=medp&NEWS=N&AN=33068751.
Generoso MB, Taiar IT, Garrocini LP, Bernardon R, Cordeiro Q, Uchida RR, et al. Effect of a 10-day transcutaneous trigeminal nerve stimulation (TNS) protocol for depression amelioration: A randomized, double blind, and sham-controlled phase II clinical trial. Epilepsy Behav. 2019;95:39–42.
Shiozawa P, da Silva ME, Netto GTM, Taiar I, Cordeiro Q. Effect of a 10-day trigeminal nerve stimulation (TNS) protocol for treating major depressive disorder: A phase II, sham-controlled, randomized clinical trial. Epilepsy Behav. 2015;44:23–6.
Shiozawa P, Duailibi MS, da Silva ME, Cordeiro Q. Trigeminal nerve stimulation (TNS) protocol for treating major depression: an open-label proof-of-concept trial. Epilepsy Behav EB. 2014;39:6–9.
Trevizol AP, Shiozawa P, Cook IA, Sato IA, Dos Santos Guimarães FB, Diniz BS, et al. Trigeminal nerve stimulation (TNS) for major depressive disorder in the elderly: an open label proof-of-concept trial. Brain Stimul. 2016;9:146–7.
Cook IA, Schrader LM, DeGiorgio CM, Miller PR, Maremont ER, Leuchter AF. Trigeminal nerve stimulation in major depressive disorder: Acute outcomes in an open pilot study. Epilepsy Behav. 2013;28:221–6.
Schrader LM, Cook IA, Miller PR, Maremont ER, DeGiorgio CM. Trigeminal nerve stimulation in major depressive disorder: first proof of concept in an open pilot trial. Epilepsy Behav EB. 2011;22:475–8.
DeGiorgio CM, Soss J, Cook IA, Markovic D, Gornbein J, Murray D, et al. Randomized controlled trial of trigeminal nerve stimulation for drug-resistant epilepsy. Neurology. 2013;80:786–91.
Soss J, Heck C, Murray D, Markovic D, Oviedo S, Corrale-Leyva G, et al. A prospective long-term study of external trigeminal nerve stimulation for drug-resistant epilepsy. Epilepsy Behav. 2015;42:44–7.
Gil-López F, Boget T, Manzanares I, Donaire A, Conde-Blanco E, Baillés E, et al. External trigeminal nerve stimulation for drug resistant epilepsy: a randomized controlled trial. Brain Stimul. 2020;13:1245–53.
Zhang C, Mao A, Jin P, Tao W, Shen J, Cai X, et al. Efficacy of transcutaneous trigeminal nerve stimulation on drug-resistant epilepsy and depression. J Clin Neurol. 2018;31:165–8.
DeGiorgio CM, Shewmon DA, Whitehurst T. Trigeminal nerve stimulation for epilepsy. Neurology. 2003;61:421–2.
DeGiorgio CM, Shewmon A, Murray D, Whitehurst T. Pilot study of trigeminal nerve stimulation (TNS) for epilepsy: a proof-of-concept trial. Epilepsia. 2006;47:1213–5.
DeGiorgio CM, Murray D, Markovic D, Whitehurst T. Trigeminal nerve stimulation for Epilepsy: longt-term feasibility and efficacy. Neurology. 2009;72:936–8.
Pop J, Murray D, Markovic D, DeGiorgio CM. Acute and long-term safety of external trigeminal nerve stimulation for drug-resistant epilepsy. Epilepsy Behav. 2011;22:574–6.
Slaght SJ, Nashef L. An audit of external trigeminal nerve stimulation (eTNS) in epilepsy. Seizure. 2017;52:60–2.
Olivié L, Giraldez BG, Sierra-Marcos A, Díaz-Gómez E, Serratosa JM. External trigeminal nerve stimulation: a long term follow up study. Seizure. 2019;69:218–20.
Bisla S, Gupta A, Agarwal S, Singh H, Sehrawat A, Singh A. Evaluation of transcutaneous electrical nerve stimulation as an adjunct therapy in trigeminal neuralgia - a randomized double-blind placebo-controlled clinical study. J Dent Anesth Pain Med. 2021;21:565–74.
Yameen F, Shahbaz NN, Hasan Y, Fauz R, Abdullah M. Efficacy of transcutaneous electrical nerve stimulation and its different modes in patients with trigeminal neuralgia. JPMA J Pak Med Assoc. 2011;61:437–9.
Schoenen JE. Migraine prevention with a supraorbital transcutaneous stimulator: a randomized controlled trial. Neurology. 2016;86:201–2.
An Z, Yin Y, Zhao L, Zhao T, Gu J, Li M, et al. Efficacy and safety of flunarizine combined with transcutaneous electrical nerve stimulation in the treatment of vestibular migraine. Clin Misdiagnosis Mistreatment. 2020;33:57–61.
Gao J. To observe the effect of nimodipine combined with transcutaneous electrical nerve stimulation in the treatment of vestibular migraine. Diet Health. 2020;44:76.
