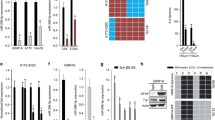
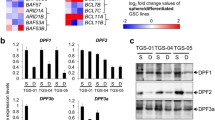
Abstract
Cancer stem-like cells represent poorly differentiated multipotent tumor-propagating cells that contribute disproportionately to therapeutic resistance and tumor recurrence. Transcriptional mechanisms that control the phenotypic conversion of tumor cells lacking tumor-propagating potential to tumor-propagating stem-like cells remain obscure. Here we show that the reprogramming transcription factors Oct4 and Sox2 induce glioblastoma cells to become stem-like and tumor–propagating via a mechanism involving direct DNA methyl transferase (DNMT) promoter transactivation, resulting in global DNA methylation- and DNMT-dependent downregulation of multiple microRNAs (miRNAs). We show that one such downregulated miRNA, miRNA-148a, inhibits glioblastoma cell stem-like properties and tumor-propagating potential. This study identifies a novel and targetable molecular circuit by which glioma cell stemness and tumor-propagating capacity are regulated.







Similar content being viewed by others
References
Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006; 444: 756–760.
Galli R, Binda E, Orfanelli U, Cipelletti B, Gritti A, De Vitis S et al. Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 2004; 64: 7011–7021.
Li Y, Laterra J . Cancer stem cells: distinct entities or dynamically regulated phenotypes? Cancer Res 2012; 72: 576–580.
Takahashi K, Yamanaka S . Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006; 126: 663–676.
Bibikova M, Laurent LC, Ren B, Loring JF, Fan JB . Unraveling epigenetic regulation in embryonic stem cells. Cell Stem Cell 2008; 2: 123–134.
Singh RP, Shiue K, Schomberg D, Zhou FC . Cellular epigenetic modifications of neural stem cell differentiation. Cell Transplant 2009; 18: 1197–1211.
Chiou SH, Wang ML, Chou YT, Chen CJ, Hong CF, Hsieh WJ et al. Coexpression of Oct4 and Nanog enhances malignancy in lung adenocarcinoma by inducing cancer stem cell-like properties and epithelial-mesenchymal transdifferentiation. Cancer Res 2010; 70: 10433–10444.
Li Y, Li A, Glas M, Lal B, Ying M, Sang Y et al. c-Met signaling induces a reprogramming network and supports the glioblastoma stem-like phenotype. Proc Natl Acad Sci USA 2011; 108: 9951–9956.
Rath P, Lal B, Ajala O, Li Y, Xia S, Kim J et al. In vivo c-Met pathway inhibition depletes human glioma xenografts of tumor-propagating stem-like cells. Transl Oncol 2013; 6: 104–111.
Bartel DP . MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116: 281–297.
Lin SL, Chang DC, Chang-Lin S, Lin CH, Wu DT, Chen DT et al. Mir-302 reprograms human skin cancer cells into a pluripotent ES-cell-like state. RNA 2008; 14: 2115–2124.
Li Y, Guessous F, Zhang Y, Dipierro C, Kefas B, Johnson E et al. MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes. Cancer Res 2009; 69: 7569–7576.
Marson A, Levine SS, Cole MF, Frampton GM, Brambrink T, Johnstone S et al. Connecting microRNA genes to the core transcriptional regulatory circuitry of embryonic stem cells. Cell 2008; 134: 521–533.
Miyoshi N, Ishii H, Nagano H, Haraguchi N, Dewi DL, Kano Y et al. Reprogramming of mouse and human cells to pluripotency using mature microRNAs. Cell Stem Cell 2011; 8: 633–638.
Sato F, Tsuchiya S, Meltzer SJ, Shimizu K . MicroRNAs and epigenetics. FEBS J 2011; 278: 1598–1609.
Kim TY, Zhong S, Fields CR, Kim JH, Robertson KD . Epigenomic profiling reveals novel and frequent targets of aberrant DNA methylation-mediated silencing in malignant glioma. Cancer Res 2006; 66: 7490–7501.
Chew JL, Loh YH, Zhang W, Chen X, Tam WL, Yeap LS et al. Reciprocal transcriptional regulation of Pou5f1 and Sox2 via the Oct4/Sox2 complex in embryonic stem cells. Mol Cell Biol 2005; 25: 6031–6046.
Schoenhals M, Kassambara A, De Vos J, Hose D, Moreaux J, Klein B . Embryonic stem cell markers expression in cancers. Biochem Biophys Res Commun 2009; 383: 157–162.
Uchida N, Buck DW, He D, Reitsma MJ, Masek M, Phan TV et al. Direct isolation of human central nervous system stem cells. Proc Natl Acad Sci USA 2000; 97: 14720–14725.
