Abstract
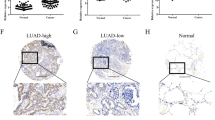
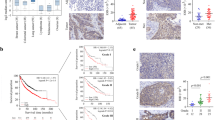
Lipoma preferred partner (LPP) is a LIM domain protein, which has multiple functions as an actin-binding protein and a transcriptional coactivator, and it has been suggested that LPP has some roles in cell migration or invasion, however, its role in cancer cells remains to be elucidated. Here, we showed that LPP degraded N-cadherin in lung cancer, PC14PE6 cells via regulating the expression of matrix metalloproteinase 15 (MMP-15), and loss-of-LPP increases collective cell migration (CCM) and dissemination consequently. Knockdown of LPP and its functional partner, Etv5, markedly restores the full-length N-cadherin and increases cell–cell adhesion. We investigated the common target of LPP and Etv5, and found that MMP-15 is transcribed as their direct transcriptional target. Furthermore, MMP-15 could directly digest the N-cadherin extracellular domain. LPP knockdown in PC14PE6 cells increases N-cadherin-dependent CCM in the three-dimensional collagen gel invasion assays, and promoted the dissemination of cancer cells when they were orthotopically implanted in nude mice. Immunohistochemistry of lung adenocarcinoma specimens revealed the heterogeneity of LPP intensity and complementary expression of LPP and N-cadherin in the primary tumors. These findings suggest that loss-of-LPP, Etv5 or MMP-15 can be a prognostic marker of increasing malignancy.






Similar content being viewed by others
References
Petit MM, Mols R, Schoenmakers EF, Mandahl N, Van de Ven WJ . LPP, the preferred fusion partner gene of HMGIC in lipomas, is a novel member of the LIM protein gene family. Genomics 1996; 36: 118–129.
Petit MM, Meulemans SM, Van de Ven WJ . The focal adhesion and nuclear targeting capacity of the LIM-containing lipoma-preferred partner (LPP) protein. J Biol Chem 2003; 278: 2157–2168.
Li B, Zhuang L, Reinhard M, Trueb B . The lipoma preferred partner LPP interacts with alpha-actinin. J Cell Sci 2003; 116: 1359–1366.
Petit MM, Fradelizi J, Golsteyn RM, Ayoubi TA, Menichi B, Louvard D et al. LPP, an actin cytoskeleton protein related to zyxin, harbors a nuclear export signal and transcriptional activation capacity. Mol Biol Cell 2000; 11: 117–129.
Guo B, Sallis RE, Greenall A, Petit MM, Jansen E, Young L et al. The LIM domain protein LPP is a coactivator for the ETS domain transcription factor PEA3. Mol Cell Biol 2006; 26: 4529–4538.
de Launoit Y, Baert JL, Chotteau-Lelievre A, Monte D, Coutte L, Mauen S et al. The Ets transcription factors of the PEA3 group: transcriptional regulators in metastasis. Biochim Biophys Acta 2006; 1766: 79–87.
Grunewald TG, Pasedag SM, Butt E . Cell adhesion and transcriptional activity - defining the role of the novel protooncogene LPP. Transl Oncol 2009; 2: 107–116.
Wang Y, Gilmore TD . Zyxin and paxillin proteins: focal adhesion plaque LIM domain proteins go nuclear. Biochim Biophys Acta 2003; 1593: 115–120.
Colas E, Muinelo-Romay L, Alonso-Alconada L, Llaurado M, Monge M, Barbazan J et al. ETV5 cooperates with LPP as a sensor of extracellular signals and promotes EMT in endometrial carcinomas. Oncogene 2012; 31: 4778–4788.
Xu J, Lai YJ, Lin WC, Lin FT . TRIP6 enhances lysophosphatidic acid-induced cell migration by interacting with the lysophosphatidic acid 2 receptor. J Biol Chem 2004; 279: 10459–10468.
Kuriyama S, Theveneau E, Benedetto A, Parsons M, Tanaka M, Charras G et al. In vivo collective cell migration requires an LPAR2-dependent increase in tissue fluidity. J Cell Biol. 2014; 206: 113–127.
Miyatani S, Shimamura K, Hatta M, Nagafuchi A, Nose A, Matsunaga M et al. Neural cadherin: role in selective cell-cell adhesion. Science 1989; 245: 631–635.
Wheelock MJ, Shintani Y, Maeda M, Fukumoto Y, Johnson KR . Cadherin switching. J Cell Sci 2008; 121: 727–735.
Hazan RB, Phillips GR, Qiao RF, Norton L, Aaronson SA . Exogenous expression of N-cadherin in breast cancer cells induces cell migration, invasion, and metastasis. J Cell Biol 2000; 148: 779–790.