Deng Y, Zheng M, He L, Yang J, Yu G, Wang J. A head-to-head comparison of percutaneous mastoid electrical stimulator and supraorbital transcutaneous stimulator in the prevention of migraine: a prospective, randomized controlled study. Neuromodulation Technol Neural Interface. 2020;23:770–7.
Hamed NS. Supraorbital electrical stimulation in management of chronic type tension headache: a randomized controlled study. Physiother Theory Pr. 2018;34:101–10.
Wang L, Jin J, Liu Y. Clinical study of transcutaneous electrical nerve stimulation combined with rizatriptan benzoate in the treatment of migraine. Chin J Pain Med. 2017;23:353–5.
Guo P. Clinical effect of transcutaneous electrical nerve stimulation combined with nimesulide capsules in the treatment of migraine. Clin Med Res Pr. 2018;3:27–8.
Fan X, Zhao Y. Transcutaneous low-frequency electrical nerve stimulation therapy and clinical application of migraine headache. Chin Med Clin. 2019;19:1684–5.
Zhao Y. Clinical analysis of 58 cases of migraine treated with transcutaneous electrical nerve stimulator Cefaly. World Latest Med Inf Dig. 2018;18;294–5.
Chen Y, Xing S, Xin P, Ning W, Chenglong R, Xizheng S. Clinical observation of electrical nerve stimulation in patients with vestibular migraine. Chin J Hear Speech Rehabil Sci. 2018;16:259–61.
Przeklasa-Muszyńska A, Skrzypiec K, Kocot-Kępska M, Dobrogowski J, Wiatr M, Mika J. Non-invasive transcutaneous Supraorbital Neurostimulation (tSNS) using Cefaly ® device in prevention of primary headaches. Neurol Neurochir Pol. 2017;51:127–34.
Gao W, Hu Y, Wang HP. A randomized controlled study on the treatment of chronic headache by transcutaneous low frequency electrical stimulation. Int J Psychiatry Neurol. 2019;8;19–23
Kuruvilla D, Mann JI, Tepper SJ, Starling AJ, Panza G, Johnson MAL. Phase 3 randomized, double-blind, sham-controlled trial of e-TNS for the acute treatment of migraine (TEAM). Sci Rep. 2022;12:5110.
Alon G. Proceedings #6: combined trigeminal & occipital nerve stimulation (CTO-NS) compared to trigeminal stimulation alone for treatment of migraine headache. Brain Stimul. 2019;12:e63–5.
Gao S. Analysis of the effect of percutaneous supraorbital nerve stimulation in patients with migraine. Health Needs. 2020;8:208.
Danno D, Iigaya M, Imai N, Igarashi H, Takeshima T. The safety and preventive effects of a supraorbital transcutaneous stimulator in Japanese migraine patients. Sci Rep. 2019;9:9900.
Russo A, Tessitore A, Conte F, Marcuccio L, Giordano A, Tedeschi G. Transcutaneous supraorbital neurostimulation in “de novo” patients with migraine without aura: the first Italian experience. J Headache Pain. 2015;16:69.
Russo A, Tessitore A, Esposito F, Di Nardo F, Silvestro M, Trojsi F, et al. Functional changes of the perigenual part of the anterior cingulate cortex after external trigeminal neurostimulation in migraine patients. Front Neurol. 2017;8:282.
Birlea M, Penning S, Callahan K, Schoenen J. Efficacy and safety of external trigeminal neurostimulation in the prevention of chronic migraine: an open-label trial. Cephalalgia Rep. 2019;2:251581631985662.
Ordás CM, Cuadrado ML, Pareja JA, de-las-Casas-Cámara G, Gómez-Vicente L, Torres-Gaona G, et al. Transcutaneous supraorbital stimulation as a preventive treatment for chronic migraine: a prospective, open-label study. Pain Med. 2020;21:415–22.
Vikelis M, Dermitzakis EV, Spingos KC, Vasiliadis GG, Vlachos GS, Kararizou E. Clinical experience with transcutaneous supraorbital nerve stimulation in patients with refractory migraine or with migraine and intolerance to topiramate: a prospective exploratory clinical study. BMC Neurol. 2017;17:97.
Chou DE, Gross GJ, Casadei CH, Yugrakh MS. External trigeminal nerve stimulation for the acute treatment of migraine: open-label trial on safety and efficacy. Neuromodulation Technol Neural Interface. 2017;20:678–83.
Beh SC. External trigeminal nerve stimulation: potential rescue treatment for acute vestibular migraine. J Neurol Sci. 2020;408:116550.
Kuruvilla D, Mann JI, Schoenen J, Penning S. Acute treatment of migraine with external trigeminal nerve stimulation: a pilot trial. Cephalalgia Rep. 2019;2:251581631982990.
Di Fiore P, Galli A, D’Arrigo G, Bussone G, Didier H, D’Amico D, et al. Transcutaneous supraorbital neurostimulation for acute treatment of chronic migraine: open-label preliminary data. Neurol Sci. 2018;39:163–4.
