Abstract
Prenatal immune-mediated events are known risk factors for neurodevelopmental disorders in the offspring (NDD). Although the brain continues to develop for years after birth and many postnatal factors alter the regular trajectory of neurodevelopment, little is known about the impact of postnatal immune factors. To fill this gap we set up ARTEMIS, a cohort of juvenile rheumatisms and systemic autoimmune and auto-inflammatory disorders (jRSAID), and assessed their neurodevelopment. We then complemented our results with a systematic review and meta-analysis. In ARTEMIS, we used unsupervised and supervised analysis to determine the influence of jRSAID age at onset (AO) and delay in introduction of disease-modifying therapy (DMT) on NDD (NCT04814862). For the meta-analysis, we searched MEDLINE, EMBASE, PsycINFO, Cochrane, and Web of Science up to April 2022 without any restrictions on language, or article type for studies investigating the co-occurence of jRSAID and NDD (PROSPERO- CRD42020150346). 195 patients were included in ARTEMIS. Classification tree isolated 3 groups of patients (i) A low-risk group (AO > 130 months (m)) with 5% of NDD (ii) A medium-risk group (AO < 130 m and DMT < 2 m) with 20% of NDD (iii) and a high-risk-group (AO < 130 m and DMT > 2 m) with almost half of NDD. For the meta-analysis, 18 studies encompassing a total of (i) 46,267 children with jRSAID; 213,930 children with NDD, and 6,213,778 children as controls were included. We found a positive association between jRSAID and NDD with an OR = 1.44 [95% CI 1.31; 1.57] p < 0.0001, [I2 = 66%, Tau2 = 0.0067, p < 0.01]. Several sensitivity analyses were performed without changing the results. Metaregression confirmed the importance of AO (p = 0.005). Our study supports the association between jRSAID and NDD. AO and DMT have pivotal roles in the risk of developing NDD. We plead for systematic screening of NDD in jRSAID to prevent the functional impact of NDD.




Similar content being viewed by others
References
Thapar A, Cooper M, Rutter M. Neurodevelopmental disorders. Lancet Psychiatry. 2017;4:339–46.
Diagnostic and statistical manual of mental disorders: DSM-5. 5th ed. Washington: American Psychiatric Association; 2013.
Georgiades S, Bishop SL, Frazier T. research in autism—introducing the concept of ‘chronogeneity’. J Child Psychol Psychiatry. 58. Longitudinal: Editorial Perspective; 2017. 634–6.
Lord C, Elsabbagh M, Baird G, Veenstra-Vanderweele J. Autism spectrum disorder. Lancet Lond Engl. 2018;392:508–20.
Robertson MM, Eapen V, Singer HS, Martino D, Scharf JM, Paschou P, et al. Gilles de la Tourette syndrome. Nat Rev Dis Prim. 2017;3:16097.
Stein DJ, Costa DLC, Lochner C, Miguel EC, Reddy YCJ, Shavitt RG, et al. Obsessive-compulsive disorder. Nat Rev Dis Prim. 2019;5:52.
Antoun S, Ellul P, Peyre H, Rosenzwajg M, Gressens P, Klatzmann D, et al. Fever during pregnancy as a risk factor for neurodevelopmental disorders: results from a systematic review and meta-analysis. Mol Autism. 2021;12:60.
Ellul P, Acquaviva E, Peyre H, Rosenzwajg M, Gressens P, Klatzmann D, et al. Parental autoimmune and autoinflammatory disorders as multiple risk factors for common neurodevelopmental disorders in offspring: a systematic review and meta-analysis. Transl Psychiatry. 2022;12:112.
Choi GB, Yim YS, Wong H, Kim S, Kim H, Kim SV, et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 2016;351:933–9.
Rudolph MD, Graham AM, Feczko E, Miranda-Dominguez O, Rasmussen JM, Nardos R, et al. Maternal IL-6 during pregnancy can be estimated from newborn brain connectivity and predicts future working memory in offspring. Nat Neurosci. 2018;21:765–72.
