Key Points
-
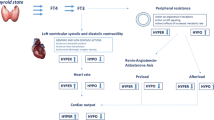
Levothyroxine monotherapy at doses that normalize serum levels of TSH does not universally restore parameters of thyroid hormone levels for patients with hypothyroidism
-
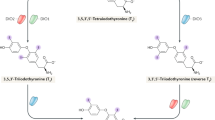
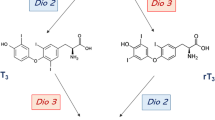
The iodothyronine deiodinases provide a cell-specific, prereceptor mechanism that controls thyroid-hormone signalling
-
Localized thyroid-hormone signalling has a critical role in different areas of the brain, as mediated by thyroid hormone transporters and the iodothyronine deiodinases
-
DIO2 ubiquitination induced by tetraiodothyronine (T4) normally decreases tri-iodothyronine (T3) production, but not in the hypothalamus
-
The levothyroxine dose that normalizes serum levels of TSH in an animal model is lower than the dose that normalizes serum levels of T3, which explains the increased serum T4 to T3 ratio observed in patients treated with levothyroxine
-
If patients carrying the Thr92AlaD2 polymorphism derive benefit from combination therapy with levothyroxine and liothyronine, then genotyping for this single nucleotide polymorphism might become a component of the management of hypothyroidism
Abstract
The coordinated expression and activity of the iodothyronine deiodinases regulate thyroid hormone levels in hypothyroidism. Once heralded as the pathway underpinning adequate thyroid-hormone replacement therapy with levothyroxine, the role of these enzymes has come into question as they have been implicated in both an inability to normalize serum levels of tri-iodothyronine (T3) and the incomplete resolution of hypothyroid symptoms. These observations, some of which were validated in animal models of levothyroxine monotherapy, challenge the paradigm that tissue levels of T3 and thyroid-hormone signalling can be fully restored by administration of levothyroxine alone. The low serum levels of T3 observed among patients receiving levothyroxine monotherapy occur as a consequence of type 2 iodothyronine deiodinase (DIO2) in the hypothalamus being fairly insensitive to ubiquitination. In addition, residual symptoms of hypothyroidism have been linked to a prevalent polymorphism in the DIO2 gene that might be a risk factor for neurodegenerative disease. Here, we discuss how these novel findings underscore the clinical importance of iodothyronine deiodinases in hypothyroidism and how an improved understanding of these enzymes might translate to therapeutic advances in the care of millions of patients with this condition.


Similar content being viewed by others
References
Aoki, Y. et al. Serum TSH and total T4 in the United States population and their association with participant characteristics: National Health and Nutrition Examination Survey (NHANES 1999–2002). Thyroid 17, 1211–1223 (2007).
Vanderpump, M. P. The epidemiology of thyroid disease. Br. Med. Bull. 99, 39–51 (2011).
Lindholm, J. & Laurberg, P. Hypothyroidism and thyroid substitution: historical aspects. J. Thyroid Res. 2011, 809341 (2011).
Braverman, L. E., Ingbar, S. H. & Sterling, K. Conversion of thyroxine (T4) to triiodothyronine (T3) in athyreotic subjects. J. Clin. Invest. 49, 855–864 (1970).
Larsen, P. R. & Ingbar, S. in Textbook of Endocrinology (eds Wilson, J. D. et al.) 357–487 (W. B. Saunders, Co., 1992).
Garber, J. R. et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr. Pract. 18, 988–1028 (2012).
Taylor, S., Kapur, M. & Adie, R. Combined thyroxine and triiodothyronine for thyroid replacement therapy. Br. Med. J. 2, 270–271 (1970).
Roberts, N. D. Psychological problems in thyroid disease. British Thyroid Foundation Newsletter 18, 3 (1996).
Saravanan, P. et al. Psychological well-being in patients on 'adequate' doses of l-thyroxine: results of a large, controlled community-based questionnaire study. Clin. Endocrinol. 57, 577–585 (2002).