Son MJ, Woolard K, Nam DH, Lee J, Fine HA . SSEA-1 is an enrichment marker for tumor-initiating cells in human glioblastoma. Cell Stem Cell 2009; 4: 440–452.
Piccirillo SG, Combi R, Cajola L, Patrizi A, Redaelli S, Bentivegna A et al. Distinct pools of cancer stem-like cells coexist within human glioblastomas and display different tumorigenicity and independent genomic evolution. Oncogene 2009; 28: 1807–1811.
Glas M, Rath BH, Simon M, Reinartz R, Schramme A, Trageser D et al. Residual tumor cells are unique cellular targets in glioblastoma. Ann Neurol 2010; 68: 264–269.
Stricker SH, Feber A, Engstrom PG, Caren H, Kurian KM, Takashima Y et al. Widespread resetting of DNA methylation in glioblastoma-initiating cells suppresses malignant cellular behavior in a lineage-dependent manner. Genes Dev 2013; 27: 654–669.
Lujambio A, Calin GA, Villanueva A, Ropero S, Sanchez-Cespedes M, Blanco D et al. A microRNA DNA methylation signature for human cancer metastasis. Proc Natl Acad Sci USA 2008; 105: 13556–13561.
Shi Y, Do JT, Desponts C, Hahm HS, Scholer HR, Ding S . A combined chemical and genetic approach for the generation of induced pluripotent stem cells. Cell Stem Cell 2008; 2: 525–528.
Huangfu D, Osafune K, Maehr R, Guo W, Eijkelenboom A, Chen S et al. Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nat Biotechnol 2008; 26: 1269–1275.
Card DA, Hebbar PB, Li L, Trotter KW, Komatsu Y, Mishina Y et al. Oct4/Sox2-regulated miR-302 targets cyclin D1 in human embryonic stem cells. Mol Cell Biol 2008; 28: 6426–6438.
Wang G, Guo X, Hong W, Liu Q, Wei T, Lu C et al. Critical regulation of miR-200/ZEB2 pathway in Oct4/Sox2-induced mesenchymal-to-epithelial transition and induced pluripotent stem cell generation. Proc Natl Acad Sci USA 2013; 110: 2858–2863.
Fowler A, Thomson D, Giles K, Maleki S, Mreich E, Wheeler H et al. miR-124a is frequently down-regulated in glioblastoma and is involved in migration and invasion. Eur J Cancer 2011; 47: 953–963.
Vrba L, Munoz-Rodriguez JL, Stampfer MR, Futscher BW . miRNA gene promoters are frequent targets of aberrant DNA methylation in human breast cancer. PLoS One 2013; 8: e54398.
Zhao WG, Yu SN, Lu ZH, Ma YH, Gu YM, Chen J . The miR-217 microRNA functions as a potential tumor suppressor in pancreatic ductal adenocarcinoma by targeting KRAS. Carcinogenesis 2010; 31: 1726–1733.
Zhang JP, Zeng C, Xu L, Gong J, Fang JH, Zhuang SM . MicroRNA-148a suppresses the epithelial-mesenchymal transition and metastasis of hepatoma cells by targeting Met/Snail signaling. Oncogene 2013; 33: 4069–4076.
Foltz G, Yoon JG, Lee H, Ryken TC, Sibenaller Z, Ehrich M et al. DNA methyltransferase-mediated transcriptional silencing in malignant glioma: a combined whole-genome microarray and promoter array analysis. Oncogene 2009; 28: 2667–2677.
Turcan S, Rohle D, Goenka A, Walsh LA, Fang F, Yilmaz E et al. IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature 2012; 483: 479–483.
Rajendran G, Shanmuganandam K, Bendre A, Muzumdar D, Goel A, Shiras A . Epigenetic regulation of DNA methyltransferases: DNMT1 and DNMT3B in gliomas. J Neuro-Oncol 2011; 104: 483–494.
Fanelli M, Caprodossi S, Ricci-Vitiani L, Porcellini A, Tomassoni-Ardori F, Amatori S et al. Loss of pericentromeric DNA methylation pattern in human glioblastoma is associated with altered DNA methyltransferases expression and involves the stem cell compartment. Oncogene 2008; 27: 358–365.
Orr BA, Haffner MC, Nelson WG, Yegnasubramanian S, Eberhart CG . Decreased 5-hydroxymethylcytosine is associated with neural progenitor phenotype in normal brain and shorter survival in malignant glioma. PLoS ONE 2012; 7: e41036.
