Abstract
Our objective is to analyse the observations related to intrauterine volvulus and assess how clinical manifestations and treatment strategies impact prognosis. We conducted a comprehensive search on Pubmed and ClinicalTrials.gov from inception to July 2022, using search terms like “intrauterine volvulus” or “foetal volvulus,” supplemented by manual scrutiny of reference lists in relevant texts and articles. Our review encompassed 57 case reports/case series, involving 88 cases. The presence of foetal bradycardia during prenatal visits (p = 0.002) and the existence of meconium cyst or pseudocyst (p = 0.038) significantly influence survival rates. Preterm labour occurred more frequently among cases resulting in mortality (54% vs 21%; p = 0.055). Our study’s limitations include the inability to access all reported cases and reliance solely on available data. We advocate for vigilant monitoring of foetuses exhibiting signs of intestinal obstruction, and consideration of an emergent caesarean section as a pre-emptive measure before foetal biophysical profile deterioration worsens.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Midgut or intestinal volvulus arises when loops of the intestine become twisted around the pedicle of the superior mesenteric artery. This condition constitutes a surgical emergency due to the heightened risk of mortality and morbidity associated with delayed diagnosis and treatment [1]. Although the volvulus is mostly observed during the infant period, intrauterine volvulus cases were also reported [2].
The causes of postnatal volvulus are mostly malrotation, followed by cystic fibrosis and gastrointestinal mass [1]. The majority of intrauterine volvulus cases occur without any predisposing factors. Less commonly, intrauterine volvulus can be attributed to conditions such as malrotation, intestinal atresia, meconium ileus (associated with cystic fibrosis), or anomalies in the mesentery [3]. For most cases, signs of intrauterine volvulus are non-specific; thus, intrauterine intestinal volvulus, a very rare life-threatening condition, is difficult to diagnose prenatally [4, 5].
We review case reports and series regarding intrauterine volvulus in English literature with the aim of identifying diagnostic indicators and assessing how clinical findings and treatment methods influence prognosis.
Methods
Search
We conducted a thorough search on PubMed (http://www.pubmed.ncbi.nlm.nih.gov) and ClinicalTrials.gov (http://www.clinicaltrials.gov) spanning from their inception to July 2022. Our search utilised the terms “intrauterine volvulus,” yielding 91 results on PubMed, and “foetal volvulus,” yielding 295 results on PubMed and 4 results on ClinicalTrials.com, all pertaining to English literature.
Study selection
The article headings were scrutinised, and potential case reports and case series concerning intrauterine volvulus were identified. Instances of repetition or duplication were treated as singular cases. The abstracts of these articles were then examined to ascertain whether they indeed reported cases of intrauterine volvulus. We selected case reports or case series with headings explicitly indicating a focus on intrauterine volvulus, where the cases were presented as instances of intrauterine volvulus with either prenatal diagnosis via prenatal ultrasonography or operative findings immediately after birth. In total, we identified 60 case reports or case series. Unfortunately, access to 8 of these articles was not possible. However, we included 2 cases reported in detail in another case review. [6] We reached 52 articles, read them and list the findings of cases to a table for analysis (dataset). Additionally, we manually examined the reference lists of the included texts and relevant articles to identify any cases of intrauterine volvulus not captured in the initial literature search. This method led to the discovery of four case reports and one case series. These findings were then incorporated into the dataset. In conclusion, we conducted a review of 57 case reports or case series, providing adequate information on a total of 88 cases (as depicted in Fig. 1 and dataset) [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57].
We omitted cases lacking volvulus findings on antenatal ultrasonography (USG) and those not promptly operated on after birth (presumed to be postnatal volvulus), as well as those documented to have undergone spontaneous resolution (specifically, three cases from the case series by Bartholmot et al.) [1].
Data İtems
One reviewer (TAD) meticulously recorded the information of these cases in a table as they were reported, leaving unreported data blank in the table. Subsequently, we conducted statistical analysis on these data. The recorded data encompassed various parameters, including antenatal USG findings, foetal biometry and biophysical profile scores, gestational age at diagnosis and birth, birth weight, gender, physical examination results, radiological and operative findings of the newborn, type of surgical procedure performed (whether anastomosis or enterostomy), associated pathologies, potential aetiological factors of volvulus, and outcomes. The duration of volvulus was determined by subtracting the gestational age at diagnosis from the gestational age at birth.
