Abstract
Asparaginase is an essential component of acute lymphoblastic leukemia (ALL) therapy, yet its associated toxicities often lead to treatment discontinuation, increasing the risk of relapse. Hypersensitivity reactions include clinical allergies, silent inactivation, or allergy-like responses. We hypothesized that even moderate increases in asparaginase clearance are related to later inactivation. We therefore explored mandatory monitoring of asparaginase enzyme activity (AEA) in patients with ALL aged 1–45 years treated according to the ALLTogether pilot protocol in the Nordic and Baltic countries to relate mean AEA to inactivation, to build a pharmacokinetic model to better characterize the pharmacokinetics of peg-asparaginase and assess whether an increased clearance relates to subsequent inactivation. The study analyzed 1631 real-time AEA samples from 253 patients, identifying inactivation in 18.2% of the patients. This inactivation presented as mild allergy (28.3%), severe allergy (50.0%), or silent inactivation (21.7%). A pharmacokinetic transit compartment model was used to describe AEA-time profiles, revealing that 93% of patients with inactivation exhibited prior increased clearance, whereas 86% of patients without hypersensitivity maintained stable clearance throughout asparaginase treatment. These findings enable prediction of inactivation and options for either dose increments or a shift to alternative asparaginase formulations to optimize ALL treatment strategies.
Similar content being viewed by others
Introduction
Acute lymphoblastic leukemia (ALL) is the most common childhood cancer. Asparaginase is a cornerstone in the treatment of ALL contributing to the high survival rate [1]. Debates persist concerning the most effective treatment regimen in terms of the quantity of doses, dosage, and timing of asparaginase extending to the broader treatment strategy [2]. Within the ALLTogether treatment protocol, the dosing and number of doses vary based on age and risk groups (EudraCT no.2018-001795-38). Escherichia coli L-asparaginase can be covalently linked to polyethylene glycol (peg-asparaginase) in order to decrease immunogenicity of the enzyme and to prolong its half-life [3]. However, the most common cause of truncation of peg-asparaginase treatment is still hypersensitivity (8–15%) [1, 4,5,6], and truncation of asparaginase treatment has been shown to increase the risk of relapse [1, 7,8,9,10]. Asparaginase hypersensitivity is defined as; clinical allergy ranging from a mild rash to anaphylaxis and silent inactivation (SI) with absence of clinical symptoms. Both conditions are associated with inactivation of asparaginase enzyme activity (AEA) and switching treatment to another asparaginase formulation is indicated [7, 11, 12] Additional allergy-like reactions is a type of asparaginase hypersensitivity with more varying symptoms mimicking true allergies, but - importantly - the AEA is unaffected and peg-asparaginase treatment may proceed if the symptoms are not too pronounced [3, 13].
Real-time therapeutic drug monitoring (TDM) of AEA is an important tool to ensure optimal treatment and to identify patients with inactivation of the drug providing a possibility to switch asparaginase preparation[3, 4, 14]. Additionally, TDM makes it possible to distinguish true allergies with inactivation from allergy-like reactions[3, 4, 6, 13, 15].
AEA trough concentration (Ctrough) ≥ 100 IU/L two weeks after an administration of peg-asparaginase has been defined as the therapeutic activity target level to ensure complete asparagine depletion[6, 11, 13, 16]. Previous studies showed that the number of peg-asparaginase doses could be reduced while maintaining high survival rates and reducing toxicity significantly[8, 17,18,19], resulting in less asparaginase treatment in many contemporary protocols compared with previous protocols. Consequently, it is crucial that all doses of peg-asparaginase are effective.
The inactivation of asparaginase has been a long-standing subject of interest, and numerous explanations have been proposed [20]. It has been hypothesized that asparaginase inactivation follows a classical immune response with initial IgM formation followed by affinity maturation and isotype switching to the IgG and IgE subclasses [21, 22] which could indicate that early changes in clearance could be detected. The detection of inactivation-based changes in clearance using AEA is obscured by drug accumulation, intra-patient variability and increased intra-dose clearance due to depegylation. Hence, the application of pharmacokinetic analysis becomes necessary to delineate these elements [23]. Wurthwein et al. [24] characterized the standard elimination in peg-asparaginase AEA-time profiles, however, the study design only included two consecutive doses and samples indication inactivation were excluded. Changes in clearance over the treatment period due to an inactivation response was not part of the scope of the study.
Prediction of inactivation from AEA levels at an early stage would provide an opportunity to decrease the risk of potentially life-threatening allergic reactions and to switch to another asparaginase preparation at the optimal time to ensure effective treatment. Furthermore, prediction of AEA-levels might be useful to decrease the peg-asparaginase dose in patients with expected high AEA, which may be associated with an increased risk of certain toxicities (pancreatitis and avascular necrosis) [25].