Pierrat V, Marchand-Martin L, Marret S, Arnaud C, Benhammou V, Cambonie G, et al. Neurodevelopmental outcomes at age 5 among children born preterm: EPIPAGE-2 cohort study. BMJ 2021;373:n741.
Delahaye-Duriez A, Dufour A, Bokobza C, Gressens P, Van Steenwinckel J. Targeting Microglial Disturbances to Protect the Brain From Neurodevelopmental Disorders Associated With Prematurity. J Neuropathol Exp Neurol. 2021;80:634–48.
Zengeler KE, Lukens JR. Innate immunity at the crossroads of healthy brain maturation and neurodevelopmental disorders. Nat Rev Immunol. 2021;21:454–68.
Green MJ, Watkeys OJ, Whitten T, Thomas C, Kariuki M, Dean K, et al. Increased incidence of childhood mental disorders following exposure to early life infection. Brain Behav Immun. 2021;97:376–82. S0889-1591(21)00299-3
Wang L, Wang F-S, Gershwin ME. Human autoimmune diseases: a comprehensive update. J Intern Med. 2015;278:369–95.
Martini A, Lovell DJ, Albani S, Brunner HI, Hyrich KL, Thompson SD, et al. Juvenile idiopathic arthritis. Nat Rev Dis Prim. 2022;8:5.
Ozen S, Bilginer Y. A clinical guide to autoinflammatory diseases: familial Mediterranean fever and next-of-kin. Nat Rev Rheumatol. 2014;10:135–47.
Kamphuis S, Silverman ED. Prevalence and burden of pediatric-onset systemic lupus erythematosus. Nat Rev Rheumatol. 2010;6:538–46.
Pappas D. ADHD Rating Scale-IV: Checklists, norms, and clinical interpretation. J Psychoeduc Assess. 2006;24:172–8.
Baron IS. Behavior rating inventory of executive function. Child Neuropsychol J Norm Abnorm Dev Child Adolesc. 2000;6:235–8.
Constantino JN, Davis SA, Todd RD, Schindler MK, Gross MM, Brophy SL, et al. Validation of a brief quantitative measure of autistic traits: comparison of the social responsiveness scale with the autism diagnostic interview-revised. J Autism Dev Disord. 2003;33:427–33.
Lenth R. emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.8.4-1. 2023. https://CRAN.R-project.org/package=emmeans.
Hothorn T, Zeileis A. partykit: a modular toolkit for recursive partytioning in R. J Mach Learn Res. 2015;16:3905-9. https://jmlr.org/papers/v16/hothorn15a.html.
Kuhn M. caret: Classification and Regression Training. R package version 6.0-93. 2022. https://CRAN.R-project.org/package=caret.
Vaissie P, Monge A, Husson F. Factoshiny: Perform Factorial Analysis from ‘FactoMineR’ with a Shiny Application. R package version 2.4. 2021. https://CRAN.R-project.org/package=Factoshiny.
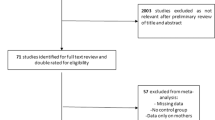
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100.
Eurofever Project - Home. https://www.printo.it/eurofever/index. Accessed 19 February 2021.
Autoimmune Disease List • AARDA. AARDA. 2016. https://www.aarda.org/diseaselist/. Accessed 19 February 2021.
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–5.
Cullen AE, Holmes S, Pollak TA, Blackman G, Joyce DW, Kempton MJ, et al. Associations Between Non-neurological Autoimmune Disorders and Psychosis: A Meta-analysis. Biol Psychiatry. 2019;85:35–48.
Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327:557–60.
Egger M, Smith GD, Phillips AN. Meta-analysis: principles and procedures. BMJ 1997;315:1533–7.
Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health. 2019;22:153–60.
Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane Handbook for Systematic Reviews of Interventions. 2nd ed. John Wiley & Sons; 2019.