In cases where certain features or findings were not reported, we assumed them to be within normal range. For prenatal diagnosis of the pathology, any suspicious prenatal ultrasound findings such as the presence of dilated bowel or ascites were accepted, influencing both prenatal and postnatal diagnoses accordingly.
Although necropsy findings from four cases reported by Ashworth [57] were documented in the dataset, they were not included in the operative findings, which were noted based on explorative laparotomies conducted after birth.
In instances where “no signal within/between loops” was reported on Doppler Ultrasound, this was defined as “Abnormal Doppler Findings.” Given that most authors did not specify whether bowel atresia occurred in the small bowel or large bowel, the term “bowel atresia” was used generally.
Synthesis of results and statistics
We compiled the data into a table (dataset) and conducted statistical analysis. Cases were grouped based on survival outcomes: Group 1 included survived cases, while Group 2 encompassed lost cases, including miscarriage, intrauterine death, and postnatal loss cases. Four cases underwent pregnancy termination, and five cases lacked outcome data; hence, these cases were excluded from grouping.
Statistical analyses were performed using the IBM SPSS Statistics Version 25.0 software program (IBM Corp., Armonk, NY, USA). Descriptive statistics for categorical variables were presented as numbers (n) and percentages (%). For numerical parameters, if parametric test assumptions were met, descriptive statistics were presented as mean ± standard deviation. If parametric test assumptions were not met, the median and minimum-maximum values were reported. Parametric continuous variables were compared using the t-test, while non-parametric continuous variables were compared using the Mann–Whitney U test. Categorical variables were compared using either the Pearson chi-square test or Fisher’s exact test. A p value of less than 0.05 was considered statistically significant.
Results
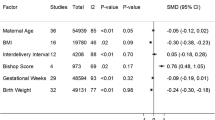
We reviewed 57 case reports or case series containing enough information on 88 cases (Fig. 1, dataset) [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57]. Tables 1, 2 present the distribution of cases and provide the p-values for the comparison between Groups 1 and 2 (Tables 1, 2). Table 3 displays the distribution of gestational age at diagnosis and birth, birth weight, and duration of volvulus, along with the corresponding p values for each finding in the comparison between Groups 1 and 2 (Table 3; Figs. 2–4).
Our analysis revealed significant differences between Groups 1 and 2. Specifically, the presence of foetal bradycardia at prenatal visits (p = 0.002) and the presence of meconium cyst or pseudocyst reported during the operation (p = 0.038) were notably lower in Group 1 compared to Group 2. Most cases in Group 1 (84%) were diagnosed prenatally, a statistically significant difference from Group 2 (44%) (p = 0.002) (Table 1). Among the five cases reported to have foetal bradycardia, four cases resulted in mortality. Additionally, among the three cases with foetal hydrops, only one survived while two died.
Although preterm labour was more frequent in Group 2 (54% vs. 21%; p = 0.055), this difference did not reach statistical significance. Among the cases with preterm labour, two out of five delivered via caesarean section, and four out of twelve delivered vaginally resulted in mortality. In Group 1, the proportion of term newborns was 22%, while in Group 2, it was 42% (p = 0.167). Among the cases, three out of 27 delivered via emergent caesarean section, four out of 13 with preterm birth without urgent caesarean section, and four out of 25 with planned delivery resulted in mortality. However, there was no statistically significant difference between urgent caesarean section and planned delivery (p = 0.689) or between preterm birth without urgent caesarean section and urgent caesarean section (p = 0.172).
Among the 67 cases with reported antenatal diagnosis time, 79% were diagnosed after 27 weeks of gestational age (Fig. 2). Regarding preterm births, 55.6% occurred before 34 weeks gestation, 30.6% were late preterm births, and 13.9% were term births.
Furthermore, additional physical examinations of three cases were reported as normal. Notably, in one of these cases, although intrauterine death occurred at the 39th gestational week, the postnatal physical examination was normal [28].
Additionally, contrast enema was conducted for six cases, yielding findings such as narrow or microcolon (3 cases), normal colon (2 cases), caecum at the right upper quadrant (1 case), and suspected stenosis at the terminal ileum (1 case). Upper gastrointestinal series were performed for six cases, resulting in reports of normal findings (3 cases), obstruction at mid-jejunum (1 case), malrotation (1 case), and barium stopping at the level of the right lower quadrant, along with a “bird beak” sign with a filling defect (1 case).