The overall aim of this study was to assess whether AEA can be used to identify patients with future peg-asparaginase inactivation. To achieve this goal, peg-asparaginase enzyme activity was monitored in patients included in the ALLTogether pilot protocol, the relationship between measured AEA were related to timing of the dose and the probability of an inactivation event and a pharmacokinetic analysis was conducted to characterize the trend and variability in AEA-time profiles and to verify whether a change or an increase in clearance are associated with the development of an inactivation response.
Methods
Study population
The included patients were aged 1–45 years and diagnosed with de novo Philadelphia chromosome negative B-cell precursor or T-cell ALL. Patients were treated according to the ALLTogether pilot protocol conducted in Denmark, Iceland, Lithuania, Norway and Sweden from November 2018 to February 2022. Patient and disease characteristics, treatment and toxicity information were retrieved from the ALLTogether study database (Castor) and by contact with the treating centers.
Risk-stratification and treatment
In the ALLTogether pilot protocol, patients were stratified into four main risk groups; standard-risk (SR), intermediate-risk low/high (IRL/IRH), or high-risk (HR) based on clinical features; age, immunophenotype (B-cell and T-cell), cytogenetics and treatment response, by evaluation of measurable residual disease at day 29 and day 71/78 (Supplementary 1).
Peg-asparaginase was used as the standard preparation. The dose was adjusted according to age (1500 IE/m2 < 16 years and 1000 IE/m2 ≥ 16 years) and was administered intravenously (IV) or intramuscularly (IM, mostly adults) over one hour. Administration of peg-asparaginase started from treatment day 4 during the induction phase for all patients. However, following amendment of September 9th, 2020, ALLTogether pilot protocol version 2.2, patients aged 25 years and older (n = 2) received their first dose of peg-asparaginase on day 18 of the treatment protocol. Peg-asparaginase was administered with two-week intervals to maintain continuous depletion of asparagine for a duration of 8–20 weeks which varied based on the prescribed number of doses according to risk group (SR 4 doses, IRL 5 doses, IRH 8 doses and HR a maximum of 10 doses) (Supplementary Table S2, Supplementary 1). Premedication was not recommended.
The ALLTogether pilot protocol was approved by the National Medicines Agencies (EudraCT no.2018-001795-38) and national or regional ethics committees in each participating country. The study was registered at ClinicalTrials.gov (NCT03911128). All patients and/or caregivers gave consent according to the Declaration of Helsinki.
AEA sampling and TDM
Sampling for AEA measurements on the ALLTogether pilot protocol was planned as follows; at baseline, on day 1, 4, 7, 11 and 14 after the first dose of peg-asparaginase and 14 days after the subsequent doses. Ctrough was defined as the AEA level 14 ± 2 days after a peg-asparaginase administration [6, 16, 26]. Lower level of detection (LLOD) was 0.5 IU/L and inter-day variation 2.5-6%. In case of >1 sample in the defined timeslot (day 12–16 after previous dose) the earliest sample in the timeslot was selected for statistical analyses. All samples were included in the pharmacokinetic analyses.
Analysis of AEA, using a validated L-aspartic β-hydroxamate assay (AHA) [27], was centralized for all countries in the Nordic Society of Paediatric Haematology and Oncology (NOPHO) at the Asparaginase laboratory at Aarhus University Hospital. AEA levels were measured in real-time and recommendations about continuation, switch or additional sampling were provided to the responsible clinicians through an online database (REDCap electronic data capture tools hosted at Aarhus University) [28, 29]. In REDCap all patients were registered with a unique record-ID with an event for all AEA samples. The clinicians provided information about peg-asparaginase administration date and potential toxicities regarding a specific dose and sample. Missing information was retrieved by direct contact with the treatment sites. The total turnaround time was between 4 and 7 days.
Inactivation
Clinical allergy was graded as mild or severe according to the Consensus Definitions by the Ponte Di Legno Working Group [4, 11]. Silent inactivation was defined as Ctrough AEA < LLQ 14 ± 2 days after a peg-asparaginase administration. Allergy-like reactions were defined as varying symptoms of intolerance e.g., vomiting, abdominal pain and/or rash but with Ctrough > 100 IU/L if infusion was completed [11]. In case of an allergic reaction where the infusion was stopped, the amount of administered peg-asparaginase was registered and extra sampling immediately after interruption of the infusion and within a few days were recommended to verify inactivation. If over half the dose was given, an extra day-seven sample was recommended after an allergic reaction. If AEA on day seven exceeded 100 IU/L, another peg-asparaginase dose would be proposed. In case of inactivation, the recommendations were to replace the remaining doses with native Erwinia chrysanthemi-derived asparaginase (20,000 IU/m2/dose) administered every second day, which means that one dose of peg-asparaginase was substituted with seven doses of Erwinia-derived asparaginase.