Posner J, Polanczyk GV, Sonuga-Barke E. Attention-deficit hyperactivity disorder. Lancet Lond Engl. 2020;395:450–62.
Filiano AJ, Xu Y, Tustison NJ, Marsh RL, Baker W, Smirnov I, et al. Unexpected role of interferon-γ in regulating neuronal connectivity and social behaviour. Nature 2016;535:425–9.
Lord C, Brugha TS, Charman T, Cusack J, Dumas G, Frazier T, et al. Autism spectrum disorder. Nat Rev Dis Prim. 2020;6:5.
Akmatov MK, Ermakova T, Bätzing J. Psychiatric and nonpsychiatric comorbidities among children with ADHD: An exploratory analysis of nationwide claims data in Germany. J Atten Disord. 2021;25:874–84.
Alabaf S, Gillberg C, Lundström S, Lichtenstein P, Kerekes N, Råstam M, et al. Physical health in children with neurodevelopmental disorders. J Autism Dev Disord. 2019;49:83–95.
Butwicka A, Lichtenstein P, Frisén L, Almqvist C, Larsson H, Ludvigsson JF. Celiac disease is associated with childhood psychiatric disorders: a population-based study. J Pediatr. 2017;184:87–93.e1.
Butwicka A, Olén O, Larsson H, Halfvarson J, Almqvist C, Lichtenstein P, et al. Association of Childhood-Onset Inflammatory Bowel Disease With Risk of Psychiatric Disorders and Suicide Attempt. JAMA Pediatr. 2019. 19 August 2019. https://doi.org/10.1001/jamapediatrics.2019.2662.
Capucilli P, Cianferoni A, Grundmeier RW, Spergel JM. Comparison of comorbid diagnoses in children with and without eosinophilic esophagitis in a large population. Ann Allergy Asthma Immunol Publ Am Coll Allergy Asthma Immunol. 2018;121:711–6.
Kapellen TM, Reimann R, Kiess W, Kostev K. Prevalence of medically treated children with ADHD and type 1 diabetes in Germany - Analysis of two representative databases. J Pediatr Endocrinol Metab JPEM. 2016;29:1293–7.
Lebwohl B, Haggård L, Emilsson L, Söderling J, Roelstraete B, Butwicka A, et al. Psychiatric disorders in patients with a diagnosis of celiac disease during childhood from 1973 to 2016. Clin Gastroenterol Hepatol Clin Pr J Am Gastroenterol Assoc. 2021;19:2093–2101.e13.
Lee M, Krishnamurthy J, Susi A, Sullivan C, Gorman GH, Hisle-Gorman E, et al. Association of autism spectrum disorders and inflammatory bowel disease. J Autism Dev Disord. 2018;48:1523–9.
Lin C-H, Lin W-D, Chou I-C, Lee I-C, Hong S-Y. Heterogeneous neurodevelopmental disorders in children with Kawasaki disease: what is new today? BMC Pediatr. 2019;19:406.
Nielsen PR, Benros ME, Dalsgaard S. Associations between autoimmune diseases and attention-deficit/hyperactivity disorder: a nationwide study. J Am Acad Child Adolesc Psychiatry. 2017;56:234–240.e1.
Robinson C, Lao F, Chanchlani R, Gayowsky A, Darling E, Batthish M. Long-term hearing and neurodevelopmental outcomes following Kawasaki disease: A population-based cohort study. Brain Dev. 2021;43:735–44.
Heifert TA, Susi A, Hisle-Gorman E, Erdie-Lalena CR, Gorman G, Min SB, et al. Feeding disorders in children with autism spectrum disorders are associated with eosinophilic esophagitis. J Pediatr Gastroenterol Nutr. 2016;63:e69–73.
Mercadante MT, Busatto GF, Lombroso PJ, Prado L, Rosário-Campos MC, do Valle R, et al. The psychiatric symptoms of rheumatic fever. Am J Psychiatry. 2000;157:2036–8.