Operative findings were available for 82 cases, with four of these cases not explicitly mentioning volvulus; however, they were reported to have pseudocyst/ meconium cyst secondary to volvulus [8, 9, 14, 56]. Four of 11 cases with pseudocyst/ meconium cyst, and seven of 49 cases without pseudocyst/meconium cyst died (p = 0.038).
Fifteen cases were found to be associated with foetal anaemia. Among these, six out of eight cases with hemoperitoneum and nine out of 56 cases without hemoperitoneum exhibited foetal anaemia (p = 0.001). Additionally, all three cases of foetal hydrops were found to have foetal anaemia (p = 0.011). Furthermore, four out of the 15 cases with foetal anaemia required blood transfusion.
Paracentesis was performed antenatally for two cases [8, 24], which drained bilious or meconium-stained ascites, and postnatally for two cases [11, 12], which drained bloody ascites.
Discussion
Finley et al. suggested that a delayed return of foetal bowel from the umbilical cord to the foetal abdomen, potentially resulting in the failure of normal fixation of the small bowel mesentery, could predispose to intrauterine volvulus, as observed in their reported case [15]. Due to the lesser or greater degree of rotation of the gut around the superior mesenteric artery, Cloutier et al. blamed the absence of broad mesenteric attachment [9]. While the majority of postnatal volvulus cases were attributed to malrotation, it is noteworthy that only 17% of intrauterine volvulus cases were associated with malrotation. Additionally, a significant proportion of intrauterine volvulus cases (46%) were found to have no identifiable aetiological or associated anomalies.
Our review identified 22 cases (29%) that were associated with atresia. Notably, the literature cites the rate of atresia in volvulus cases as 25% [5, 38]. However, it remains unclear from the reports whether these atresias occurred secondary to the volvulus or if the volvuli occurred secondary to the atresia. We posit that both scenarios may be true [1, 58].
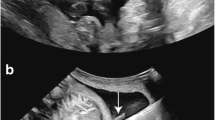
In the literature, the prenatal ultrasonographic findings of intrauterine volvulus were reported as polyhydramnios, dilated intestinal loops, static abdominal mass, ascites, peritoneal calcifications and increased abdominal circumference [2,3,4, 17, 26, 30, 32, 36, 38, 40, 41, 46, 51]. In our review, we identified similar lists of prenatal USG findings, although peritoneal calcifications were less frequent while meconium cysts/pseudocysts were more common. Additionally, the literature reported foetal biophysical profile findings as foetal distress with decreased foetal movements and decreased variability during foetal monitorisation, along with foetal cardiac sinus rhythm with anaemia and increased peak systolic velocity in the middle cerebral artery Doppler [2,3,4, 17, 26, 30, 32, 36, 38, 40, 41, 46, 52] which was mostly observed after 27 weeks of gestation [4, 25, 26, 41]. We found similar foetal biophysical profile findings and more frequent foetal bradycardia, which also increases mortality.
The ultrasonographic signs of intrauterine volvulus can be indicative of several differential diagnoses, including intestinal atresia, meconium ileus, meconium peritonitis, segmental small bowel dilatations, cystic mass lesions such as mesenteric cysts, lymphangioma, or teratoma, Meckel’s diverticulum, duplication cysts, and omphalomesenteric cysts [45, 51, 53]. Definitive diagnosis of volvulus could only be made by the presence of the “whirlpool or snail configuration” or the “coffee bean sign” [1, 3, 4, 26, 32, 36, 38, 40, 43, 49, 53], which are difficult to define [5, 30] and not always associated with volvulus [59]. The sensitivity and specificity of the whirlpool sign were reported as 89% and 92%, respectively in neonatal volvulus cases [54, 60]. We found that the “whirlpool or snail configuration” was reported in 19 cases (24%) and the “coffee bean sign” was reported in 4 cases (5%). Due to the decreased occurrence of specific signs of volvulus and the increased prevalence of nonspecific signs that share many potential differential diagnoses, diagnosing and effectively treating these cases in a timely manner becomes challenging. Consequently, this contributes to elevated mortality and morbidity rates [3,4,5,6, 9, 13, 25, 30, 41, 43, 49, 51, 52].