Statistical analyses
A linear mixed model was used to evaluate the relationship between mean AEA Ctrough levels and dose number with dose number entered as a categorical variable. A random effect (intercept) per patient was included to model the dependency between measurements from the same patient. Results were reported by no inactivation versus inactivation and stratified by route of administration (IM and IV administration), age (</≥16 years) and separated for time of allergic reaction.
A logistic regression model was used to model the relationship between AEA levels at day seven and inactivation after the first peg-asparaginase dose with AEA levels entered via a restricted cubic spline with four knots. This enabled us to evaluate the predictive performance of AEA levels at day seven on the risk of asparaginase inactivation.
Pharmacokinetic model
A population pharmacokinetic model was developed based on all the available AEA measurements, using the nonlinear mixed effects software NONMEM version 7.4.4 (ICON Development Solutions) [24]. The pharmacokinetics of peg-asparaginase are characterized by an increased clearance within the dosing interval as a consequence of depegylation, in addition to an increased clearance over the treatment period in some subjects due to development of inactivation. To capture these patterns, several structural models were assessed to describe the quantified data (Supplementary data 2. Structured models) [24], simultaneously accounting for readings below the lower level of quantification (LLQ) (≤5 IU/L) [30]. The model estimated the probability that patients belonged to one of two subpopulations; i.e. I) constant clearance or II) increased clearance over time. In a posthoc step, each patient was assigned to the group with their highest probability. The sensitivity and specificity of the model to identify increased clearance in patients that developed an inactivation was assessed. Inter-individual variability was evaluated on all structural parameters. To account for different body sizes, pharmacokinetic parameters were adjusted to body weight using allometric scaling [31]. The impact of age was additionally assessed on the clearance parameters.
Results
Patient characteristics
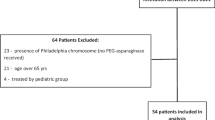
A total of 320 patients were included in the ALLTogether pilot protocol during the study period. Following the exclusion of patients with outlier AEA (>16 days after previous dose) and/or insufficient sampling (\(\le\)1 sample), 253 patients (79%) with 1631 AEA measurements (mean 6.5 samples per patient) were included in the statistical analyses, out of these, 86.6% (n = 219) were children 1–17 years and 13.4% (n = 34) young adults 18–45 years (Fig. 1).
The median age at diagnosis was 5.9 years and 61.3% of the patients were male. Patients were stratified to the following risk groups; SR 35.6%, IRL 24.9%, IRH 31.6% and HR 7.1%. The majority of patients (229/253, 90.5%) were treated with IV peg-asparaginase. IM administration was used in 3/219 children (1.4%) and 22/34 (61.1%) adults. Baseline characteristics of the patients are described in Table 1. Dosing of peg-asparaginase was 1500 IE/m2 for 80.5% of patients (<16 years) and 1000 IE/m2 for 19.5% of patients (≥16 years).
Sampling days 1–16 after the first dose and AEA Ctrough (12–16 days) after doses 2–7 were included in the basic statistical evaluation. Samples from day 7 after the first dose were used in the logistic regression model. AEA Ctrough after dose 8 were too few to be included.
Asparaginase enzyme activity and inactivation
Of 253 patients, 46 (18.2%) experienced hypersensitivity with inactivation during peg-asparaginase treatment; 36/46 patients (78.3%) developed clinical allergy, of those 23/36 patients (63.9%) had severe allergy and 13/36 patients (36.1%) had mild reactions. For all patients with IV administration and allergic symptoms, the reactions were observed during the infusion. None were fatal. Silent inactivation was found in 10/46 (21.7%). One patient had an allergy-like reaction. Two patients who experienced allergic reactions were excluded from the linear mixed model because samples were not collected within the designated timeframe following any dose. Both patients remained in the pharmacokinetic model with AEA measurements still within 14 days but outside the desired range of 14 +/− 2 days (Ctrough).
Clinical allergy occurred during administration of the third (n = 11/36, 30.6%), the fourth (n = 19/36, 52.8%) or the fifth (n = 6/36, 16.7%) dose of peg-asparaginase. For 23 of 36 (63.9%) of the patients with clinical allergy, AEA Ctrough were available reflecting the AEA level after the previous dose given. In all cases Ctrough was <LLQ. For the remaining 13 patients (36.1%) with clinical allergy no AEA Ctrough before the allergy-triggering dose was available. However, in three of these cases, Ctrough at an earlier stage during treatment showed AEA Ctrough < LLQ. SI was seen as Ctrough < LLQ after the second dose in two cases (20%), after the third dose in six cases (60%) and after the fourth dose in two cases (20%).
Statistical analyses
The AEA Ctrough for patients with and without inactivation are illustrated in Table 2 and Fig. 2. The inactivation group consisted of 45 patients with AEA Ctrough in the defined time slot (12–16 days after previous dose); 42 patients were under 16 years treated with IV peg-asparaginase (93.3%), 2 patients ≥16 years treated with IV peg-asparaginase (4.4%) and one patient (<16 years) treated with IM peg-asparaginase (2.2%).