Gorman, C. A., Jiang, N. S., Ellefson, R. D. & Elveback, L. R. Comparative effectiveness of dextrothyroxine and levothyroxine in correcting hypothyroidism and lowering blood lipid levels in hypothyroid patients. J. Clin. Endocrinol. Metab. 49, 1–7 (1979).
Franklyn, J. A. et al. Thyroxine replacement therapy and circulating lipid concentrations. Clin. Endocrinol. 38, 453–459 (1993).
Gullo, D. et al. Levothyroxine monotherapy cannot guarantee euthyroidism in all athyreotic patients. PLoS ONE 6, e22552 (2011).
Panicker, V. et al. Common variation in the DIO2 gene predicts baseline psychological well-being and response to combination thyroxine plus triiodothyronine therapy in hypothyroid patients. J. Clin. Endocrinol. Metab. 94, 1623–1629 (2009).
Bunevicius, R., Kazanavicius, G., Zalinkevicius, R. & Prange, A. J. Jr. Effects of thyroxine as compared with thyroxine plus triiodothyronine in patients with hypothyroidism. N. Engl. J. Med. 340, 424–429 (1999).
Escobar-Morreale, H. F. et al. Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann. Intern. Med. 142, 412–424 (2005).
Jonklaas, J. et al. Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid 24, 1670–1751 (2014).
Werneck de Castro, J. P. et al. Differences in hypothalamic type 2 deiodinase ubiquitination explain localized sensitivity to thyroxine. J. Clin. Invest. 125, 769–781 (2015).
Escobar-Morreale, H. F., Obregon, M. J., Escobar del Rey, F. & Morreale de Escobar, G. Replacement therapy for hypothyroidism with thyroxine alone does not ensure euthyroidism in all tissues, as studied in thyroidectomized rats. J. Clin. Invest. 96, 2828–2838 (1995).
Escobar-Morreale, H. F., Rey, F., Obregon, M. J. & Escobar, G. M. Only the combined treatment with thyroxine and triiodothyronine ensures euthyroidism in all tissues of the thyroidectomized rat. Endocrinology 137, 2490–2502 (1996).
Escobar-Morreale, H. F., Obregon, M. J., Hernandez, A., Escobar del Rey, F. & Morreale de Escobar, G. Regulation of iodothyronine deiodinase activity as studied in thyroidectomized rats infused with thyroxine or triiodothyronine. Endocrinology 138, 2559–2568 (1997).
Bianco, A. C. et al. American Thyroid Association guide to investigating thyroid hormone economy and action in rodent and cell models. Thyroid 24, 88–168 (2014).
Gereben, B., Salvatore, D., Harney, J. W., Tu, H. M. & Larsen, P. R. The human, but not rat, Dio2 gene is stimulated by thyroid transcription factor-1 (TTF-1). Mol. Endocrinol. 15, 112–124 (2001).
Salvatore, D., Tu, H., Harney, J. W. & Larsen, P. R. Type 2 iodothyronine deiodinase is highly expressed in human thyroid. J. Clin. Invest. 98, 962–968 (1996).
Campos-Barros, A. et al. Phenolic and tyrosyl ring iodothyronine deiodination and thyroid hormone concentrations in the human central nervous system. J. Clin. Endocrinol. Metab. 81, 2179–2185 (1996).
Visser, T. J., Leonard, J. L., Kaplan, M. M. & Larsen, P. R. Kinetic evidence suggesting two mechanisms for iodothyronine 5′-deiodination in rat cerebral cortex. Proc. Natl Acad. Sci. USA 79, 5080–5084 (1982).
Christoffolete, M. A. et al. Mice with impaired extrathyroidal thyroxine to 3,5,3′-triiodothyronine conversion maintain normal serum 3,5,3′-triiodothyronine concentrations. Endocrinology 148, 954–960 (2007).
Fonseca, T. L. et al. Tissue-specific inactivation of type 2 deiodinase reveals multilevel control of fatty acid oxidation by thyroid hormone in the mouse. Diabetes 63, 1594–1604 (2014).