Half of all the cases showed findings of foetal distress. Bartholmot et al. reported that 61.5% of mothers applied with the complaint of decreased foetal movements [1]. The increased risk of foetal loss and foetal distress may occur because of (1) Elevated intraabdominal pressure due to distended bowel and ascites, if present, can impede umbilical venous return and subsequently reduce cardiac output; (2) anaemia resulting from hemoperitoneum can lead to diminished perfusion; (3) the release of toxins from gangrenous or necrotic bowel tissue can exacerbate the physiological stress on the foetus [12, 22]; and (4) fluid escape into the third cavity (dilated bowel lumen) leading to hypovolemic shock and cardiovascular failure [33]. Leung et al. observed that following intrauterine paracentesis of ascites induced by meconium peritonitis, there was an improvement in foetal movements and suboptimal foetal heart rate patterns on non-stress test (NST). This improvement was attributed to the removal of irritant meconium, alleviation of compression on the foetal thorax, and potentially reducing compression on the vena cava, leading to enhanced venous return [24].
The literature mentioned that the outcome of intrauterine volvulus depends on the length of the viable intestine, level of obstruction, presence of meconium peritonitis, associated anomalies, birth weight, and gestational age at birth [17, 30, 38, 49, 54]. We found that the presence of foetal bradycardia at prenatal visits, and presence of meconium cyst or pseudocyst reported in operation were significantly related to decreased survival. Notably, 11 cases (17%) had short bowel syndrome, and two of them died.
One of the factors influencing mortality was the presence of meconium cyst/pseudocyst during operation. In deceased patients, a lower occurrence of dilated bowel in prenatal ultrasonography (44% vs 75%) and a higher incidence of meconium cyst/pseudocyst during operation (36% vs 9%) were observed. Thus, we hypothesize that mortality rises with necrosis and progression of volvulus to perforation, resulting in reduced bowel diameter and formation of meconium cysts.
Foetal anaemia, recognised for increasing mortality and morbidity in both the foetus and newborn, was more prevalent in cases with intrauterine volvulus [13, 36, 38, 42, 43]. While approximately one fourth of intrauterine volvulus cases in the literature were associated with foetal anaemia, we were unable to demonstrate its impact on mortality in our review. However, we observed that hemoperitoneum was a contributing factor to foetal anaemia. In line with the guidance provided by Kornacki et al., we advocate for evaluating the hemodynamic status of the foetus by measuring the peak systolic velocity of the middle cerebral artery in cases presenting with foetal bowel pathology, particularly in foetuses with intrauterine volvulus [36].
In severe cases, volvulus can manifest early in pregnancy, leading to the development of intestinal atresia and meconium peritonitis. Such instances may necessitate immediate intervention or preterm delivery due to the presence of foetal distress [6, 18]. Additionally, severe intrauterine volvulus led to ischaemic necrosis of intestines and might activate both feto-placental and hypothalamic release of stress hormones that results in uterine contractions and preterm labour [3, 20, 28, 30, 32, 42, 55]. We found that in the deceased patients, the preterm labour was more frequent (54% vs 21%), while urgent caesarean section and preterm newborn ratio was less frequent (27% vs 47%, 58% vs 78%, respectively). Therefore, we deduced that in cases where survival was achieved, obstetricians closely monitored these patients to detect signs of foetal distress and promptly conducted urgent caesarean sections to prevent foetal demise.
Two studies highlight that delays in diagnosing and treating intrauterine volvulus can lead to high complication rates, including loss of intestinal segments and the development of short bowel syndrome [17, 18]. Some authors suggest that delivery can be promptly performed via caesarean section after 34 weeks of gestation, while the timing of delivery before 34 weeks depends on the severity of the foetal condition and the need for prophylactic measures for foetal lung maturation [43, 45], Additionally, in cases where prenatal diagnosis is established, some authors recommend preterm delivery and emergent surgical intervention [26, 42]. However, others emphasize that the timing of delivery should be based on obstetrical indications, given the uncertainty in differentiating between intestinal atresia, meconium ileus, and volvulus [14, 17]. It’s also suggested to closely monitor foetuses presenting with signs of bowel obstruction and to consider urgent caesarean section in the presence of acute abdominal pathology or non-reassuring foetal status [1, 21, 55] when findings of acute abdominal pathology such as the heterogeneous echogenicity within the dilated bowel or the disappearance of peristalsis are presented [21] or when there are findings of non-reassuring foetal status [52, 55]. For instance, Herrera et al. recommend urgent delivery in the presence of specific indicators such as ascites, absence of intestinal peristalsis, decreased foetal movements, or sudden changes in bowel diameter [51], while Alvarez et al. suggest delivery when findings of thoracic compression are detected [14]. We recommend closely monitoring foetuses exhibiting signs of intestinal obstruction and administering betamethasone therapy for pulmonary maturity, particularly given the observed frequency of preterm labour. Urgent caesarean section should be considered as an option before foetal biophysical profile findings deteriorate further. It’s important to note that planned preterm delivery solely based on ultrasound findings is not advised, as these findings may also be indicative of conditions such as atresia or meconium peritonitis that do not necessitate urgent intervention. However, the likelihood of preterm delivery may still be higher due to a greater incidence of non-reassuring foetal tests.