Nieman MT, Prudoff RS, Johnson KR, Wheelock MJ . N-cadherin promotes motility in human breast cancer cells regardless of their E-cadherin expression. J Cell Biol 1999; 147: 631–644.
Li G, Satyamoorthy K, Herlyn M . N-cadherin-mediated intercellular interactions promote survival and migration of melanoma cells. Cancer Res 2001; 61: 3819–3825.
Kuphal S, Bosserhoff AK . Influence of the cytoplasmic domain of E-cadherin on endogenous N-cadherin expression in malignant melanoma. Oncogene 2006; 25: 248–259.
Tanaka H, Kono E, Tran CP, Miyazaki H, Yamashiro J, Shimomura T et al. Monoclonal antibody targeting of N-cadherin inhibits prostate cancer growth, metastasis and castration resistance. Nat Med 2010; 16: 1414–1420.
Asano K, Duntsch CD, Zhou Q, Weimar JD, Bordelon D, Robertson JH et al. Correlation of N-cadherin expression in high grade gliomas with tissue invasion. J Neurooncol 2004; 70: 3–15.
Camand E, Peglion F, Osmani N, Sanson M, Etienne-Manneville S . N-cadherin expression level modulates integrin-mediated polarity and strongly impacts on the speed and directionality of glial cell migration. J Cell Sci 2012; 125: 844–857.
Theveneau E, Marchant L, Kuriyama S, Gull M, Moepps B, Parsons M et al. Collective chemotaxis requires contact-dependent cell polarity. Dev Cell 2010; 19: 39–53.
Yano S, Nokihara H, Hanibuchi M, Parajuli P, Shinohara T, Kawano T et al. Model of malignant pleural effusion of human lung adenocarcinoma in SCID mice. Oncol Res 1997; 9: 573–579.
Cui ZY, Ahn JS, Lee JY, Kim WS, Lim HY, Jeon HJ et al. Mouse orthotopic lung cancer model induced by PC14PE6. Cancer Res Treat 2006; 38: 234–239.
Yamaura T, Murakami K, Doki Y, Sugiyama S, Misaki T, Yamada Y et al. Solitary lung tumors and their spontaneous metastasis in athymic nude mice orthotopically implanted with human non-small cell lung cancer. Neoplasia 2000; 2: 315–324.
Doki Y, Murakami K, Yamaura T, Sugiyama S, Misaki T, Saiki I . Mediastinal lymph node metastasis model by orthotopic intrapulmonary implantation of Lewis lung carcinoma cells in mice. Br J Cancer 1999; 79: 1121–1126.
Kohutek ZA, diPierro CG, Redpath GT, Hussaini IM . ADAM-10-mediated N-cadherin cleavage is protein kinase C-alpha dependent and promotes glioblastoma cell migration. J Neurosci 2009; 29: 4605–4615.
Covington MD, Burghardt RC, Parrish AR . Ischemia-induced cleavage of cadherins in NRK cells requires MT1-MMP (MMP-14). Am J Physiol Renal Physiol 2006; 290: F43–F51.
Firlej V, Ladam F, Brysbaert G, Dumont P, Fuks F, de Launoit Y et al. Reduced tumorigenesis in mouse mammary cancer cells following inhibition of Pea3- or Erm-dependent transcription. J Cell Sci 2008; 121: 3393–3402.
Tanaka M, Sato H, Takino T, Iwata K, Inoue M, Seiki M . Isolation of a mouse MT2-MMP gene from a lung cDNA library and identification of its product. FEBS Lett 1997; 402: 219–222.
Kobayashi K, Nishioka M, Kohno T, Nakamoto M, Maeshima A, Aoyagi K et al. Identification of genes whose expression is upregulated in lung adenocarcinoma cells in comparison with type II alveolar cells and bronchiolar epithelial cells in vivo. Oncogene 2004; 23: 3089–3096.
Tanaka M, Kuriyama S, Aiba N . Nm23-H1 regulates contact inhibition of locomotion, which is affected by ephrin-B1. J Cell Sci 2012; 125: 4343–4353.
d'Ortho MP, Will H, Atkinson S, Butler G, Messent A, Gavrilovic J et al. Membrane-type matrix metalloproteinases 1 and 2 exhibit broad-spectrum proteolytic capacities comparable to many matrix metalloproteinases. Eur J Biochem 1997; 250: 751–757.
Lettice LA, Williamson I, Wiltshire JH, Peluso S, Devenney PS, Hill AE et al. Opposing functions of the ETS factor family define Shh spatial expression in limb buds and underlie polydactyly. Dev Cell 2012; 22: 459–467.