Ucuz I, Altunisik N, Sener S, Turkmen D, Kavuran NA, Marsak M, et al. Quality of life, emotion dysregulation, attention deficit and psychiatric comorbidity in children and adolescents with vitiligo. Clin Exp Dermatol. 2021;46:510–5.
Zelnik N, Pacht A, Obeid R, Lerner A. Range of neurologic disorders in patients with celiac disease. Pediatrics 2004;113:1672–6.
Zerbo O, Leong A, Barcellos L, Bernal P, Fireman B, Croen LA. Immune mediated conditions in autism spectrum disorders. Brain Behav Immun. 2015;46:232–6.
Güngör S, Celiloğlu OS, Ozcan OO, Raif SG, Selimoğlu MA. Frequency of celiac disease in attention-deficit/hyperactivity disorder. J Pediatr Gastroenterol Nutr. 2013;56:211–4.
Ho H-Y, Wong C-K, Wu S-Y, Hsiao RC, Chen Y-L, Yen C-F. Increased Alopecia Areata risk in children with attention-deficit/hyperactivity disorder and the impact of methylphenidate use: a nationwide population-based cohort study. Int J Environ Res Public Health. 2021;18:1286.
Granjon M, Rohmer O, Doignon-Camus N, Popa-Roch M, Pietrement C, Gavens N. Neuropsychological functioning and academic abilities in patients with juvenile idiopathic arthritis. Pediatr Rheumatol Online J. 2021;19:53.
von Weiss RT, Rapoff MA, Varni JW, Lindsley CB, Olson NY, Madson KL, et al. Daily hassles and social support as predictors of adjustment in children with pediatric rheumatic disease. J Pediatr Psychol. 2002;27:155–65.
Daltroy LH, Larson MG, Eaton HM, Partridge AJ, Pless IB, Rogers MP, et al. Psychosocial adjustment in juvenile arthritis. J Pediatr Psychol. 1992;17:277–89.
Özer S, Bozkurt H, Yılmaz R, Sönmezgöz E, Bütün I. Evaluation of executive functions in children and adolescents with familial Mediterranean fever. Child Neuropsychol J Norm Abnorm Dev Child Adolesc. 2017;23:332–42.
Borsini A, Zunszain PA, Thuret S, Pariante CM. The role of inflammatory cytokines as key modulators of neurogenesis. Trends Neurosci. 2015;38:145–57.
Salvador AF, de Lima KA, Kipnis J. Neuromodulation by the immune system: a focus on cytokines. Nat Rev Immunol. 2021;21:526–41.
Ross G, Sammaritano L, Nass R, Lockshin M. Effects of mothers’ autoimmune disease during pregnancy on learning disabilities and hand preference in their children. Arch Pediatr Adolesc Med. 2003;157:397–402.
Alves de Lima K, Rustenhoven J, Da Mesquita S, Wall M, Salvador AF, Smirnov I, et al. Meningeal γδ T cells regulate anxiety-like behavior via IL-17a signaling in neurons. Nat Immunol. 2020;21:1421–9.
Ribeiro M, Brigas HC, Temido-Ferreira M, Pousinha PA, Regen T, Santa C, et al. Meningeal γδ T cell-derived IL-17 controls synaptic plasticity and short-term memory. Sci Immunol. 2019;4:eaay5199.
Krol A, Feng G. Windows of opportunity: timing in neurodevelopmental disorders. Curr Opin Neurobiol. 2018;48:59–63.
Faust TE, Gunner G, Schafer DP. Mechanisms governing activity-dependent synaptic pruning in the developing mammalian CNS. Nat Rev Neurosci. 2021;22:657–73.
Kaul D, Habbel P, Derkow K, Krüger C, Franzoni E, Wulczyn FG, et al. Expression of Toll-like receptors in the developing brain. PloS One. 2012;7:e37767.
Hung Y-Y. Antidepressants improve negative regulation of toll-like receptor signaling in monocytes from patients with major depression. Neuroimmunomodulation 2018;25:42–48.
