Abstract
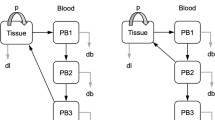
Chronic lymphocytic leukemia (CLL) is a progressive malignancy of mature B-cells that involves the peripheral blood (PB), lymph nodes (LNs) and bone marrow (BM). Although the majority of CLL cells are in a resting state, small populations of proliferating cells exist; however, the anatomical site of active cell proliferation remains to be definitively determined. Based on findings that CLL cells in LNs have increased expression of B-cell activation genes, we tested the hypothesis that the fraction of ‘newly born’ cells would be highest in the LNs. Using a deuterium oxide (2H) in vivo labeling method in which patients consumed deuterated (heavy) water (2H2O), we determined CLL cell kinetics in concurrently obtained samples from LN, PB and BM. The LN was identified as the anatomical site harboring the largest fraction of newly born cells, compared to PB and BM. In fact, the calculated birth rate in the LN reached as high a 3.3% of the clone per day. Subdivision of the bulk CLL population by flow cytometry identified the subpopulation with the CXCR4dimCD5bright phenotype as containing the highest proportion of newly born cells within each compartment, including the LN, identifying this subclonal population as an important target for novel treatment approaches.





Similar content being viewed by others
References
Chiorazzi N, Rai KR, Ferrarini M . Chronic lymphocytic leukemia. N Engl J Med 2005; 352: 804–815.
Santos FP, O'Brien S . Small lymphocytic lymphoma and chronic lymphocytic leukemia: are they the same disease? Cancer J 2012; 18: 396–403.
Zenz T, Mertens D, Kuppers R, Dohner H, Stilgenbauer S . From pathogenesis to treatment of chronic lymphocytic leukaemia. Nat Rev Cancer 2010; 10: 37–50.
Sun C, Wiestner A . Prognosis and therapy of chronic lymphocytic leukemia and small lymphocytic lymphoma. Cancer Treat Res 2015; 165: 147–175.
Wierda WG, O'Brien S, Wang X, Faderl S, Ferrajoli A, Do KA et al. Multivariable model for time to first treatment in patients with chronic lymphocytic leukemia. J Clin Oncol 2011; 29: 4088–4095.
Chiorazzi N . Implications of new prognostic markers in chronic lymphocytic leukemia. Hematology Am Soc Hematol Educ Program 2012; 2012: 76–87.
Bulian P, Shanafelt TD, Fegan C, Zucchetto A, Cro L, Nuckel H et al. CD49d is the strongest flow cytometry-based predictor of overall survival in chronic lymphocytic leukemia. J Clin Oncol 2014; 32: 897–904.
Rossi D, Rasi S, Spina V, Bruscaggin A, Monti S, Ciardullo C et al. Integrated mutational and cytogenetic analysis identifies new prognostic subgroups in chronic lymphocytic leukemia. Blood 2013; 121: 1403–1412.
Stilgenbauer S, Schnaiter A, Paschka P, Zenz T, Rossi M, Dohner K et al. Gene mutations and treatment outcome in chronic lymphocytic leukemia: results from the CLL8 trial. Blood 2014; 123: 3247–3254.
Messmer BT, Messmer D, Allen SL, Kolitz JE, Kudalkar P, Cesar D et al. In vivo measurements document the dynamic cellular kinetics of chronic lymphocytic leukemia B cells. J Clin Invest 2005; 115: 755–764.
Defoiche J, Debacq C, Asquith B, Zhang Y, Burny A, Bron D et al. Reduction of B cell turnover in chronic lymphocytic leukaemia. Br J Haematol 2008; 143: 240–247.
Hayes GM, Busch R, Voogt J, Siah IM, Gee TA, Hellerstein MK et al. Isolation of malignant B cells from patients with chronic lymphocytic leukemia (CLL) for analysis of cell proliferation: validation of a simplified method suitable for multi-center clinical studies. Leuk Res 2009; 34: 809–815.
van Gent R, Kater AP, Otto SA, Jaspers A, Borghans JA, Vrisekoop N et al. In vivo dynamics of stable chronic lymphocytic leukemia inversely correlate with somatic hypermutation levels and suggest no major leukemic turnover in bone marrow. Cancer Res 2008; 68: 10137–10144.
Herishanu Y, Perez-Galan P, Liu D, Biancotto A, Pittaluga S, Vire B et al. The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood 2011; 117: 563–574.
Klein U, Tu Y, Stolovitzky GA, Mattioli M, Cattoretti G, Husson H et al. Gene expression profiling of B cell chronic lymphocytic leukemia reveals a homogeneous phenotype related to memory B cells. J Exp Med 2001; 194: 1625–1638.