AEA measurements day 1,4,7 and 11 after first dose and AEA Ctrough after every dose of peg-asparaginase. Patients with inactivation are illustrated as the blue line (including one patient with IM administered PEG-asparaginase and three patients ≥16 years) and patients in the non-inactivation group as the red, black and green line according to route of administration and age group.
Mean AEA Ctrough after the first IV peg-asparaginase administration was 322 IU/L (95% Confidence Interval (CI): 303–340 IU/L) <16 years (all were IV treated), 168 IU/L ≥ 16 years in the non-inactivation group (all were IV treated) and 295 IU/L in the inactivation group (97.8% IV treated, 2.2% IM treated) (Table 2).
Mean AEA Ctrough after the second IV administration was 203 IU/L in patients <16 years with inactivation. In patients <16 years without inactivation, the mean AEA Ctrough was 413 IU/L and 189 IU/L in patients ≥16 years. In patients <16 years mean AEA Ctrough after the 2nd dose of peg-asparaginase (AEA 203 IU/L) was significantly lower for patients with inactivation than without inactivation (AEA 413 IU/L) (p < 0.001) (Table 2). Meaningful statistical analyses were not feasible due to the limited number of patients and samples in the group of individuals aged ≥16 years.
The overall mean AEA Ctrough across the first four doses in the non-inactivation group of IV patients was in range 322–516 IU/L for patients <16 years, and 168–211 IU/L in patients ≥16 years. Finally, mean AEA Ctrough was 179 IU/L in the group of patients treated with IM peg-asparaginase (mean age 27.1, (3–44.8)) (Fig. 2). Considering the timing of allergic reaction (dose 3, 4, or 5), the variation in AEA Ctrough following the second dose continues to be distinct across all three time points for the allergy (Fig. 3).
AEA measurements day 1,4,7 and 11 after first dose and AEA Ctrough after every dose of peg-asparaginase for patients <16 years (IV administration). Patients without inactivation are shown in red, those experiencing allergic reactions on the 3rd dose in blue, on the 4th dose in green, and on the 5th dose in black.
The logistic regression analysis (Fig. 4) examined the relationship between the level of AEA measured seven days after the initial dose and the likelihood of inactivation. When evaluating an AEA level of <200 IU/L seven days after the first dose of peg-asparaginase, the risk of inactivation was estimated to be 15% (95% CI: 5.4–25.8). The analysis did not find a statistically significant association between AEA levels at that specific time point and the risk of inactivation.
Pharmacokinetic model
A pharmacokinetic transit model with a chain of 10-compartments captured the AEA data the best (Supplementary 2). A first-order absorption rate constant was added to describe the IM data. Initial clearance (CLinitial) for each compartment represented peg-asparaginase clearance at baseline, whereas clearance induced (CLinduced) out of the last compartment explained the increased clearance between and over dosing occasions. Volume of distribution was estimated at 4.81 L/70 kg (i.e., 1.71 L/25 kg, 3.44 L/50 kg) and CLinitial at 0.20 L/day/70 kg (i.e., 0.09 L/day/25 kg, 0.16 L/day/50 kg).
The pharmacokinetic model classified 28% of patients to have increased clearance over time, whereas 72% of patients were classified to have stable clearance over the dosing period. Most patients that developed an inactivation over the course of treatment were classified as having increased clearance over time (93% increased clearance, 7% stable clearance), while most patients without inactivation were classified as having constant clearance over the dosing occasions (86% stable clearance, 14% increased clearance). Based on these results, pharmacokinetic model classifies patients with inactivation based on increased clearance with a sensitivity of 93% and a specificity of 86%.
Discussion
The aim of this study was to investigate the comprehensive pharmacokinetics of peg-asparaginase and to assess the significance of AEA measurements in predicting inactivation and potentially life-threatening allergic reactions among patients diagnosed with ALL who were undergoing treatment with peg-asparaginase. AEA levels were measured in a specific patient cohort comprising individuals aged 1–45 years with ALL, who received peg-asparaginase treatment in accordance with the ALLTogether pilot protocol implemented in the Nordic and Baltic regions. We demonstrated that a pharmacokinetic model based on the AEA could estimate the probability that patients belonged to one of two subpopulations of either increased or constant clearance over the treatment period. Patients allocated to the increased clearance group over dosing occasions were frequently individuals who experienced inactivation, while those assigned to stable clearance were less prone to inactivate. The difference in clearance was present from the period between 2nd and 3rd dose administered in patients inactivating at 3rd as well as 4th and 5th dose (Fig. 3).
This study presents a pioneering methodology for predicting inactivation by employing a pharmacokinetic model to identify early alterations in clearance. Notably, our investigation is grounded on a robust dataset comprising a substantial number of samples. The adherence to TDM of asparaginase within the NOPHO consortium was exceptional, resulting in near-complete sampling and empowering both statistical and pharmacokinetic analyses. This remarkable compliance is the primary strength of our study. Moreover, the availability of high-quality data pertaining to clinical symptoms further reinforces the study’s findings.