However, our study has several limitations. Firstly, we were unable to access all the available case reports and case series, and our investigation was confined to English-language literature. Secondly, our data were constrained by what was reported, resulting in missing information on many features and findings. Thirdly, we assumed that certain non-reported features and findings were normal, such as the absence of necrotic bowel or mesenteric anomalies. If authors did not explicitly mention necrosis of bowel segments, we presumed the bowel to be viable. At the same time, it is possible that mesenteric anomalies were not universally investigated or documented by all surgeons, leading to potential oversight of many cases.
Due to the predominance of non-specific signs over specific signs of volvulus, which can mimic various differential diagnoses, diagnosing and treating these cases in a timely manner posed significant challenges. We concluded that the severity of volvulus may contribute to preterm labour, thereby increasing mortality. We posit that mortality escalates with necrosis and progression of volvulus to perforation, resulting in a reduction in bowel diameter and formation of meconium cysts. Consequently, we recommend close monitoring of foetuses exhibiting signs of intestinal obstruction, administration of betamethasone therapy for pulmonary maturity given the frequent occurrence of preterm labour, and consideration of urgent caesarean section before foetal biophysical profile findings deteriorate further.
References
Bartholmot C, Faure JM, Grosjean F, Couture A, Forgues D, Fuchs F, et al. Prenatal diagnosis of antenatal midgut volvulus: specific ultrasound features. Prenat Diagn. 2019;39:16–25. https://doi.org/10.1002/pd.5392.
Crisera C, Ginsburg HB, Gittes GK. Fetal midgut volvulus presenting at term. J Pediatr Surg. 1999;34:1280–1. https://doi.org/10.1016/s0022-3468(99)90169-0.
Kaba M, Oksuzoglu A, Kaba G, Timur H, Akbaba E, Turgut K. Fetal midgut volvulus with a cystic appearance, accompanying a sinus rhythm and an increased peak systolic velocity without anemia. Case Rep Obstet Gynecol. 2015;2015:354619. https://doi.org/10.1155/2015/354619.
Yoo SJ, Park KW, Cho SY, Sim JS, Hhan KS. Definitive diagnosis of intestinal volvulus in utero. Ultrasound Obstet Gynecol. 1999;13:200–3. https://doi.org/10.1046/j.1469-0705.1999.13030200.x.
Pederiva F, De Cunto A, Paviotti G, Cadrich D, Demarini S. Neonatal Cullen’s sign: a distinguishing feature of intrauterine volvulus with hemorrhagic ascites. APSP J Case Rep. 2013;4:53.
Morikawa N, Namba S, Fujii Y, Sato Y, Fukuba K. Intrauterine volvulus without malrotation associated with segmental absence of small intestinal musculature. J Pediatr Surg. 1999;34:1549–51. https://doi.org/10.1016/s0022-3468(99)90127-6.
Seward JF, Zusman J. Hydrops fetalis associated with small-bowel volvulus. Lancet. 1978;2:52–53. https://doi.org/10.1016/s0140-6736(78)91370-3.
Baxi LV, Yeh MN, Blanc WA, Schullinger JN. Antepartum diagnosis and management of in utero intestinal volvulus with perforation. N. Engl J Med. 1983;308:1519–21. https://doi.org/10.1056/NEJM198306233082508.
Cloutier MG, Fried AM, Selke AC. Antenatal observation of midgut volvulus by ultrasound. J Clin Ultrasound. 1983;11:286–8. https://doi.org/10.1002/jcu.1870110509.
Samuel N, Dicker D, Feldberg D, Goldman JA. Ultrasound diagnosis and management of fetal intestinal obstruction and volvulus in utero. J Perinat Med. 1984;12:333–7. https://doi.org/10.1515/jpme.1984.12.6.333.
Nogami W, Weber T, Lemons JA. Hydrops fetalis associated with midgut volvulus. J Pediatr Surg. 1985;20:177–8. https://doi.org/10.1016/s0022-3468(85)80297-9.