Fonseca, T. L. et al. Coordination of hypothalamic and pituitary T3 production regulates TSH expression. J. Clin. Invest. 123, 1492–1500 (2013).
Brent, G. A. Mechanisms of thyroid hormone action. J. Clin. Invest. 122, 3035–3043 (2012).
Heuer, H. & Visser, T. J. The pathophysiological consequences of thyroid hormone transporter deficiencies: Insights from mouse models. Biochim. Biophys. Acta 1830, 3974–3978 (2013).
Callebaut, I. et al. The iodothyronine selenodeiodinases are thioredoxin-fold family proteins containing a glycoside hydrolase clan GH-A-like structure. J. Biol. Chem. 278, 36887–36896 (2003).
Schweizer, U., Schlicker, C., Braun, D., Kohrle, J. & Steegborn, C. Crystal structure of mammalian selenocysteine-dependent iodothyronine deiodinase suggests a peroxiredoxin-like catalytic mechanism. Proc. Natl Acad. Sci. USA 111, 10526–10531 (2014).
Zeold, A. et al. Metabolic instability of type 2 deiodinase is transferable to stable proteins independently of subcellular localization. J. Biol. Chem. 281, 31538–31543 (2006).
Baqui, M. M., Gereben, B., Harney, J. W., Larsen, P. R. & Bianco, A. C. Distinct subcellular localization of transiently expressed types 1 and 2 iodothyronine deiodinases as determined by immunofluorescence confocal microscopy. Endocrinology 141, 4309–4312 (2000).
McAninch, E. A. et al. Prevalent polymorphism in thyroid hormone-activating enzyme leaves a genetic fingerprint that underlies associated clinical syndromes. J. Clin. Endocrinol. Metab. 100, 920–933 (2015).
Schneider, M. J. et al. Targeted disruption of the type 1 selenodeiodinase gene (Dio1) results in marked changes in thyroid hormone economy in mice. Endocrinology 147, 580–589 (2006).
Baqui, M. et al. Human type 3 iodothyronine selenodeiodinase is located in the plasma membrane and undergoes rapid internalization to endosomes. J. Biol. Chem. 278, 1206–1211 (2003).
Kallo, I. et al. A novel pathway regulates thyroid hormone availability in rat and human hypothalamic neurosecretory neurons. PLoS ONE 7, e37860 (2012).
Jo, S. et al. Neuronal hypoxia induces hsp40-mediated nuclear import of type 3 deiodinase as an adaptive mechanism to reduce cellular metabolism. J. Neurosci. 32, 8491–8500 (2012).
Gereben, B. et al. Cellular and molecular basis of deiodinase-regulated thyroid hormone signaling. Endocr. Rev. 29, 898–938 (2008).
Bianco, A. C. & McAninch, E. A. The role of thyroid hormone and brown adipose tissue in energy homoeostasis. Lancet Diabetes Endocrinol. 1, 250–258 (2013).
Peeters, R. P. et al. Reduced activation and increased inactivation of thyroid hormone in tissues of critically ill patients. J. Clin. Endocrinol. Metab. 88, 3202–3211 (2003).
Simonides, W. S. et al. Hypoxia-inducible factor induces local thyroid hormone inactivation during hypoxic-ischemic disease in rats. J. Clin. Invest. 118, 975–983 (2008).
Olivares, E. L. et al. Thyroid function disturbance and type 3 iodothyronine deiodinase induction after myocardial infarction in rats a time course study. Endocrinology 148, 4786–4792 (2007).
Bianco, A. C. & Casula, S. Thyroid hormone replacement therapy: three 'simple' questions, complex answers. Eur. Thyroid J. 1, 88–98 (2012).
Dumitrescu, A. M. et al. Mutations in SECISBP2 result in abnormal thyroid hormone metabolism. Nat. Genet. 37, 1247–1252 (2005).
Ord, W. M. Report of the committee of the Clinical Society of London nominated December 14, 1883, to investigate the subject of myxoedema. Trans. Clin. Soc. Lond. 21 (Suppl.), 1–215 (1888).