In studies including TDM no difference has been found in the incidence of inactivation when AEA measurements have been used to compare IM and IV administration of peg-asparaginase.(4, 32) Overall, the incidence of inactivation (18.2%) in this study was high compared with concurrent protocols [4, 32] This might be explained by extensive sampling and a high compliance rate or by some type of change in the peg-asparaginase formulation, but this must be considered as speculative. The number of patients (n = 45) excluded due to missing ethical approval for sampling in their country did not participate with sampling for TDM measurements making the assessment of the incidence somewhat uncertain. The majority of such patients (56%) were adults, who are generally considered to have a lower incidence of inactivation of asparaginase [33]. Consistent with literature only three of the 46 patients with inactivation in this study were ≥16 years (6.5%).
The mean AEA Ctrough in the group of patients without inactivation treated with IV peg-asparaginase <16 years was high (322–516 IU/L over the first four doses). In the DCOG and Dana Farber 00–01 studies, attempts were made to reduce or increase the dosage of asparaginase based on enzyme activities. These studies demonstrated unchanged survival rates with the potential for dose reduction [6, 34]. Thus, the higher Ctrough values for patients <16 years in this cohort indicate that dose reductions may be possible without compromising the efficacy of the asparaginase treatment assuming that higher Ctrough is not correlated to a better outcome. Higher AEA-values might also be associated with an increased risk of certain toxicities [3, 6, 17].
Previous studies have shown that some patients developed inactivation after the first dose of peg-asparaginase leading to the hypothesis that anti-peg antibodies were present before the start of treatment [5, 35,36,37,38]. In the present study all patients had AEA Ctrough > 100 IU/L after the first dose, which suggests that sensitization to peg-asparaginase occurred after the first administration. Additionally, sampling between dose 1 and 2 showed no significant difference in mean AEA Ctrough (Figs. 2 and 3) in the groups, supporting that most inactivation were seen following second dose or later. The variation in the triggering dose in different treatment protocols could potentially be attributed to the simultaneous administration of dexamethasone during induction, which may delay the immune response and the occurrence of inactivation reactions [39] and/or the early introduction of peg-asparaginase treatment compared with introduction of peg-asparaginase post-induction e.g. the NOPHO ALL2008 protocol [40] and the CoALL 08–09 [41].
The remarkable sensitivity (93%) and specificity (86%) observed in the pharmacokinetic model highlight its potential value for clinicians. By leveraging this model, healthcare professionals can gain valuable insights to proactively prepare for potential adverse reactions. This preparation may involve extending the infusion duration, implementing premedication, or exploring desensitization techniques. While completely averting inactivation remains a challenge, delaying the reaction opens the door to administering more effective peg-asparaginase doses before a switch. This intervention also offers the potential to act proactively before a life-threatening allergic reaction occurs, thereby providing a means to mitigate distressing experiences for both patients and their families. Importantly, these measures may contribute to optimizing peg-asparaginase treatment and ensuring favorable survival rates.
Few patients (n = 18, 7%) demonstrated induced clearance in the pharmacokinetic model but did not develop an allergic reaction during the peg-asparaginase treatment. This could be explained by the low number of peg-asparaginase doses administered according to the risk group (SR patients = 4 doses), which might lead to completion of peg-asparaginase treatment before the full allergic reaction occurred.
Additionally, it is essential to assess the quantity and precise timepoints of pharmacokinetic samples needed to anticipate inactivation events. In our study, the timing of extended sampling was determined by existing evidence. The time point (seven days after first dose) in the logistic regression model showed no value in predicting inactivation why extended sampling for AEA should occur shortly and less than 14 days after administration of the second dose. We strongly recommend incorporating this approach in future studies. Additionally, the implementation of model-based optimal design can offer valuable support in determining the appropriate sampling times [42]. Increasing the number of samples (day four and/or seven) following the second dose is likely to enhance the sensitivity for detecting accelerated clearance as an early indication of impending inactivation, allowing for timely adjustments to the preceding dose or appropriate adjustments to the upcoming dose. Moreover, an additional factor that impacts the timing of hypersensitivity reactions is the presence of breaks or extended intervals between doses [4]. This is supported by unpublished data from the current ALLTogether1 main protocol. A pause in the peg-asparaginase treatment during consolidation 1 has been introduced due to an unacceptable frequency of acute toxicities (unpublished data). Despite the pause in the peg-asparaginase treatment between the second and third doses for patients <25 years, the model is anticipated to detect increased clearance shortly after the second dose. Lastly, although outside of the scope of this project, the pharmacokinetic model could be useful to enable dose reductions in patients that are far above the treatment threshold, potentially resulting in reduced treatment costs and most importantly perhaps reducing the incidence of asparaginase-related toxicities. However, more studies are needed to support this.