Witter FR, Molteni RA. Intrauterine intestinal volvulus with hemoperitoneum presenting as fetal distress at 34 weeks’ gestation. Am J Obstet Gynecol. 1986;155:1080–1. https://doi.org/10.1016/0002-9378(86)90354-6.
Usmani SS, Kenigsberg K. Intrauterine volvulus without malrotation. J Pediatr Surg. 1991;26:1409–10. https://doi.org/10.1016/0022-3468(91)91049-5.
Alvarez M, Lasker MR, Friedman F Jr, Chitkara U, Berkowitz RL, Holzman IR. Sonographic findings in a twin with congenital fetal volvulus. J Clin Ultrasound. 1991;19:98–100. https://doi.org/10.1002/jcu.1870190207.
Finley BE, Burlbaw J, Bennett TL, Levitch L. Delayed return of the fetal midgut to the abdomen resulting in volvulus, bowel obstruction, and gangrene of the small intestine. J Ultrasound Med. 1992;11:233–5. https://doi.org/10.7863/jum.1992.11.5.233.
Dell’Agnola CA, Tomaselli V, Teruzzi E, Tadini B, Coran AG. Prenatal diagnosis of gastrointestinal obstruction: a correlation between prenatal ultrasonic findings and postnatal operative findings. Prenat Diagn. 1993;13:629–32. https://doi.org/10.1002/pd.1970130713.
Mercado MG, Bulas DI, Chandra R. Prenatal diagnosis and management of congenital volvulus. Pediatr Radio. 1993;23:601–2. https://doi.org/10.1007/bf02014977.
Black PR, Mueller D, Crow J, Morris RC, Husain AN. Mesenteric defects as a cause of intestinal volvulus without malrotation and as the possible primary etiology of intestinal atresia. J Pediatr Surg. 1994;29:1339–43. https://doi.org/10.1016/0022-3468(94)90111-2.
Bronshtein M, Zimmer EZ. Early sonographic detection of fetal intestinal obstruction and possible diagnostic pitfalls. Prenat Diagn. 1996;16:203–6. https://doi.org/10.1002/(SICI)1097-0223(199603)16:3<203::AID-PD830>3.0.CO;2-#.
De Felice C, Massafra C, Centini G, Di Maggio G, Tota G, Bracci R. Relationship between intrauterine midgut volvulus without malrotation and preterm delivery. Acta Obstet Gynecol Scand. 1997;76:386. https://doi.org/10.1111/j.1600-0412.1997.tb08001.x.
Miyakoshi K, Tanaka M, Miyazaki T, Yoshimura Y. Prenatal ultrasound diagnosis of small-bowel torsion. Obstet Gynecol. 1998;91:802–3. https://doi.org/10.1016/s0029-7844(98)00026-x.
Ogunyemi D. Prenatal ultrasonographic diagnosis of ileal atresia and volvulus in a twin pregnancy. J Ultrasound Med. 2000;19:723–6. https://doi.org/10.7863/jum.2000.19.10.723.
Kubota A, Nakayama T, Yonekura T, Hoki M, Hirai H, Kosumi T, et al. Congenital ileal atresia presenting as a single cyst-like lesion on prenatal sonography. J Clin Ultrasound. 2000;28:206–8. https://doi.org/10.1002/(sici)1097-0096(200005)28:4<206::aid-jcu10>3.0.co;2-7.
Leung WC, Pandya P, Seaward G, Windrim R, Ryan G. Changes in fetal heart rate pattern after intrauterine paracentesis in one twin with meconium peritonitis. Prenat Diagn. 2001;21:693–6. https://doi.org/10.1002/pd.146.
Uerpairojkit B, Charoenvidhya D, Tanawattanacharoen S, Manotaya S, Wacharaprechanont T, Tannirandorn Y. Fetal intestinal volvulus: a clinico-sonographic finding. Ultrasound Obstet Gynecol. 2001;18:186–7. https://doi.org/10.1046/j.1469-0705.2001.00425.x.
Has R, Gunay S. ‘Whirlpool’ sign in the prenatal diagnosis of intestinal volvulus. Ultrasound Obstet Gynecol. 2002;20:307–8. https://doi.org/10.1046/j.1469-0705.2002.00779_4.x.
Jéquier S, Hanquinet S, Bugmann P, Pfizenmaier M. Antenatal small-bowel volvulus without malrotation: ultrasound demonstration and discussion of pathogenesis. Pediatr Radio. 2003;33:263–5. https://doi.org/10.1007/s00247-003-0871-3.