Missig G, Robbins JO, Mokler EL, McCullough KM, Bilbo SD, McDougle CJ, et al. Sex-dependent neurobiological features of prenatal immune activation via TLR7. Mol Psychiatry. 2020;25:2330–41.
Park SJ, Lee JY, Kim SJ, Choi S-Y, Yune TY, Ryu JH. Toll-like receptor-2 deficiency induces schizophrenia-like behaviors in mice. Sci Rep. 2015;5:8502.
Okun E, Griffioen KJ, Mattson MP. Toll-like receptor signaling in neural plasticity and disease. Trends Neurosci. 2011;34:269–81.
Chen R, Davis LK, Guter S, Wei Q, Jacob S, Potter MH, et al. Leveraging blood serotonin as an endophenotype to identify de novo and rare variants involved in autism. Mol Autism. 2017;8:14.
Meredith RM, Dawitz J, Kramvis I. Sensitive time-windows for susceptibility in neurodevelopmental disorders. Trends Neurosci. 2012;35:335–44.
Millan MJ, Andrieux A, Bartzokis G, Cadenhead K, Dazzan P, Fusar-Poli P, et al. Altering the course of schizophrenia: progress and perspectives. Nat Rev Drug Discov. 2016;15:485–515.
Fitzpatrick Z, Frazer G, Ferro A, Clare S, Bouladoux N, Ferdinand J, et al. Gut-educated IgA plasma cells defend the meningeal venous sinuses. Nature 2020;587:472–6.
Liston A, Dooley J, Yshii L. Brain-resident regulatory T cells and their role in health and disease. Immunol Lett. 2022;248:26–30. S0165-2478(22)00092-X
Meltzer A, Van de Water J. The role of the immune system in autism spectrum disorder. Neuropsychopharmacol Publ Am Coll Neuropsychopharmacol. 2017;42:284–98.
Bennabi M, Gaman A, Delorme R, Boukouaci W, Manier C, Scheid I, et al. HLA-class II haplotypes and Autism spectrum disorders. Sci Rep. 2018;8:7639.
Cross-Disorder Group of the Psychiatric Genomics Consortium. Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet Lond Engl. 2013;381:1371–9.
Schanberg LE, Lefebvre JC, Keefe FJ, Kredich DW, Gil KM. Pain coping and the pain experience in children with juvenile chronic arthritis. Pain 1997;73:181–9.
La Hausse de Lalouvière L, Ioannou Y, Fitzgerald M. Neural mechanisms underlying the pain of juvenile idiopathic arthritis. Nat Rev Rheumatol. 2014;10:205–11.
Acknowledgements
The authors warmly thank Camille Aupiais and Charlène Da Silveira for their precious help in the inclusion of the patients.
Funding
The authors received funding from DM’UP (Robert Debré).
Author information
Authors and Affiliations
Contributions
PE and IM: have designed the ARTEMIS study, did the statistics, and wrote the first draft of the article. IM, FA, BBM, AB, GD, CD, AF, MLF, UM, PQ, IS, CV: inclusion of ARTEMIS patients. LL: extracted the data of the patients included in ARTEMIS and participated in the first statistical analyses. NT: Supervision of ARTEMIS statistical analysis. PE and SA: Searched the studies for the meta-analysis, extracted the data, and did the statistics. HP: helped for meta-analysis statistics. RD and DK: Overall supervision and guidance of the whole article. All authors contributed significantly to the proofreading and improvement of the first draft. All authors confirm that they had full access to all the data and accept responsibility to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ellul, P., Melki, I., Antoun, S. et al. Early systemic inflammation induces neurodevelopmental disorders: results from ARTEMIS, a French multicenter study of juvenile rheumatisms and systemic autoimmune and auto-inflammatory disorders and meta-analysis. Mol Psychiatry 28, 1516–1526 (2023). https://doi.org/10.1038/s41380-023-01980-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-023-01980-w
- Springer Nature Limited