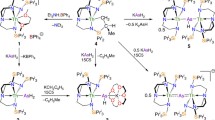
Abstract
To further our fundamental understanding of the nature and extent of covalency in uranium–ligand bonding, and the benefits that this may have for the design of new ligands for nuclear waste separation, there is burgeoning interest in the nature of uranium complexes with soft- and multiple-bond-donor ligands. Despite this, there have so far been no examples of structurally authenticated molecular uranium–arsenic bonds under ambient conditions. Here, we report molecular uranium(IV)–arsenic complexes featuring formal single, double and triple U–As bonding interactions. Compound formulations are supported by a range of characterization techniques, and theoretical calculations suggest the presence of polarized covalent one-, two- and threefold bonding interactions between uranium and arsenic in parent arsenide [U–AsH2], terminal arsinidene [U=AsH] and arsenido [U≡AsK2] complexes, respectively. These studies inform our understanding of the bonding of actinides with soft donor ligands and may be of use in future ligand design in this area.

Similar content being viewed by others
References
Dam, H. H., Reinhoudt, D. N. & Verboom, W. Multicoordinate ligands for actinide/lanthanide separations. Chem. Soc. Rev. 36, 367–377 (2007).
Jones, M. B. & Gaunt, A. J. Recent developments in synthesis and structural chemistry of nonaqueous actinide complexes. Chem. Rev. 113, 1137–1198 (2013).
Kozimor, S. A. et al. Trends in covalency for d- and f-element metallocene dichlorides identified using chlorine K-edge X-ray absorption spectroscopy and time-dependent density functional theory. J. Am. Chem. Soc. 131, 12125–12136 (2009).
Seaman, L. A. et al. Probing the 5f orbital contribution to the bonding in a U(V) ketimide complex. J. Am. Chem. Soc. 134, 4931–4940 (2012).
Minasian, S. G. et al. Determining relative f and d orbital contributions to M–Cl covalency in MCl62− (M = Ti, Zr, Hf, U) and UOCl5− using Cl K-edge X-ray absorption spectroscopy and time-dependent density functional theory. J. Am. Chem. Soc. 134, 5586–5597 (2012).
Spencer, L. P. et al. Tetrahalide complexes of the [U(NR)2]2+ ion: synthesis, theory, and chlorine K-edge X-ray absorption spectroscopy. J. Am. Chem. Soc. 135, 2279–2290 (2013).
Lukens, W. W. et al. Quantifying the σ and π interactions between U(V) f orbitals and halide, alkyl, alkoxide, amide, and ketimide ligands. J. Am. Chem. Soc. 135, 10742–10754 (2013).
Jensen, M. P. & Bond, A. H. Comparison of covalency in the complexes of trivalent actinide and lanthanide cations. J. Am. Chem. Soc. 124, 9870–9877 (2002).
Miguirditchian, M. et al. Thermodynamic study of the complexation of trivalent actinide and lanthanide cations by ADPTZ, a tridentate N-donor ligand. Inorg. Chem. 44, 1404–1412 (2005).
Gaunt, A. J. et al. Experimental and theoretical comparison of actinide and lanthanide bonding in M[N(EPR2)2]3 complexes (M = U, Pu, La, Ce; E = S, Se, Te; R = Ph, iPr, H). Inorg. Chem. 47, 29–41 (2007).
Ingram, K. I. M., Tassell, M. J., Gaunt, A. J. & Kaltsoyannis, N. Covalency in the f element–chalcogen bond. Computational studies of M[N(EPR2)2]3 (M = La, Ce, Pr, Pm, Eu, U, Np, Pu, Am, Cm; E = O, S, Se, Te; R = H, iPr, Ph). Inorg. Chem. 47, 7824–7833 (2008).
Jones, M. B. et al. Uncovering f-element bonding differences and electronic structure in a series of 1:3 and 1:4 complexes with a diselenophosphinate ligand. Chem. Sci. 4, 1189–1203 (2013).
Ephritikhine, M. The vitality of uranium molecular chemistry at the dawn of the XXIst century. Dalton Trans. 2006, 2501–2516 (2006).