One limitation of this study, as well as in other relevant literature, is the potential risk of misinterpreting inactivation reactions. In cases where a severe allergic reaction occurred during infusion, the administration was often terminated prematurely, resulting in the patient receiving only a fraction of the intended dose. This situation makes it challenging to accurately assess the subsequent AEA Ctrough when it falls below LLQ. Within this study, 26 out of 36 patients who experienced an allergic reaction had AEA Ctrough values below the LLQ prior to the reaction. Additionally, the majority of these reactions were severe, providing reasonable grounds to assume the presence of true inactivation [13]. However, in 10 out of 36 patients, AEA Ctrough data were not available to reflect the previously administered dose, potentially leading to misdiagnosis if only a minimal amount of the dose was given. Nonetheless, the severity (seven severe, three mild) and timing of the allergic reactions argue against the likelihood of allergy-like reactions [3, 13]. Future studies are likely to place greater emphasis on maintaining patients on peg-asparaginase, which could potentially influence the treatment strategy in situations of uncertainty, favouring the administration of an additional dose of peg-asparaginase with close TDM supervision. This approach could also involve considering pre-medication and adjusting the infusion duration.
In general, the patient groups ≥16 years, as well as those who received IM peg-asparaginase treatment, were limited in size. This limited sample size within these subgroups led to reduced statistical power and made it impossible to compare the level of AEA between those who experienced inactivation and those who maintained sufficient activity.
Conclusion
In conclusion, this study utilized a pharmacokinetic model based on AEA measurements to predict inactivation in patients undergoing peg-asparaginase treatment for ALL. The model effectively distinguished patients with increased clearance, who were more likely to experience inactivation, from those with stable clearance. This distinction was evident as early as immediately after 2nd dose in patients who later experienced inactivation.
This methodology represents a pioneering approach for predicting inactivation by identifying early changes in clearance. It offers clinicians a valuable tool to proactively manage treatment, switch asparaginase formulation timely, and potentially prevent ALL relapses.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Silverman LB, Gelber RD, Dalton VK, Asselin BL, Barr RD, Clavell LA, et al. Improved outcome for children with acute lymphoblastic leukemia: results of Dana-Farber Consortium Protocol 91-01. Blood. 2001;97:1211–8.
Brigitha LJ, Pieters R, van der Sluis IM. How much asparaginase is needed for optimal outcome in childhood acute lymphoblastic leukaemia? A systematic review. Eur J Cancer. 2021;157:238–49.
van der Sluis IM, Vrooman LM, Pieters R, Baruchel A, Escherich G, Goulden N, et al. Consensus expert recommendations for identification and management of asparaginase hypersensitivity and silent inactivation. Haematologica. 2016;101:279–85.
Brigitha LJ, Fiocco M, Pieters R, Albertsen BK, Escherich G, Lopez-Lopez E, et al. Hypersensitivity to Pegylated E.coli asparaginase as first-line treatment in contemporary paediatric acute lymphoblastic leukaemia protocols: a meta-analysis of the Ponte di Legno Toxicity working group. Eur J Cancer. 2022;162:65–75.
Henriksen LT, Harila-Saari A, Ruud E, Abrahamsson J, Pruunsild K, Vaitkeviciene G, et al. PEG-asparaginase allergy in children with acute lymphoblastic leukemia in the NOPHO ALL2008 protocol. Pediatr Blood Cancer 2015;62:427–33.
Kloos RQH, Pieters R, Jumelet FMV, de Groot-Kruseman HA, van den Bos C, van der Sluis IM. Individualized asparaginase dosing in childhood acute lymphoblastic leukemia. J Clin Oncol. 2020;38:715–24.
Gottschalk Højfeldt S, Grell K, Abrahamsson J, Lund B, Vettenranta K, Jónsson ÓG, et al. Relapse risk following truncation of pegylated asparaginase in childhood acute lymphoblastic leukemia. Blood. 2021;137:2373–82.
Albertsen BK, Grell K, Abrahamsson J, Lund B, Vettenranta K, Jónsson ÓG, et al. Intermittent versus continuous PEG-asparaginase to reduce asparaginase-associated toxicities: a NOPHO ALL2008 randomized study. J Clin Oncol. 2019;37:1638–46.
Rank CU, Toft N, Tuckuviene R, Grell K, Nielsen OJ, Frandsen TL, et al. Thromboembolism in acute lymphoblastic leukemia: results of NOPHO ALL2008 protocol treatment in patients aged 1 to 45 years. Blood. 2018;131:2475–84.
Gupta S, Wang C, Raetz EA, Schore R, Salzer WL, Larsen EC, et al. Impact of asparaginase discontinuation on outcome in childhood acute lymphoblastic leukemia: a report from the Children’s Oncology Group. J Clin Oncol. 2020;38:1897–905.