Di Fiore P, Bussone G, Galli A, Didier H, Peccarisi C, D’Amico D, et al. Transcutaneous supraorbital neurostimulation for the prevention of chronic migraine: a prospective, open-label preliminary trial. Neurol Sci. 2017;38:201–6.
Um YH, Wang SM, Kang DW, Kim NY, Lim HK. Impact of transdermal trigeminal electrical neuromodulation on subjective and objective sleep parameters in patients with insomnia: a pilot study. Sleep Breath. 2021. https://link.springer.com/10.1007/s11325-021-02459-0.
Raghuveer R, Marbate S, Ruchi. Effect of cervical mobilization and transcutaneous supraorbital nerve stimulation in migraine without aura. Int J Res Pharm Sci. 2021;12:2316–24.
Tao H, Wang T, Dong X, Guo Q, Xu H, Wan Q. Effectiveness of transcutaneous electrical nerve stimulation for the treatment of migraine: a meta-analysis of randomized controlled trials. J Headache Pain. 2018;19:42.
Caous CA, de Sousa Buck H, Lindsey CJ. Neuronal connections of the paratrigeminal nucleus: a topographic analysis of neurons projecting to bulbar, pontine and thalamic nuclei related to cardiovascular, respiratory and sensory functions. Auton Neurosci. 2001;94:14–24.
de Sousa Buck H, Caous CA, Lindsey CJ. Projections of the paratrigeminal nucleus to the ambiguus, rostroventrolateral and lateral reticular nuclei, and the solitary tract. Auton Neurosci. 2001;87:187–200.
Gura EV, Garkavenko VV, Limansky Y. Influences of central gray matter stimulation on thalamic neuron responses to high- and low-threshold stimulation of trigeminal nerve structures. Neuroscience. 1991;41:681–93.
Farmer AD, Strzelczyk A, Finisguerra A, Gourine AV, Gharabaghi A, Hasan A, et al. International consensus based review and recommendations for minimum reporting standards in research on transcutaneous vagus nerve stimulation (version 2020). Front Hum Neurosci. 2020;14:568051.
Parkin BL, Ekhtiari H, Walsh VF. Non-invasive human brain stimulation in cognitive neuroscience: a primer. Neuron. 2015;87:932–45.
Gallop L, Westwood S, Lewis Y, Campbell I, Schmidt U. Effects of transcranial direct current stimulation in children and young people with psychiatric disorders: a systematic review. Eur Child Adolesc Psychiatry. https://doi.org/10.1101/2022.03.17.22272541.
Rubia K, Westwood S, Aggensteiner PM, Brandeis D. Neurotherapeutics for Attention Deficit/Hyperactivity Disorder (ADHD): A Review. Cells. 2021;10:2156.
Funding
AAC, SC, and KR were supported by the Efficacy and Mechanism Evaluation (EME) programme, an MRC and NIHR partnership (project ref: NIHR130077), SC and KR by the NIHR Programme Grant (NIHR203684), and SC by 101095568-HORIZON- HLTH-2022-DISEASE-07-03)(European Research Agency) and NIHR203684, NIHR203035, NIHR130077, NIHR128472, RP-PG-0618-20003 (National Institute for Health and Care Research. The design, management, analysis and reporting of the study are independent of the funders. KR is supported by the National Institute for Health Research (NIHR) and the UK Department of Health via the National Institute for Health Research (NIHR) Biomedical Research Centre (BRC) for Mental Health at South London and the Maudsley NHS Foundation Trust and Institute of Psychiatry, Psychology and Neuroscience, King’s College London. WT is supported by the China Scholarship Council (No.202006240034). The views expressed are those of the authors and not necessarily those of the funders.
Author information
Authors and Affiliations
Contributions
Conceptualization: SJW, AAC, WT, SC, and KR. Data curation: SJW, AAC, and WT. Formal analysis: SJW and AAC. Investigation: SJW, AAC, WT, and SX. Methodology: SJW, AAC, WT, and SC. Project administration: SJW, SC, and KR. Software: AAC. Supervision: SJW, SC, and KR. Validation: SJW, AAC, WT, SX, SC, and KR. Visualization: SJW. Writing - original draft: SJW and AAC. Writing - review & editing: SJW, AAC, WT, S.X, SC, and KR.
Corresponding author
Ethics declarations
Competing interests
SJW, AAC, WT, and SX declare no conflicts of interest. KR has received a grant from Takeda pharmaceuticals for another project and consulting fees from Supernus and Lundbeck. SC declares honoraria and reimbursement for travel and accommodation expenses for lectures from the following non-profit associations: Association for Child and Adolescent Central Health (ACAMH), Canadian ADHD Alliance Resource (CADDRA), British Association of Pharmacology (BAP), and from Healthcare Convention for educational activity on ADHD.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Westwood, S.J., Conti, A.A., Tang, W. et al. Clinical and cognitive effects of external trigeminal nerve stimulation (eTNS) in neurological and psychiatric disorders: a systematic review and meta-analysis. Mol Psychiatry 28, 4025–4043 (2023). https://doi.org/10.1038/s41380-023-02227-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-023-02227-4
- Springer Nature Limited