Oppenheimer, J. H. & Schwartz, H. L. Molecular basis of thyroid hormone-dependent brain development. Endocr. Rev. 18, 462–475 (1997).
Obregon, M. J., Escobar del Rey, F. & Morreale de Escobar, G. The effects of iodine deficiency on thyroid hormone deiodination. Thyroid 15, 917–929 (2005).
Desouza, L. A. et al. Thyroid hormone regulates hippocampal neurogenesis in the adult rat brain. Mol. Cell. Neurosci. 29, 414–426 (2005).
Bernal, J. Thyroid hormone receptors in brain development and function. Nat. Clin. Pract. Endocrinol. Metab. 3, 249–259 (2007).
Morte, B. & Bernal, J. Thyroid hormone action: astrocyte-neuron communication. Front. Endocrinol. 5, 82 (2014).
Greenberg, J. H. et al. Imaging triiodothyronine binding kinetics in rat brain: a model for studies in human subjects. Synapse 60, 212–222 (2006).
Visser, T. J. Thyroid hormone transporters. Horm. Res. 68 (Suppl. 5), 28–30 (2007).
Ceballos, A. et al. Importance of monocarboxylate transporter 8 for the blood-brain barrier-dependent availability of 3,5,3′-triiodo-L-thyronine. Endocrinology 150, 2491–2496 (2009).
Liao, X. H. et al. Distinct roles of deiodinases on the phenotype of Mct8 defect: a comparison of eight different mouse genotypes. Endocrinology 152, 1180–1191 (2011).
Wittmann, G. et al. Parallel regulation of thyroid hormone transporters OATP1c1 and MCT8 during and after endotoxemia at the blood-brain barrier of male rodents. Endocrinology 156, 1552–1564 (2015).
Friesema, E. C. et al. Association between mutations in a thyroid hormone transporter and severe X-linked psychomotor retardation. Lancet 364, 1435–1437 (2004).
Dumitrescu, A. M., Liao, X. H., Best, T. B., Brockmann, K. & Refetoff, S. A novel syndrome combining thyroid and neurological abnormalities is associated with mutations in a monocarboxylate transporter gene. Am. J. Hum. Genet. 74, 168–175 (2004).
Crantz, F. R., Silva, J. E. & Larsen, P. R. Analysis of the sources and quantity of 3,5,3′-triiodothyronine specifically bound to nuclear receptors in rat cerebral cortex and cerebellum. Endocrinology 110, 367–375 (1982).
Galton, V. A. et al. Thyroid hormone homeostasis and action in the type 2 deiodinase-deficient rodent brain during development. Endocrinology 148, 3080–3088 (2007).
Silva, J. E. & Matthews, P. S. Production rates and turnover of triiodothyronine in rat-developing cerebral cortex and cerebellum: responses to hypothyroidism. J. Clin. Invest. 74, 1035–1049 (1984).
Peeters, R. et al. Regional physiological adaptation of the central nervous system deiodinases to iodine deficiency. Am. J. Physiol. Endocrinol. Metab. 281, E54–E61 (2001).
Guadano-Ferraz, A., Obregon, M. J., St Germain, D. L. & Bernal, J. The type 2 iodothyronine deiodinase is expressed primarily in glial cells in the neonatal rat brain. Proc. Natl Acad. Sci. USA 94, 10391–10396 (1997).
Tu, H. M. et al. Regional distribution of type 2 thyroxine deiodinase messenger ribonucleic acid in rat hypothalamus and pituitary and its regulation by thyroid hormone. Endocrinology 138, 3359–3368 (1997).
Mohacsik, P., Zeold, A., Bianco, A. C. & Gereben, B. Thyroid hormone and the neuroglia: both source and target. J. Thyroid Res. 2011, 215718 (2011).
Cahoy, J. D. et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J. Neurosci. 28, 264–278 (2008).
Bernal, J., Guadano-Ferraz, A. & Morte, B. Perspectives in the study of thyroid hormone action on brain development and function. Thyroid 13, 1005–1012 (2003).