Calissano C, Damle RN, Marsilio S, Yan XJ, Yancopoulos S, Hayes G et al. Intraclonal complexity in chronic lymphocytic leukemia: fractions enriched in recently born/divided and older/quiescent cells. Mol Med 2011; 17: 1374–1382.
Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Dohner H et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 2008; 111: 5446–5456.
Saba NS, Liu D, Herman SE, Underbayev C, Tian X, Behrend D et al. Pathogenic role of B-cell receptor signaling and canonical NF-kappaB activation in mantle cell lymphoma. Blood 2016; 128: 82–92.
Lis G, Wassenaar LI, Hendry MJ . High-precision laser spectroscopy D/H and 18O/16O measurements of microliter natural water samples. Anal Chem 2008; 80: 287–293.
Busch R, Neese RA, Awada M, Hayes GM, Hellerstein MK . Measurement of cell proliferation by heavy water labeling. Nat Protoc 2007; 2: 3045–3057.
Voogt JN, Awada M, Murphy EJ, Hayes GM, Busch R, Hellerstein MK . Measurement of very low rates of cell proliferation by heavy water labeling of DNA and gas chromatography/pyrolysis/isotope ratio-mass spectrometric analysis. Nat Protoc 2007; 2: 3058–3062.
Calissano C, Damle RN, Hayes G, Murphy EJ, Hellerstein MK, Moreno C et al. In vivo intraclonal and interclonal kinetic heterogeneity in B-cell chronic lymphocytic leukemia. Blood 2009; 114: 4832–4842.
Sivina M, Hartmann E, Kipps TJ, Rassenti L, Krupnik D, Lerner S et al. CCL3 (MIP-1alpha) plasma levels and the risk for disease progression in chronic lymphocytic leukemia. Blood 2011; 117: 1662–1669.
Dohner H, Stilgenbauer S, Benner A, Leupolt E, Krober A, Bullinger L et al. Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med 2000; 343: 1910–1916.
Burger JA, Burger M, Kipps TJ . Chronic lymphocytic leukemia B cells express functional CXCR4 chemokine receptors that mediate spontaneous migration beneath bone marrow stromal cells. Blood 1999; 94: 3658–3667.
Bennett F, Rawstron A, Plummer M, de Tute R, Moreton P, Jack A et al. B-cell chronic lymphocytic leukaemia cells show specific changes in membrane protein expression during different stages of cell cycle. Br J Haematol 2007; 139: 600–604.
Vlad A, Deglesne PA, Letestu R, Saint-Georges S, Chevallier N, Baran-Marszak F et al. Down-regulation of CXCR4 and CD62L in chronic lymphocytic leukemia cells is triggered by B-cell receptor ligation and associated with progressive disease. Cancer Res 2009; 69: 6387–6395.
Patten PE, Chu CC, Albesiano E, Damle RN, Yan XJ, Kim D et al. IGHV-unmutated and IGHV-mutated chronic lymphocytic leukemia cells produce activation-induced deaminase protein with a full range of biologic functions. Blood 2012; 120: 4802–4811.
Acknowledgements
We are grateful to all patients who participated in this study. We thank Ajunae Wells for assistance in the clinic, the NIH Surgery Branch, Judy Starling of the NIH Clinical Center Pharmacy and Theresa Davies-Hill for preparing LN single-cell suspensions. In addition, we thank Keyvan Keyvanfar for assistance with flow cytometry. This work was supported by the Intramural Research Program of the National, Heart, Lung and Blood Institute and the National Cancer Institute, the National Institutes of Health and an RO1 grant from the National Cancer Institute, NIH to NC. CUN was supported by the Danish Cancer Society. This research was supported by the Intramural Research Program of the National, Heart, Lung and Blood Institute and the National Cancer Institute. CUN received support from the Danish Cancer Society. Research support was also provided by an RO1 grant from the NIH National Cancer Institute (CA081554) to NC.
Author contributions
Designed the study: TMH, S-SC, NS, CE, XT, TEH, NC, AW; cared for patients: TMH, JV, TEH, GEM, GA, SS, MF, AW; collected data: TMH, S-SC, NS, JV, CE, SS, DCA, MS-S, CMY, CUN, SEMH, NC, AW; analyzed data: TMH, S-SC, NS, CE, MG, XT, DCA, MS-S, CMY, CUN, NC, AW; wrote manuscript: TMH, S-SC, CE, NC, AW.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
CE and MG are employees of KineMed Inc. CUN reports consultancy for Roche, Janssen, Gilead, and receipt of travel grants from Roche, Gilead and Novartis. The remaining authors declare no competing financial interests.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Herndon, T., Chen, SS., Saba, N. et al. Direct in vivo evidence for increased proliferation of CLL cells in lymph nodes compared to bone marrow and peripheral blood. Leukemia 31, 1340–1347 (2017). https://doi.org/10.1038/leu.2017.11
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.11
- Springer Nature Limited