Abstract
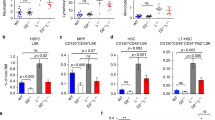
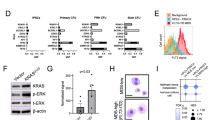
Fanconi anemia (FA) is an inherited bone marrow failure syndrome with extremely high risk of leukemic transformation. Here we investigate the relationship between DNA damage response (DDR) and leukemogenesis using the Fanca knockout mouse model. We found that chronic exposure of the Fanca−/− hematopoietic stem cells to DNA crosslinking agent mitomycin C in vivo leads to diminished DDR, and the emergence/expansion of pre-leukemia stem cells (pre-LSCs). Surprisingly, although genetic correction of Fanca deficiency in the pre-LSCs restores DDR and reduces genomic instability, but fails to prevent pre-LSC expansion or delay leukemia development in irradiated recipients. Furthermore, we identified transcription program underlying dysregulated DDR and cell migration, myeloid proliferation, and immune response in the Fanca−/− pre-LSCs. Forced expression of the downregulated DNA repair genes, Rad51c or Trp53i13, in the Fanca−/− pre-LSCs partially rescues DDR but has no effect on leukemia, whereas shRNA knockdown of the upregulated immune receptor genes Trem1 or Pilrb improves leukemia-related survival, but not DDR or genomic instability. Furthermore, Trem1 cooperates with diminished DDR in vivo to promote Fanca−/− pre-LSC expansion and leukemia development. Our study implicates diminishing DDR as a root cause of FA leukemogenesis, which subsequently collaborates with other signaling pathways for leukemogenic transformation.






Similar content being viewed by others
References
Bagby GC . Genetic basis of Fanconi anemia. Curr Opin Hematol 2003; 10: 68–76.
Kennedy RD, D'Andrea AD . The Fanconi anemia/BRCA pathway: new faces in the crowd. Genes Dev 2000; 19: 2925–2940.
Bogliolo M, Schuster B, Stoepker C, Derkunt B, Su Y, Raams A et al. Mutations in ERCC4, encoding the DNA-repair endonuclease XPF, cause Fanconi anemia. Am J Hum Genet 2013; 92: 800–806.
Garaycoechea JI, Patel KJ . Why does the bone marrow fail in Fanconi anemia? Blood 2014; 123: 26–34.
Hodskinson MR, Silhan J, Crossan GP, Garaycoechea JI, Mukherjee S, Johnson CM et al. Mouse SLX4 is a tumor suppressor that stimulates the activity of the nuclease XPF-ERCC1 in DNA crosslink repair. Mol Cell 2014; 54: 472–484.
Sawyer SL, Tian L, Kähkönen M, Schwartzentruber J, Kircher M et alUniversity of Washington Centre for Mendelian Genomics. Biallelic mutations in BRCA1 cause a new Fanconi anemia subtype. Cancer Discov 2015; 5: 135–142.
Kottemann MC, Smogorzewska A . Fanconi anaemia and the repair of Watson and Crick DNA crosslinks. Nature 2013; 493: 356–363.
Deans AJ, West SC . DNA interstrand crosslink repair and cancer. Nat Rev Cancer 2011; 11: 467–480.
Auerbach AD, Allen RG . Leukemia and preleukemia in Fanconi anemia patients. A review of the literature and report of the International Fanconi Anemia Registry. Cancer Genet Cytogenet 1991; 51: 1–12.
Mehta P, Locatelli F, Stary J, Smith FO . Bone marrow transplantation for inherited bone marrow failure syndromes. Pediatr Clin North Am 2010; 57: 147–170.
Smith AR, Wagner JE . Current clinical management of Fanconi anemia. Expert Rev Hematol 2012; 5: 513–522.
Liu J Fanconi’s anemia. In: Young NS, ed. Bone Marrow Failure Syndromes. WB Saunders: Philadelphia, PA, 2000.
Du W, Adam Z, Rani R, Zhang X, Pang Q . Oxidative stress in Fanconi anemia hematopoiesis and disease progression. Antioxid Redox Signal 2008; 10: 1909–1921.
Kutler DI, Singh B, Satagopan J, Batish SD, Berwick M, Giampietro PF et al. A 20-year perspective on the International Fanconi Anemia Registry (IFAR). Blood 2003; 101: 1249–1256.
Lensch MW, Rathbun RK, Olson SB, Jones GR, Bagby GC . Selective pressure as an essential force in molecular evolution of myeloid leukemic clones: a view from the window of Fanconi anemia. Leukemia 1999; 13: 1784–1789.
Liu JM . Gene transfer for the eventual treatment of Fanconi’s anemia. Semin Hematol 1998; 35: 168–179.
Bartkova J, Horejsí Z, Koed K, Krämer A, Tort F, Zieger K et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 2005; 434: 864–870.
Di Micco R, Fumagalli M, Cicalese A, Piccinin S, Gasparini P, Luise C et al. Oncogene induced senescence is a DNA damage response triggered by DNA hyperreplication. Nature 2006; 444: 638–642.
Gorgoulis VG, Vassiliou LV, Karakaidos P, Zacharatos P, Kotsinas A, Liloglou T et al. Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 2005; 434: 907–913.