Visser, W. E., Friesema, E. C., Jansen, J. & Visser, T. J. Thyroid hormone transport by monocarboxylate transporters. Best Pract. Res. Clin. Endocrinol. Metab. 21, 223–236 (2007).
Freitas, B. C. et al. Paracrine signaling by glial cell-derived triiodothyronine activates neuronal gene expression in the rodent brain and human cells. J. Clin. Invest. 120, 2206–2217 (2010).
Dentice, M. et al. The Hedgehog-inducible ubiquitin ligase subunit WSB-1 modulates thyroid hormone activation and PTHrP secretion in the developing growth plate. Nat. Cell Biol. 7, 698–705 (2005).
Dentice, M. et al. Sonic hedgehog-induced type 3 deiodinase blocks thyroid hormone action enhancing proliferation of normal and malignant keratinocytes. Proc. Natl Acad. Sci. USA 104, 14466–14471 (2007).
Fliers, E., Alkemade, A., Wiersinga, W. M. & Swaab, D. F. Hypothalamic thyroid hormone feedback in health and disease. Prog. Brain Res. 153, 189–207 (2006).
Yoshimura, T. et al. Light-induced hormone conversion of T4 to T3 regulates photoperiodic response of gonads in birds. Nature 426, 178–181 (2003).
Campos-Barros, A. et al. Type 2 iodothyronine deiodinase expression in the cochlea before the onset of hearing. Proc. Natl Acad. Sci. USA 97, 1287–1292 (2000).
Fekete, C. & Lechan, R. M. Central regulation of hypothalamic–pituitary–thyroid axis under physiological and pathophysiological conditions. Endocr. Rev. 35, 159–194 (2014).
Friesema, E. C. et al. Thyroid hormone transporters and deiodinases in the developing human hypothalamus. Eur. J. Endocrinol. 167, 379–386 (2012).
Ng, L. et al. Type 3 deiodinase, a thyroid-hormone-inactivating enzyme, controls survival and maturation of cone photoreceptors. J. Neurosci. 30, 3347–3357 (2010).
Kester, M. H. et al. Iodothyronine levels in the human developing brain: major regulatory roles of iodothyronine deiodinases in different areas. J. Clin. Endocrinol. Metab. 89, 3117–3128 (2004).
Vose, L. R. et al. Treatment with thyroxine restores myelination and clinical recovery after intraventricular hemorrhage. J. Neurosci. 33, 17232–17246 (2013).
Zavacki, A. M. et al. Type 1 iodothyronine deiodinase is a sensitive marker of peripheral thyroid status in the mouse. Endocrinology 146, 1568–1575 (2005).
Bianco, A. C., Salvatore, D., Gereben, B., Berry, M. J. & Larsen, P. R. Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocr. Rev. 23, 38–89 (2002).
Roti, E., Fang, S. L., Green, K., Emerson, C. H. & Braverman, L. E. Human placenta is an active site of thyroxine and 3,3′,5-triiodothyronine tyrosyl ring deiodination. J. Clin. Endocrinol. Metab. 53, 498–501 (1981).
Alexander, E. K. et al. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N. Engl. J. Med. 351, 241–249 (2004).
Huang, S. A. et al. Severe hypothyroidism caused by type 3 iodothyronine deiodinase in infantile hemangiomas. N. Engl. J. Med. 343, 185–189 (2000).
Abdalla, S. M. & Bianco, A. C. Defending plasma T3 is a biological priority. Clin. Endocrinol. 81, 633–641 (2014).
Bianco, A. C. & Kim, B. W. Deiodinases: implications of the local control of thyroid hormone action. J. Clin. Invest. 116, 2571–2579 (2006).
Steinsapir, J., Harney, J. & Larsen, P. R. Type 2 iodothyronine deiodinase in rat pituitary tumor cells is inactivated in proteasomes. J. Clin. Invest. 102, 1895–1899 (1998).
Gereben, B., Goncalves, C., Harney, J. W., Larsen, P. R. & Bianco, A. C. Selective proteolysis of human type 2 deiodinase: a novel ubiquitin-proteasomal mediated mechanism for regulation of hormone activation. Mol. Endocrinol. 14, 1697–1708 (2000).