Zwolinska AK, Heagle Whiting A, Beekman C, Sedivy JM, Marine JC . Suppression of Myc oncogenic activity by nucleostemin haploinsufficiency. Oncogene 2012; 31: 3311–3321.
Satoyoshi R, Kuriyama S, Aiba N, Yashiro M, Tanaka M . Asporin activates coordinated invasion of scirrhous gastric cancer and cancer-associated fibroblasts. Oncogene 2014; 34: 650–660.
Breindel JL, Haskins JW, Cowell EP, Zhao M, Nguyen DX, Stern DF . EGF receptor activates MET through MAPK to enhance non-small cell lung carcinoma invasion and brain metastasis. Cancer Res 2013; 73: 5053–5065.
Gorenne I, Nakamoto RK, Phelps CP, Beckerle MC, Somlyo AV, Somlyo AP . LPP, a LIM protein highly expressed in smooth muscle. Am J Physiol Cell Physiol 2003; 285: C674–C685.
Friedl P, Gilmour D . Collective cell migration in morphogenesis, regeneration and cancer. Nat Rev Mol Cell Biol 2009; 10: 445–457.
Kessenbrock K, Plaks V, Werb Z . Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 2010; 141: 52–67.
Wolf K, Wu YI, Liu Y, Geiger J, Tam E, Overall C et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat Cell Biol 2007; 9: 893–904.
Hegerfeldt Y, Tusch M, Brocker EB, Friedl P . Collective cell movement in primary melanoma explants: plasticity of cell-cell interaction, beta1-integrin function, and migration strategies. Cancer Res 2002; 62: 2125–2130.
Haeger A, Krause M, Wolf K, Friedl P . Cell jamming: collective invasion of mesenchymal tumor cells imposed by tissue confinement. Biochim Biophys Acta 2014; 1840: 2386–2395.
Shih W, Yamada S . N-cadherin-mediated cell-cell adhesion promotes cell migration in a three-dimensional matrix. J Cell Sci 2012; 125: 3661–3670.
Maret D, Gruzglin E, Sadr MS, Siu V, Shan W, Koch AW et al. Surface expression of precursor N-cadherin promotes tumor cell invasion. Neoplasia 2010; 12: 1066–1080.
Gerlinger M, Rowan AJ, Horswell S, Larkin J, Endesfelder D, Gronroos E et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med 2012; 366: 883–892.
Petit M M, Lindskog H, Larsson E, Wasteson P, Athley E, Breuer S et al. Smooth muscle expression of lipoma preferred partner is mediated by an alternative intronic promoter that is regulated by serum response factor/myocardin. Circ Res 2008; 103: 61–69.
Wells A, Yates C, Shepard CR . E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clin Exp Metastasis 2008; 25: 621–628.
Gottlicher M, Minucci S, Zhu P, Kramer OH, Schimpf A, Giavara S et al. Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. EMBO J 2001; 20: 6969–6978.
Phiel CJ, Zhang F, Huang EY, Guenther MG, Lazar MA, Klein PS . Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. J Biol Chem 2001; 276: 36734–36741.
Collisson EA, C J, Brooks AN, Berger AH, Lee W, Chmielecki J et al. Comprehensive molecular profiling of lung adenocarcinoma. Nature 2014; 511: 543–550.
Ikebe D, Wang B, Suzuki H, Kato M . Suppression of keratinocyte stratification by a dominant negative JunB mutant without blocking cell proliferation. Genes Cells 2007; 12: 197–207.
Carey MF, Peterson CL, Smale ST . Chromatin immunoprecipitation (ChIP). Cold Spring Harb Protoc 2009; 114: 124941.
Acknowledgements
We thank Dr Yuichiro Hamanaka for kindly providing the melanoma cell lines. The MNCD2 antibody developed by Takeichi and Matsunami was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of NICHD and maintained by Department of Biology, The University of Iowa, Iowa City, IA, USA. This work is supported by JST KAKENHI (grant number 24700962, SK; grant number 25290042, 26640068, MT) and MEXT KAKENHI (grant number 25111702, SK).
Author Contributions
SK performed the experiments. SY and TK provided the set of lung cancer cell lines. MY, YM and AG made human cancer patient specimens. NA, MT and AG analyzed IHC of patient specimens. SK and MT designed the experiments, and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Kuriyama, S., Yoshida, M., Yano, S. et al. LPP inhibits collective cell migration during lung cancer dissemination. Oncogene 35, 952–964 (2016). https://doi.org/10.1038/onc.2015.155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.155
- Springer Nature Limited