Hayton, T. W. Metal-ligand multiple bonding in uranium: structure and reactivity. Dalton Trans. 39, 1145–1158 (2010).
Hayton, T. W. Recent developments in actinide-ligand multiple bonding. Chem. Commun. 49, 2956–2973 (2013).
Allen, F. H. The Cambridge Structural Database: a quarter of a million crystal structures and rising. Acta Cryst. Sect. B 58, 380 (2002).
Cantat, T. et al. The U=C double bond: synthesis and study of uranium nucleophilic carbene complexes. J. Am. Chem. Soc. 131, 963–972 (2009).
Cooper, O. J. et al. Uranium–carbon multiple bonding: facile access to the pentavalent uranium carbene [U{C(PPh2NSiMe3)2}(Cl)2(I)] and comparison of UV=C and UIV=C double bonds. Angew. Chem. Int. Ed. 50, 2383–2386 (2011).
Tourneux, J-C. et al. Exploring the uranyl organometallic chemistry: from single to double uranium–carbon bonds. J. Am. Chem. Soc. 133, 6162–6165 (2011).
Fortier, S., Walensky, J. R., Wu, G. & Hayton, T. W. Synthesis of a phosphorano-stabilized U(IV)-carbene via one-electron oxidation of a U(III)-ylide adduct. J. Am. Chem. Soc. 133, 6894–6897 (2011).
Mills, D. P. et al. Synthesis of a uranium(VI)-carbene: reductive formation of uranyl(V)-methanides, oxidative preparation of a [R2C=U=O]2+ analogue of the [O=U=O]2+ uranyl ion (R = Ph2PNSiMe3), and comparison of the nature of UIV=C, UV=C and UVI=C double bonds. J. Am. Chem. Soc. 134, 10047–10054 (2012).
Lu, E. et al. Synthesis, characterization, and reactivity of a uranium(VI) carbene imido oxo complex. Angew. Chem. Int. Ed. 53, 6696–6700 (2014).
Cramer, R. E., Panchanatheswaran, K. & Gilje, J. W. Uranium carbon multiple-bond chemistry. 3. Insertion of acetonitrile and the formation of a uranium nitrogen multiple bond. J. Am. Chem. Soc. 106, 1853–1854 (1984).
Brennan, J. G. & Andersen, R. A. Electron-transfer reactions of trivalent uranium. Preparation and structure of the uranium metallocene compounds (MeC5H4)3U=NPh and [(MeC5H4)3U]2[μ-η1,η2-PhNCO]. J. Am. Chem. Soc. 107, 514–516 (1985).
Burns, C. J., Smith, W. H., Huffman, J. C. & Sattelberger, A. P. Uranium(VI) organoimido complexes. J. Am. Chem. Soc. 112, 3237–3239 (1990).
Arney, D. S. J., Burns, C. J. & Smith, D. C. Synthesis and structure of the first uranium(VI) organometallic complex. J. Am. Chem. Soc. 114, 10068–10069 (1992).
Arney, D. S. J. & Burns, C. J. Synthesis and properties of high-valent organouranium complexes containing terminal organoimido and oxo functional groups. A new class of organo-f-element complexes. J. Am. Chem. Soc. 117, 9448–9460 (1995).
Hayton, T. W. et al. Synthesis of imido analogs of the uranyl ion. Science 310, 1941 (2005).
Castro-Rodríguez, I., Nakai, H. & Meyer, K. Multiple-bond metathesis mediated by sterically pressured uranium complexes. Angew. Chem. Int. Ed. 45, 2389–2392 (2006).
Graves, C. R. et al. Organometallic uranium(V)-imido halide complexes from synthesis to electronic structure and bonding. J. Am. Chem. Soc. 130, 5272–5285 (2008).
Bart, S. C. et al. Carbon dioxide activation with sterically pressured mid- and high-valent uranium complexes. J. Am. Chem. Soc. 130, 12536–12546 (2008).