Sagar, G. D. et al. Ubiquitination-induced conformational change within the deiodinase dimer is a switch regulating enzyme activity. Mol. Cell. Biol. 27, 4774–4783 (2007).
Egri, P. & Gereben, B. Minimal requirements for ubiquitination-mediated regulation of thyroid hormone activation. J. Mol. Endocrinol. 53, 217–226 (2014).
Curcio-Morelli, C. et al. In vivo dimerization of types 1, 2, and 3 iodothyronine selenodeiodinases. Endocrinology 144, 937–946 (2003).
Arrojo, E. D. R., Egri, P., Jo, S., Gereben, B. & Bianco, A. C. The type II deiodinase is retrotranslocated to the cytoplasm and proteasomes via p97/Atx3 complex. Mol. Endocrinol. 27, 2105–2115 (2013).
Curcio-Morelli, C. et al. Deubiquitination of type 2 iodothyronine deiodinase by von Hippel-Lindau protein-interacting deubiquitinating enzymes regulates thyroid hormone activation. J. Clin. Invest. 112, 189–196 (2003).
Christoffolete, M. A. et al. Atypical expression of type 2 iodothyronine deiodinase in thyrotrophs explains the thyroxine-mediated pituitary thyrotropin feedback mechanism. Endocrinology 147, 1735–1743 (2006).
Schneider, M. J. et al. Targeted disruption of the type 2 selenodeiodinase gene (DIO2) results in a phenotype of pituitary resistance to T4 . Mol. Endocrinol. 15, 2137–2148 (2001).
Fekete, C. & Lechan, R. M. Negative feedback regulation of hypophysiotropic thyrotropin-releasing hormone (TRH) synthesizing neurons: role of neuronal afferents and type 2 deiodinase. Front. Neuroendocrinol. 28, 97–114 (2007).
Boelen, A. et al. Simultaneous changes in central and peripheral components of the hypothalamus–pituitary–thyroid axis in lipopolysaccharide-induced acute illness in mice. J. Endocrinol. 182, 315–323 (2004).
Fekete, C. et al. Lipopolysaccharide induces type 2 iodothyronine deiodinase in the mediobasal hypothalamus: implications for the nonthyroidal illness syndrome. Endocrinology 145, 1649–1655 (2004).
Sawin, C. T., Surks, M. I., London, M., Ranganathan, C. & Larsen, P. R. Oral thyroxine: variation in biologic action and tablet content. Ann. Intern. Med. 100, 641–645 (1984).
Ito, M. et al. TSH-suppressive doses of levothyroxine are required to achieve preoperative native serum triiodothyronine levels in patients who have undergone total thyroidectomy. Eur. J. Endocrinol. 167, 373–378 (2012).
Wiersinga, W. M., Duntas, L., Fadeyev, V., Nygaard, B. & Vanderpump, M. P. 2012 ETA Guidelines: The use of L-T4 + L-T3 in the treatment of hypothyroidism. Eur. Thyroid J. 1, 55–71 (2012).
Williams, G. R. Neurodevelopmental and neurophysiological actions of thyroid hormone. J. Neuroendocrinol. 20, 784–794 (2008).
Ritchie, M. & Yeap, B. B. Thyroid hormone: influences on mood and cognition in adults. Maturitas 81, 266–275 (2015).
Parsaik, A. K. et al. Hypothyroidism and risk of mild cognitive impairment in elderly persons: a population-based study. JAMA Neurol. 71, 201–207 (2014).
Wekking, E. M. et al. Cognitive functioning and well-being in euthyroid patients on thyroxine replacement therapy for primary hypothyroidism. Eur. J. Endocrinol. 153, 747–753 (2005).
Nasr, A., Lauterio, T. J. & Davis, M. W. Unapproved drugs in the United States and the Food and Drug Administration. Adv. Ther. 28, 842–856 (2011).