Allahdin S, Kay V. Ischaemic haemorrhagic necrosis of the intestine secondary to volvulus of the midgut: a silent cause of intrauterine death. J Obstet Gynaecol. 2004;24:310. https://doi.org/10.1080/01443610410001660940.
Trachsel D, Heinimann K, Bösch N, Hammer J. Cystic fibrosis and intrauterine death. J Perinatol. 2007;27:181–2. https://doi.org/10.1038/sj.jp.7211652.
Molvarec A, Bábinszki A, Kovács K, Toth F, Szalay J. Intrauterine intestinal obstruction due to fetal midgut volvulus: a report of two cases. Fetal Diagn Ther. 2007;22:38–40. https://doi.org/10.1159/000095841.
Rajab KE, Al Juffairi Z, Issa AA. Antenatal diagnosis and management of fetal midgut volvulus. Bahrain Med Bull. 2007;29:106–8.
Park JS, Cha SJ, Kim BG, Kim YS, Choi YS, Chang IT, et al. Intrauterine midgut volvulus without malrotation: diagnosis from the ‘coffee bean sign’. World J Gastroenterol. 2008;14:1456–8. https://doi.org/10.3748/wjg.14.1456.
Steffensen TS, Gilbert-Barness E, DeStefano KA, Kontopoulos EV. Midgut volvulus causing fetal demise in utero. Fetal Pediatr Pathol. 2008;27:223–31. https://doi.org/10.1080/15513810802319608.
Noreldeen SA, Hodgett SG, Venkat-Raman N. Midgut volvulus with hemorrhagic ascites: a rare cause of fetal anemia. Ultrasound Obstet Gynecol. 2008;31:352–4. https://doi.org/10.1002/uog.5223.
Durand M, Coste K, Martin A, Scheye T, Creveaux I, Vanlieferinghen P, et al. Fetal midgut volvulus as a sign for cystic fibrosis. Prenat Diagn. 2008;28:973–4. https://doi.org/10.1002/pd.2086.
Kornacki J, Czarnecka M, Błaszczyński M, Skrzypczak J, Gadzinowski J, Jankowski A, et al. Congenital midgut volvulus associated with fetal anemia. Fetal Diagn Ther. 2010;28:119–22. https://doi.org/10.1159/000315051.
Valladares E, Rodríguez D, Vela A, Cabre S, Lailla JM. Meconium pseudocyst secondary to ileum volvulus perforation without peritoneal calcification: a case report. J Med Case Rep. 2010;4:292. https://doi.org/10.1186/1752-1947-4-292.
Yu W, Ailu C, Bing W. Sonographic diagnosis of fetal intestinal volvulus with ileal atresia: a case report. J Clin Ultrasound. 2013;41:255–7. https://doi.org/10.1002/jcu.21896.
Shankar R, Rao S, Shetty KB. Primary segmental volvulus mimicking ileal atresia. J Neonatal Surg. 2013;2:6. https://doi.org/10.21699/jns.v2i1.17.
Ohuoba E, Fruhman G, Olutoye O, Zacharias N. Perinatal survival of a fetus with intestinal volvulus and intussusception: a case report and review of the literature. AJP Rep. 2013;3:107–12. https://doi.org/10.1055/s-0033-1349367.
Davidson SL, Hird MF, Ebel R, Gabani RU. Congenital volvulus without associated malrotation. OA Case Rep. 2013;2:160–2.
Chung JH, Lim GY, We JS. Fetal primary small bowel volvulus in a child without intestinal malrotation. J Pediatr Surg. 2013;48:e1–5. https://doi.org/10.1016/j.jpedsurg.2013.05.011.
Nakagawa T, Tachibana D, Kitada K, Kurihara Y, Terada H, Koyama M, et al. A case of fetal intestinal volvulus without malrotation causing severe anemia. Jpn Clin Med. 2015;6:1–3. https://doi.org/10.4137/JCM.S20760.
Artul S, Habib G, Adawi A, Mansour B, Nseir W. Intrauterine volvulus of terminal ileum without malrotation. J Clin Imaging Sci. 2013;3:50. https://doi.org/10.4103/2156-7514.122317.
Sciarrone A, Teruzzi E, Pertusio A, Bastonero S, Errante G, Todros T, et al. Fetal midgut volvulus: report of eight cases. J Matern Fetal Neonatal Med. 2016;29:1322–7. https://doi.org/10.3109/14767058.2015.1047336.