Matson, E. M., Crestani, M. G., Fanwick, P. E. & Bart, S. C. Synthesis of U(IV) imidos from Tp*2U(CH2Ph) (Tp* = hydrotris(3,5-dimethylpyrazolyl)borate) by extrusion of bibenzyl. Dalton Trans. 41, 7952–7958 (2012).
Jilek, R. E. et al. A direct route to bis(imido) uranium(V) halides via metathesis of uranium tetrachloride. J. Am. Chem. Soc. 134, 9876–9878 (2012).
Camp, C., Pécaut, J. & Mazzanti, M. Tuning uranium–nitrogen multiple bond formation with ancillary siloxide ligands. J. Am. Chem. Soc. 135, 12101–12111 (2013).
Lam, O. P. et al. Observation of the inverse trans influence (ITI) in a uranium(V) imide coordination complex: an experimental study and theoretical evaluation. Inorg. Chem. 51, 6190–6199 (2012).
Anderson, N. H. et al. Harnessing redox activity for the formation of uranium tris(imido) compounds. Nature Chem. 6, 919–926 (2014).
King, D. M. et al. Synthesis and characterization of an f-block terminal parent imido [U=NH] complex a masked uranium(IV) nitride. J. Am. Chem. Soc. 136, 5619–5622 (2014).
King, D. M. et al. Synthesis and structure of a terminal uranium nitride complex. Science 337, 717–720 (2012).
King, D. M. et al. Isolation and characterization of a uranium(VI)-nitride triple bond. Nature Chem. 5, 482–488 (2013).
Cleaves, P. A. et al. Two-electron reductive carbonylation of terminal uranium(V) and uranium(VI) nitrides to cyanate by carbon monoxide. Angew. Chem. Int. Ed. 53, 10412–10415 (2014).
Duttera, M. R., Day, V. W. & Marks, T. J. Organoactinide phosphine/phosphite coordination chemistry. Facile hydride-induced dealkoxylation and the formation of actinide phosphinidene complexes. J. Am. Chem. Soc. 106, 2907–2912 (1984).
Arney, D. S. J., Schnabel, R. C., Scott, B. C. & Burns, C. J. Preparation of actinide phosphinidene complexes: steric control of reactivity. J. Am. Chem. Soc. 118, 6780–6781 (1996).
Gardner, B. M. et al. Triamidoamine-uranium(IV)-stabilized terminal parent phosphide and phosphinidene complexes. Angew. Chem. Int. Ed. 53, 4484–4488 (2014).
Brown, J. L., Fortier, S., Lewis, R. A., Wu, G. & Hayton, T. W. A complete family of terminal uranium chalcogenides, [U(E)(N{SiMe3}2)3]− (E = O, S, Se, Te). J. Am. Chem. Soc. 134, 15468–15475 (2012).
Brown, J. L., Fortier, S., Wu, G., Kaltsoyannis, N. & Hayton, T. W. Synthesis and spectroscopic and computational characterization of the chalcogenido-substituted analogues of the uranyl ion, [OUE]2+ (E = S, Se). J. Am. Chem. Soc. 135, 5352–5355 (2013).
Scherer, O. J., Werner, B., Heckmann, G. & Wolmershäuser, G. Bicyclic P6 as complex ligand. Angew. Chem. Int. Ed. 30, 553–555 (1991).
Stephens, F. H. Activation of White Phosphorus by Molybdenum- and Uranium Tris-Amides PhD thesis, Massachusetts Institute of Technology (2004).
Frey, A. S. P., Cloke, F. G. N., Hitchcock, P. B. & Green, J. C. Activation of P4 by U(η5-C5Me5)(η8-C8H6(SiiPr3)2-1,4)(THF); the X-ray structure of [U(η5-C5Me5)(η8-C8H6(SiiPr3)2-1,4)]2(μ-η2:η2-P4). New. J. Chem. 35, 2022–2026 (2011).
Patel, D. et al. An actinide-zintl cluster: a tris(triamidouranium)μ3-η2:η2:η2-heptaphosphanortricyclane and its diverse synthetic utility. Angew. Chem. Int. Ed. 52, 13334–13337 (2013).