Markova, N. et al. Hippocampal gene expression of deiodinases 2 and 3 and effects of 3,5-diiodo-L-thyronine T2 in mouse depression paradigms. Biomed. Res. Int. 2013, 565218 (2013).
de Jong, F. J. et al. Thyroid function, the risk of dementia and neuropathologic changes: the Honolulu-Asia aging study. Neurobiol. Aging 30, 600–606 (2009).
Bauer, M., Heinz, A. & Whybrow, P. C. Thyroid hormones, serotonin and mood: of synergy and significance in the adult brain. Mol. Psychiatry 7, 140–156 (2002).
Morrissette, D. A. & Stahl, S. M. Modulating the serotonin system in the treatment of major depressive disorder. CNS Spectr. 19 (Suppl. 1), 57–67 (2014).
Zhang, Q. et al. Monitoring glutamate levels in the posterior cingulate cortex of thyroid dysfunction patients with TE-averaged PRESS at 3 T. Magn. Reson. Imaging 33, 774–778 (2015).
Schreckenberger, M. F. et al. Positron emission tomography reveals correlations between brain metabolism and mood changes in hyperthyroidism. J. Clin. Endocrinol. Metab. 91, 4786–4791 (2006).
Hernandez, A., Morte, B., Belinchon, M. M., Ceballos, A. & Bernal, J. Critical role of types 2 and 3 deiodinases in the negative regulation of gene expression by T3 in the mouse cerebral cortex. Endocrinology 153, 2919–2928 (2012).
Eravci, M., Pinna, G., Meinhold, H. & Baumgartner, A. Effects of pharmacological and nonpharmacological treatments on thyroid hormone metabolism and concentrations in rat brain. Endocrinology 141, 1027–1040 (2000).
Campos-Barros, A. et al. The influence of desipramine on thyroid hormone metabolism in rat brain. J Pharmacol. Exp. Ther. 268, 1143–1152 (1994).
Baumgartner, A., Dubeyko, M., Campos-Barros, A., Eravci, M. & Meinhold, H. Subchronic administration of fluoxetine to rats affects triiodothyronine production and deiodination in regions of the cortex and in the limbic forebrain. Brain Res. 635, 68–74 (1994).
Barez-Lopez, S. et al. Abnormal motor phenotype at adult stages in mice lacking type 2 deiodinase. PLoS ONE 9, e103857 (2014).
Hernandez, A., Martinez, M. E., Fiering, S., Galton, V. A. & St Germain, D. Type 3 deiodinase is critical for the maturation and function of the thyroid axis. J. Clin. Invest. 116, 476–484 (2006).
Sittig, L. J., Shukla, P. K., Herzing, L. B. & Redei, E. E. Strain-specific vulnerability to alcohol exposure in utero via hippocampal parent-of-origin expression of deiodinase-III. FASEB J. 25, 2313–2324 (2011).
Wilcoxon, J. S., Kuo, A. G., Disterhoft, J. F. & Redei, E. E. Behavioral deficits associated with fetal alcohol exposure are reversed by prenatal thyroid hormone treatment: a role for maternal thyroid hormone deficiency in FAE. Mol. Psychiatry 10, 961–971 (2005).
Wilcoxon, J. S. & Redei, E. E. Prenatal programming of adult thyroid function by alcohol and thyroid hormones. Am. J. Physiol. Endocrinol. Metab. 287, E318–E326 (2004).
Philibert, R. A. et al. The relationship of deiodinase 1 genotype and thyroid function to lifetime history of major depression in three independent populations. Am. J. Med. Genet. B Neuropsychiatr. Genet. 156B, 593–599 (2011).
Cooper-Kazaz, R. et al. Preliminary evidence that a functional polymorphism in type 1 deiodinase is associated with enhanced potentiation of the antidepressant effect of sertraline by triiodothyronine. J. Affect. Disord. 116, 113–116 (2009).
Mentuccia, D. et al. Association between a novel variant of the human type 2 deiodinase gene Thr92Ala and insulin resistance: evidence of interaction with the Trp64Arg variant of the β-3-adrenergic receptor. Diabetes 51, 880–883 (2002).