Best EJ, O’Brien CM, Carseldine W, Deshpande A, Glover R, Park F. Fetal midgut volvulus with meconium peritonitis detected on prenatal ultrasound. Case Rep. Obstet Gynecol. 2018;2018:5312179. https://doi.org/10.1155/2018/5312179.
Bharti D, Krishnan S. A neonate with bilateral vitreous hemorrhages after intra-uterine bowel infarction secondary to volvulus. J Natl Med Assoc. 2018;110:242–4. https://doi.org/10.1016/j.jnma.2017.05.006.
Gultekin ND, Salihoglu O, Alptekin A, Yılmaz FH, Altunhan H. Intrauterine volvulus that had been misunderstood as an antenatal mesenteric cyst. Pediatr Neonatol. 2019;60:467–8. https://doi.org/10.1016/j.pedneo.2018.08.009.
Vazquez DC, Renau MI, Clemente EJI, Serrano MB, Capella MS, Palomo AM. Intrauterine intestinal volvulus. J Pediatr. 2020;219:278–9. https://doi.org/10.1016/j.jpeds.2019.11.017.
Park KH, Bae MH, Lee NR, Han YM, Byun SY, Kim HY. Meconium peritonitis resulting from different etiologies in siblings: a case report. BMC Pediatr. 2020;20:106. https://doi.org/10.1186/s12887-020-2016-3.
Herrera TT, Rueda K, Espinosa H, Britton GB. Intestinal volvulus in the pump twin of a twin reversed arterial perfusion (TRAP) sequence after laser therapy at 18 weeks: a case report. J Med Case Rep. 2020;14:123. https://doi.org/10.1186/s13256-020-02444-3.
Nagano A, Onishi S, Tazaki Y, Kobayashi H, Ieiri S. Fetal intestinal volvulus without malrotation detected on prenatal ultrasound. Pediatr Int. 2021;63:845–6. https://doi.org/10.1111/ped.14550.
Gerçel G, Anadolulu Aİ. Intrauterine midgut volvulus as a rare cause of intestinal obstruction: a case report. J Med Case Rep. 2021;15:239. https://doi.org/10.1186/s13256-021-02778-6.
Khoury V, Omran A, Taishori N. A rare case of idiopathic intrauterine intestinal volvulus complicated with intestinal perforation: a case report from Syria. J Surg Case Rep. 2021;2021:rjab192. https://doi.org/10.1093/jscr/rjab192.
Mise N, Terui K, Mitsunaga T, Ohara Y, Hishiki T. Delivery time of two cases of fetal intestinal volvulus. Pediatr Int. 2022;64:e15137. https://doi.org/10.1111/ped.15137.
Sahin-Uysal N, Acer-Demir T, Güney LH, Sagnak-Akilli M. Intrauterine Volvulus. Turkiye Klinikleri J Case Rep. 2022;30:226–9. https://doi.org/10.5336/caserep.2022-90351.
Ashworth TG. Fetal volvulus of the small intestine. J Clin Pathol. 1988;41:594–5. https://doi.org/10.1136/jcp.41.5.594.
Nichol PF, Reeder A, Botham R. Humans, mice, and mechanisms of intestinal atresias: a window into understanding early intestinal development. J Gastrointest Surg. 2011;15:694–700. https://doi.org/10.1007/s11605-010-1400-y.
Van Winckel M, Voet D, Robberecht E. Whirlpool sign”: not always associated with volvulus in intestinal malrotation. J Clin Ultrasound. 1996;24:367–70.
Chao HC, Kong MS, Chen JY, Lin SJ, Lin JN. Sonogrphic features related to volvulus in neonatal intestinal malrotation. J Ultrasound Med. 2000;19:371–6.
Acknowledgements
We express our gratitude to Ülkü Ceren Köksoy for her valuable assistance in English language editing.
Funding
Open access funding provided by the Scientific and Technological Research Council of Türkiye (TÜBİTAK).
Author information
Authors and Affiliations
Contributions
The concept for the article was conceived by TAD. TAD and NSU conducted the literature search and data analysis. The initial draft of the manuscript was authored by TAD, while NSU provided critical revisions. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Acer-Demir, T., Şahin-Uysal, N. Intrauterine volvulus: systemic review of the literature with pooled analysis. J Perinatol (2024). https://doi.org/10.1038/s41372-024-01984-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41372-024-01984-6
- Springer Nature America, Inc.