Schmiegelow K, Attarbaschi A, Barzilai S, Escherich G, Frandsen TL, Halsey C, et al. Consensus definitions of 14 severe acute toxic effects for childhood lymphoblastic leukaemia treatment: a Delphi consensus. Lancet Oncol. 2016;17:e231–e9.
Tong WH, Pieters R, Kaspers GJ, te Loo DM, Bierings MB, van den Bos C, et al. A prospective study on drug monitoring of PEGasparaginase and Erwinia asparaginase and asparaginase antibodies in pediatric acute lymphoblastic leukemia. Blood. 2014;123:2026–33.
Kloos RQH, Pieters R, Escherich G, van der Sluis IM. Allergic-like reactions to asparaginase: atypical allergies without asparaginase inactivation. Pediatr Blood Cancer 2016;63:1928–34.
Asselin B, Rizzari C. Asparaginase pharmacokinetics and implications of therapeutic drug monitoring. Leuk Lymphoma. 2015;56:2273–80.
Aldoss I, Pourhassan H, Douer D. SOHO State of the Art Updates and Next Questions | asparaginase-understanding and overcoming toxicities in adults with ALL. Clin Lymphoma Myeloma Leuk. 2022;22:787–94.
Tram Henriksen L, Gottschalk Højfeldt S, Schmiegelow K, Frandsen TL, Skov Wehner P, Schrøder H, et al. Prolonged first-line PEG-asparaginase treatment in pediatric acute lymphoblastic leukemia in the NOPHO ALL2008 protocol-Pharmacokinetics and antibody formation. Pediatr Blood Cancer. 2017;64. https://doi.org/10.1002/pbc.26686.
Jeha S, Pei D, Choi J, Cheng C, Sandlund JT, Coustan-Smith E, et al. Improved CNS control of childhood acute lymphoblastic leukemia without cranial irradiation: St Jude total therapy study 16. J Clin Oncol. 2019;37:3377–91.
Mattano LA Jr., Devidas M, Maloney KW, Wang C, Friedmann AM, Buckley P, et al. Favorable trisomies and ETV6-RUNX1 predict cure in low-risk B-cell acute lymphoblastic leukemia: results from Children’s Oncology Group Trial AALL0331. J Clin Oncol. 2021;39:1540–52.
Vrooman LM, Blonquist TM, Stevenson KE, Supko JG, Hunt SK, Cronholm SM, et al. Efficacy and toxicity of pegaspargase and calaspargase pegol in childhood acute lymphoblastic leukemia: results of DFCI 11-001. J Clin Oncol. 2021;39:3496–505.
Burke MJ, Zalewska-Szewczyk B. Hypersensitivity reactions to asparaginase therapy in acute lymphoblastic leukemia: immunology and clinical consequences. Future Oncol. 2022;18:1285–99.
Hillion S, Arleevskaya MI, Blanco P, Bordron A, Brooks WH, Cesbron JY, et al. The innate part of the adaptive immune system. Clin Rev Allergy Immunol. 2020;58:151–4.
Avramis VI, Tiwari PN. Asparaginase (native ASNase or pegylated ASNase) in the treatment of acute lymphoblastic leukemia. Int J Nanomed. 2006;1:241–54.
Würthwein G, Lanvers-Kaminsky C, Siebel C, Gerß J, Möricke A, Zimmermann M, et al. Population pharmacokinetics of PEGylated asparaginase in children with acute lymphoblastic leukemia: treatment phase dependency and predictivity in case of missing data. Eur J Drug Metab Pharmacokinetics. 2021;46:289–300.
Würthwein G, Lanvers-Kaminsky C, Hempel G, Gastine S, Möricke A, Schrappe M, et al. Population pharmacokinetics to model the time-varying clearance of the PEGylated Asparaginase Oncaspar(®) in children with acute lymphoblastic leukemia. Eur J Drug Metab Pharmacokinet. 2017;42:955–63.
Lynggaard LS, Rank CU, Hansen SN, Gottschalk Højfeldt S, Henriksen LT, Jarvis KB, et al. Asparaginase enzyme activity levels and toxicity in childhood acute lymphoblastic leukemia: a NOPHO ALL2008 study. Blood Adv. 2022;6:138–47.
Müller HJ, Löning L, Horn A, Schwabe D, Gunkel M, Schrappe M, et al. Pegylated asparaginase (Oncaspar) in children with ALL: drug monitoring in reinduction according to the ALL/NHL-BFM 95 protocols. Br J Haematol. 2000;110:379–84.
Lanvers C, Vieira Pinheiro JP, Hempel G, Wuerthwein G, Boos J. Analytical validation of a microplate reader-based method for the therapeutic drug monitoring of L-asparaginase in human serum. Anal Biochem. 2002;309:117–26.
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: Building an international community of software platform partners. J Biomed Inf. 2019;95:103208.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inf. 2009;42:377–81.