Estivalet, A. A. et al. D2 Thr92Ala and PPARγ2 Pro12Ala polymorphisms interact in the modulation of insulin resistance in type 2 diabetic patients. Obesity (Silver Spring) 19, 825–832 (2010).
Dora, J. M., Machado, W. E., Rheinheimer, J., Crispim, D. & Maia, A. L. Association of the type 2 deiodinase Thr92Ala polymorphism with type 2 diabetes: case-control study and meta-analysis. Eur. J. Endocrinol. 163, 427–434 (2010).
Guo, T. W. et al. Positive association of the DIO2 (deiodinase type 2) gene with mental retardation in the iodine-deficient areas of China. J. Med. Genet. 41, 585–590 (2004).
He, B. et al. Association of genetic polymorphisms in the type II deiodinase gene with bipolar disorder in a subset of Chinese population. Prog. Neuropsychopharmacol. Biol. Psychiatry 33, 986–990 (2009).
Taylor, P. et al. Effect of low thyroid hormone bioavailability on childhood cognitive development: data from the Avon Longitudinal Study of Parents and Children birth cohort. Lancet 383, S100 (2014).
Galecka, E. et al. Association of the DIO2 gene single nucleotide polymorphisms with recurrent depressive disorder. Acta Biochim. Pol. 62, 297–302 (2015).
Canani, L. H. et al. The type 2 deiodinase A/G (Thr92Ala) polymorphism is associated with decreased enzyme velocity and increased insulin resistance in patients with type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 90, 3472–3478 (2005).
Peeters, R. P. et al. Polymorphisms in thyroid hormone pathway genes are associated with plasma TSH and iodothyronine levels in healthy subjects. J. Clin. Endocrinol. Metab. 88, 2880–2888 (2003).
Torlontano, M. et al. Type 2 deiodinase polymorphism (threonine 92 alanine) predicts L-thyroxine dose to achieve target thyrotropin levels in thyroidectomized patients. J. Clin. Endocrinol. Metab. 93, 910–913 (2008).
Butler, P. W. et al. The Thr92Ala 5′ type 2 deiodinase gene polymorphism is associated with a delayed triiodothyronine secretion in response to the thyrotropin-releasing hormone-stimulation test: a pharmacogenomic study. Thyroid 20, 1407–1412 (2011).
Zhang, C., Kim, S., Harney, J. W. & Larsen, P. R. Further characterization of thyroid hormone response elements in the human type 1 iodothyronine deiodinase gene. Endocrinology 139, 1156–1163 (1998).
Acknowledgements
B.G. is supported by the Hungarian Brain Research Program and Hungarian Scientific Research Fund (OTKA 109415). M.O.R. is supported by CAPES and FAPESP. A.C.B. is supported by the NIDDK.
Author information
Authors and Affiliations
Contributions
All authors contributed to researching the data for the article and the writing the article. B.G., E.A.M. and A.C.B. provided a substantial contribution to discussions of the content and reviewed and/or edited the manuscript before submission. B.G. and E.A.M. contributed equally to this Review.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Gereben, B., McAninch, E., Ribeiro, M. et al. Scope and limitations of iodothyronine deiodinases in hypothyroidism. Nat Rev Endocrinol 11, 642–652 (2015). https://doi.org/10.1038/nrendo.2015.155
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2015.155
- Springer Nature Limited
This article is cited by
-
Pharmacodynamic and pharmacokinetic properties of the combined preparation of levothyroxine plus sustained- release liothyronine; a randomized controlled clinical trial
BMC Endocrine Disorders (2023)
-
Primary hypothyroidism and quality of life
Nature Reviews Endocrinology (2022)
-
Developmental changes of the fetal and neonatal thyroid gland and functional consequences on the cardiovascular system
Journal of Perinatology (2022)
-
Plasticizers: negative impacts on the thyroid hormone system
Environmental Science and Pollution Research (2022)
-
Thyroid hormone: sex-dependent role in nervous system regulation and disease
Biology of Sex Differences (2021)