Selbin, J., Ahmad, N. & Pribble, M. J. Novel complexes of uranium(V). J. Chem. Soc. Chem. Commun. 759–760 (1969).
Deutscher, R. L. & Kepert, D. L. Eight-coordinate complexes of niobium, tantalum, and uranium tetrahalides. Inorg. Chem. 9, 2305–2310 (1970).
Scherer, O. J., Schulze, J. & Wolmershäuser, G. Bicyclisches As6 als komplexligand. J. Organomet. Chem. 484, C5–C7 (1994).
Wu, Q-Y. et al. Terminal U≡E (E = N, P, As, Sb, and Bi) bonds in uranium complexes: a theoretical perspective. J. Phys. Chem. A 119, 922–930 (2015).
Andrews, L., Wang, X., Lindh, R., Roos, B. O. & Marsden, C. J. Simple N≡UF3 and P≡UF3 molecules with triple bonds to uranium. Angew. Chem. Int. Ed. 47, 5366–5370 (2008).
Andrews, L., Wang, X. & Roos, B. O. As≡UF3 molecule with a weak triple bond to uranium. Inorg. Chem. 48, 6594–6598 (2009).
Ebsworth, E. A. V., Gould, R. O., Mayo, R. A. & Walkinshaw, M. Reactions of phosphine, arsine, and stibene with carbonylbis(triethylphosphine)iridium(I) halides. Part 1. Reactions in toluene; X-ray crystal; structures of [Ir(CO)ClH(PEt3)2(AsH2)] and [Ir(CO)XH(PEt3)2(μ-ZH2)RuCl2(η6-MeC6H4CHMe2-p)] (X = Br, Z = P; X = Cl, Z = As). J. Chem. Soc. Dalton Trans. 1987, 2831–2838 (1987).
Becker, G. et al. Metal derivatives of molecular compounds. IX Bis(1,2-dimethoxyethane-O,O′)lithium phosphanide, arsanide, and chloride—three new representations of the bis(1,2-dimethoxyethane-O,O')lithium bromide type. Z. Anorg. Allg. Chem. 624, 469–482 (1998).
Dixon, R. N., Duxbury, G. & Lamberton, H. M. Arsenic hydride radicals. Chem. Commun. 460–461 (1966).
Herrmann, W. A., Koumbouris, B., Zahn, T. & Ziegler, M. L. Arsanediyl (arsinidene) and diarsene complexes by metal-induced degradation of monoarsane. Angew. Chem. Int. Ed. Engl. 23, 812–814 (1984).
Bachman, R. E., Miller, S. K. & Whitmire, K. H. Synthesis and structure of an anionic arsenic hydride complex: [PPN]2[HAs{Fe(CO)4}3]•0.5THF. Inorg. Chem. 33, 2075–2076 (1994).
Präsang, C., Stoelzel, M., Inoue, S., Meltzer, A. & Driess, M. Metal-free activation of EH3 (E = P, As) by an ylide-like silylene and formation of a donor-stabilized arsasilene with a HSi=AsH subunit. Angew. Chem. Int. Ed. 49, 10002–10005 (2010).
Johnson, B. P., Balázs, G. & Scheer, M. Low-coordinate E1 ligand complexes of group 15 elements—a developing area. Coord. Chem. Rev. 250, 1178–1195 (2006).
Spinney, H. A., Piro, N. A. & Cummins, C. C. Triple-bond reactivity of an AsP complex intermediate: synthesis stemming from molecular arsenic, As4 . J. Am. Chem. Soc. 131, 16233–16243 (2009).
Curley, J. J., Piro, N. A. & Cummins, C. C. A terminal molybdenum arsenide complex synthesized from yellow arsenic. Inorg. Chem. 48, 9599–9601 (2009).
Johnson, W. C. & Pechukas, A. Hydrogen compounds of arsenic. II. Sodium and potassium dihydrogen arsenides. J. Am. Chem. Soc. 59, 2068–2071 (1937).