Zheng B, Liang L, Wang C, Huang S, Cao X, Zha R et al. MicroRNA-148a suppresses tumor cell invasion and metastasis by downregulating ROCK1 in gastric cancer. Clin Cancer Res 2011; 17: 7574–7583.
Kim J, Zhang Y, Skalski M, Hayes J, Kefas B, Schiff D et al. microRNA-148a is a prognostic oncomiR that targets MIG6 and BIM to regulate EGFR and apoptosis in glioblastoma. Cancer Res 2014; 74: 1541–1553.
Li M, Hale JS, Rich JN, Ransohoff RM, Lathia JD . Chemokine CXCL12 in neurodegenerative diseases: an SOS signal for stem cell-based repair. Trends Neurosci 2012; 35: 619–628.
Gailhouste L, Gomez-Santos L, Hagiwara K, Hatada I, Kitagawa N, Kawaharada K et al. miR-148a plays a pivotal role in the liver by promoting the hepatospecific phenotype and suppressing the invasiveness of transformed cells. Hepatology 2013; 58: 1153–1165.
Guo SL, Peng Z, Yang X, Fan KJ, Ye H, Li ZH et al. miR-148a promoted cell proliferation by targeting p27 in gastric cancer cells. Int J Biol Sci 2011; 7: 567–574.
Watanabe S, Peterson CL . The INO80 family of chromatin-remodeling enzymes: regulators of histone variant dynamics. Cold Spring Harb Symp Quant Biol 2010; 75: 35–42.
Reijntjes S, Francis-West P, Mankoo BS . Retinoic acid is both necessary for and inhibits myogenic commitment and differentiation in the chick limb. Int J Dev Biol 2010; 54: 125–134.
Artner I, Blanchi B, Raum JC, Guo M, Kaneko T, Cordes S et al. MafB is required for islet beta cell maturation. Proc Natl Acad Sci USA 2007; 104: 3853–3858.
Braconi C, Huang N, Patel T . MicroRNA-dependent regulation of DNA methyltransferase-1 and tumor suppressor gene expression by interleukin-6 in human malignant cholangiocytes. Hepatology 2010; 51: 881–890.
Duursma AM, Kedde M, Schrier M, le Sage C, Agami R . miR-148 targets human DNMT3b protein coding region. RNA 2008; 14: 872–877.
Sun P, Xia S, Lal B, Eberhart CG, Quinones-Hinojosa A, Maciaczyk J et al. DNER, an epigenetically modulated gene, regulates glioblastoma-derived neurosphere cell differentiation and tumor propagation. Stem Cells 2009; 27: 1473–1486.
Rohde C, Zhang Y, Reinhardt R, Jeltsch A . BISMA—fast and accurate bisulfite sequencing data analysis of individual clones from unique and repetitive sequences. BMC Bioinform 2010; 11: 230.
Schmittgen TD, Jiang J, Liu Q, Yang L . A high-throughput method to monitor the expression of microRNA precursors. Nucleic Acids Res 2004; 32: e43.
Li Y, Lal B, Kwon S, Fan X, Saldanha U, Reznik TE et al. The scatter factor/hepatocyte growth factor: c-met pathway in human embryonal central nervous system tumor malignancy. Cancer Res 2005; 65: 9355–9362.
Acknowledgements
We thank Daniel Trageser for technical assistance. This work was financially supported by grants from the American Brain Tumor Association (YL), James S McDonnell Foundation (JL), and the United States NIH grants RO1NS073611 (JL) and R01NS070024 (AQ-H).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Lopez-Bertoni, H., Lal, B., Li, A. et al. DNMT-dependent suppression of microRNA regulates the induction of GBM tumor-propagating phenotype by Oct4 and Sox2. Oncogene 34, 3994–4004 (2015). https://doi.org/10.1038/onc.2014.334
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.334
- Springer Nature Limited
This article is cited by
-
Regulation Mechanisms and Maintenance Strategies of Stemness in Mesenchymal Stem Cells
Stem Cell Reviews and Reports (2024)
-
Nucleic acid drug vectors for diagnosis and treatment of brain diseases
Signal Transduction and Targeted Therapy (2023)
-
Silencing of microRNA-135b inhibits invasion, migration, and stemness of CD24+CD44+ pancreatic cancer stem cells through JADE-1-dependent AKT/mTOR pathway
Cancer Cell International (2020)
-
Downregulation of FOXO3a by DNMT1 promotes breast cancer stem cell properties and tumorigenesis
Cell Death & Differentiation (2020)
-
DNMT3b/OCT4 expression confers sorafenib resistance and poor prognosis of hepatocellular carcinoma through IL-6/STAT3 regulation
Journal of Experimental & Clinical Cancer Research (2019)