Bergstrand M, Karlsson MO. Handling data below the limit of quantification in mixed effect models. AAPS J. 2009;11:371–80.
Anderson BJ, Holford NH. Mechanistic basis of using body size and maturation to predict clearance in humans. Drug Metab Pharmacokinet. 2009;24:25–36.
Place AE, Stevenson KE, Vrooman LM, Harris MH, Hunt SK, O’Brien JE, et al. Intravenous pegylated asparaginase versus intramuscular native Escherichia coli L-asparaginase in newly diagnosed childhood acute lymphoblastic leukaemia (DFCI 05-001): a randomised, open-label phase 3 trial. Lancet Oncol. 2015;16:1677–90.
Stock W, Douer D, DeAngelo DJ, Arellano M, Advani A, Damon L, et al. Prevention and management of asparaginase/pegasparaginase-associated toxicities in adults and older adolescents: recommendations of an expert panel. Leuk Lymphoma. 2011;52:2237–53.
Vrooman LM, Stevenson KE, Supko JG, O’Brien J, Dahlberg SE, Asselin BL, et al. Postinduction dexamethasone and individualized dosing of Escherichia Coli L-asparaginase each improve outcome of children and adolescents with newly diagnosed acute lymphoblastic leukemia: results from a randomized study–Dana-Farber Cancer Institute ALL Consortium Protocol 00-01. J Clin Oncol. 2013;31:1202–10.
Armstrong JK, Hempel G, Koling S, Chan LS, Fisher T, Meiselman HJ, et al. Antibody against poly(ethylene glycol) adversely affects PEG-asparaginase therapy in acute lymphoblastic leukemia patients. Cancer. 2007;110:103–11.
Khalil A, Würthwein G, Golitsch J, Hempel G, Fobker M, Gerss J, et al. Pre-existing antibodies against polyethylene glycol reduce asparaginase activities on first administration of pegylated E. coli asparaginase in children with acute lymphocytic leukemia. Haematologica. 2022;107:49–57.
Kloos R, van der Sluis IM, Mastrobattista E, Hennink W, Pieters R, Verhoef JJ. Acute lymphoblastic leukaemia patients treated with PEGasparaginase develop antibodies to PEG and the succinate linker. Br J Haematol. 2020;189:442–51.
Kloos RQH, Mathôt R, Pieters R, van der Sluis IM. Individualized dosing guidelines for PEGasparaginase and factors influencing the clearance: a population pharmacokinetic model. Haematologica. 2021;106:1254–61.
Coutinho AE, Chapman KE. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol Cell Endocrinol. 2011;335:2–13.
Toft N, Birgens H, Abrahamsson J, Griškevičius L, Hallböök H, Heyman M, et al. Results of NOPHO ALL2008 treatment for patients aged 1–45 years with acute lymphoblastic leukemia. Leukemia. 2018;32:606–15.
Escherich G, Zur Stadt U, Zimmermann M, Horstmann MA. Clofarabine in combination with pegylated asparaginase in the frontline treatment of childhood acute lymphoblastic leukaemia: a feasibility report from the CoALL 08-09 trial. Br J Haematol. 2013;163:240–7.
Retout S, Duffull S, Mentré F. Development and implementation of the population Fisher information matrix for the evaluation of population pharmacokinetic designs. Comput Methods Prog Biomed. 2001;65:141–51.
Acknowledgements
The authors thank all principal investigators, research nurses and other staff members from participating treatment sites for their valuable contribution to TDM measurements and the study in general. The work was supported by the Danish Childhood Cancer Foundation (2018–3682 and 2020–6755), The Swedish Childhood Cancer Fund (PR2021–0064), The Swedish Cancer Society (20 1226 PjF), The Health Research Foundation of Central Denmark Region, Ølufgaard Foundation, Lizzy and Mogens Staal Foundation and Brogaard Foundation. The computations were enabled by resources provided by the Swedish National Infrastructure for Computing (SNIC) at [SNIC CENTRE] partially funded by the Swedish Research Council through grant agreement no. 2018-05973.
Funding
Open access funding provided by Aarhus University Hospital.
Author information
Authors and Affiliations
Contributions
BKA conceived and designed the study, MC, LEF, DC, MOK developed the pharmacokinetic model, MD collected and analyzed data in collaboration with SNA. MD, MC, LEF, LSL and BKA wrote the manuscript, and all authors gave final approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
Jazz Pharmaceuticals has BKA serving on its advisory board and MD served as a consultant for Servier on a single occasion. All other authors state that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dam, M., Centanni, M., Friberg, L.E. et al. Increase in peg-asparaginase clearance as a predictor for inactivation in patients with acute lymphoblastic leukemia. Leukemia 38, 712–719 (2024). https://doi.org/10.1038/s41375-024-02153-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-024-02153-6
- Springer Nature Limited