Peters, J. C., Odom, A. L. & Cummins, C. C. A terminal molybdenum carbide prepared by methylidyne deprotonation. Chem. Commun. 1995–1996 (1997).
Gardner, B. M. et al. The role of 5f-orbital participation in unexpected inversion of the σ-bond metathesis reactivity trend of triamidoamine thorium(IV) and uranium(IV) alkyls. Chem. Sci. 5, 2489–2497 (2014).
Pyykkö, P. & Atsumi, M. Molecular single-bond covalent radii for elements 1–118. Chem. Eur. J. 15, 186–197 (2009).
Pyykkö, P. & Atsumi, M. Molecular double-bond covalent radii for elements Li–E112. Chem. Eur. J. 15, 12770–12779 (2009).
Pyykkö, P., Riedel, S. & Patzschke, M. Triple-bond covalent radii. Chem. Eur. J. 11, 3511–3520 (2005).
Kindra, D. R. & Evans, W. J. Magnetic susceptibility of uranium complexes. Chem. Rev. 114, 8865–8882 (2014).
Bader, R. F. W., Slee, T. S., Cremer, D. & Kraka, E. Descriptions of conjugation and hyperconjugation in terms of electron distributions. J. Am. Chem. Soc. 105, 5061–5068 (1983).
Halter, D. P., La Pierre, H. S., Heinemann, F. W. & Meyer, K. Uranium(IV) halide (F−, Cl−, Br−, and I−) monoarene complexes. Inorg. Chem. 53, 8418–8424 (2014).
Patel, D., McMaster, J., Lewis, W., Blake, A. J. & Liddle, S. T. Reductive assembly of cyclobutadienyl and diphosphacyclobutadienyl rings at uranium. Nature Commun. 4, 2323 (2013).
Acknowledgements
The authors acknowledge the Royal Society, the European Research Council, the Engineering and Physical Sciences Research Council, the Universities of Nottingham, Manchester and Regensburg, the Deutsche Forschungsgemeinschaft, the UK National Nuclear Laboratory, COST Action CM1006 and the EPSRC UK National EPR Facility for supporting this work.
Author information
Authors and Affiliations
Contributions
B.M.G. synthesized and characterized the compounds. G.B. prepared the parent potassium arsenide complex. F.T. and E.J.L.M. recorded and interpreted the magnetic data. J.M. and S.T.L. conducted and analysed the theoretical calculations. W.L. and A.J.B. carried out the X-ray single-crystal diffraction work. S.T.L. and M.S. originated the central idea, supervised the work, analysed the data and wrote the manuscript with contributions from all co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 5407 kb)
Supplementary information
First set of crystallographic data for compound 2c (CIF 29 kb)
Supplementary information
Second set of crystallographic data for compound 2c (CIF 55 kb)
Supplementary information
Crystallographic data for compound 3c (CIF 84 kb)
Supplementary information
Crystallographic data for compound 4 (CIF 116 kb)
Supplementary information
Crystallographic data for compound 5 (CIF 112 kb)
Supplementary information
Crystallographic data for compound 7 (CIF 81 kb)
Rights and permissions
About this article
Cite this article
Gardner, B., Balázs, G., Scheer, M. et al. Triamidoamine uranium(IV)–arsenic complexes containing one-, two- and threefold U–As bonding interactions. Nature Chem 7, 582–590 (2015). https://doi.org/10.1038/nchem.2279
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2279
- Springer Nature Limited
This article is cited by
-
Evidence for ligand- and solvent-induced disproportionation of uranium(IV)
Nature Communications (2021)
-
Complexes featuring a linear [N≡U≡N] core isoelectronic to the uranyl cation
Nature Chemistry (2020)
-
Terminal uranium(V)-nitride hydrogenations involving direct addition or Frustrated Lewis Pair mechanisms
Nature Communications (2020)
-
Thorium-nitrogen multiple bonds provide evidence for pushing-from-below for early actinides
Nature Communications (2019)
-
A diuranium carbide cluster stabilized inside a C80 fullerene cage
Nature Communications (2018)