Halazonetis TD, Gorgoulis VG, Bartek J . An oncogene induced DNA damage model for cancer development. Science 2008; 319: 1352–1355.
Boehrer S, Adès L, Tajeddine N, Hofmann WK, Kriener S, Bug G et al. Suppression of the DNA damage response in acute myeloid leukemia versus myelodysplastic syndrome. Oncogene 2009; 28: 2205–2218.
Wajapeyee N, Wang SZ, Serra RW, Solomon PD, Nagarajan A, Zhu X et al. Senescence induction in human fibroblasts and hematopoietic progenitors by leukemogenic fusion proteins. Blood 2010; 115: 5057–5060.
Takacova S, Slany R, Bartkova J, Stranecky V, Dolezel P, Luzna P et al. DNA damage response and inflammatory signaling limit the MLL-ENL-induced leukemogenesis in vivo. Cancer Cell 2012; 21: 517–531.
Cozzio A, Passegué E, Ayton PM, Karsunky H, Cleary ML, Weissman I . Similar MLL-associated leukemias arising from self-renewing stem cells and short-lived myeloid progenitors. Genes Dev 2003; 17: 3029–3035.
Krivtsov AV, Twomey D, Feng Z, Stubbs MC, Wang Y, Faber J et al. Transformation from committed progenitor to leukaemia stem cell initiated by MLL-AF9. Nature 2006; 442: 818–822.
Wong JC, Alon N, Mckerlie C, Huang JR, Meyn MS, Buchwald M . Targeted disruption of exons 1 to 6 of the Fanconi Anemia group A gene leads to growth retardation, strain-specific microphthalmia, meiotic defects and primordial germ cell hypoplasia. Hum Mol Genet 2003; 12: 2063–2076.
Chen M, Tomkins DJ, Auerbach W, McKerlie C, Youssoufian H, Liu L et al. Inactivation of Fac in mice produces inducible chromosomal instability and reduced fertility reminiscent of Fanconi anaemia. Nat Genet 1996; 12: 448–451.
Zhang N, Ahsan MH, Zhu L, Sambucetti LC, Purchio AF, West DB . NF-kappaB and not the MAPK signaling pathway regulates GADD45beta expression during acute inflammation. J Biol Chem 2005; 280: 21400–21408.
Carreau M, Gan OI, Liu L, Doedens M, McKerlie C, Dick JE et al. Bone marrow failure in the Fanconi anemia group C mouse model after DNA damage. Blood 1998; 91: 2737–2744.
Oostra AB, Nieuwint AW, Joenje H, de Winter JP . Diagnosis of Fanconi anemia: chromosomal breakage analysis. Anemia 2012; 2012: 238731.
Salvador JM, Brown-Clay JD, Fornace AJ Jr . Gadd45 in stress signaling, cell cycle control, and apoptosis. Adv Exp Med Biol 2013; 793: 1–19.
Liebermann DA, Tront JS, Sha X, Mukherjee K, Mohamed-Hadley A, Hoffman B . Gadd45 stress sensors in malignancy and leukemia. Crit Rev Oncog 2011; 16: 129–140.
Lu Q, Lu G, Qi J, Wang H, Xuan Y, Wang Q et al. PILRα and PILRβ have a siglec fold and provide the basis of binding to sialic acid. Proc Natl Acad Sci USA 2014; 111: 8221–8226.
Wang J, Sun Q, Morita Y, Jiang H, Gross A, Lechel A et al. A differentiation checkpoint limits hematopoietic stem cell self-renewal in response to DNA damage. Cell 2012; 148: 1001–1014.
Masson JY, Tarsounas MC, Stasiak AZ, Stasiak A, Shah R, McIlwraith MJ et al. Identification and purification of two distinct complexes containing the five RAD51 paralogs. Genes Dev 2001; 15: 3296–3307.
Reinhardt HC, Schumacher B . The p53 network: cellular and systemic DNA damage responses in aging and cancer. Trends Genet 2012; 28: 128–136.
Wu J, Li J, Salcedo R, Mivechi NF, Trinchieri G, Horuzsko A . The proinflammatory myeloid cell receptor TREM-1 controls Kupffer cell activation and development of hepatocellular carcinoma. Cancer Res 2012; 72: 3977–3986.
Lukens JR, Gurung P, Shaw PJ, Barr MJ, Zaki MH, Brown SA et al. The NLRP12 sensor negatively regulates autoinflammatory disease by modulating interleukin-4 production in T cells. Immunity 2015; 42: 654–664.
Klesney-Tait J, Keck K, Li X, Gilfillan S, Otero K, Baruah S et al. Transepithelial migration of neutrophils into the lung requires TREM-1. J Clin Invest 2013; 123: 138–149.
Arthur JC, Lich JD, Ye Z, Allen IC, Gris D, Wilson JE et al. Cutting edge: NLRP12 controls dendritic and myeloid cell migration to affect contact hypersensitivity. J Immunol 2010; 185: 4515–4519.
Kuroki K, Wang J, Ose T, Yamaguchi M, Tabata S, Maita N et al. Structural basis for simultaneous recognition of an O-glycan and its attached peptide of mucin family by immune receptor PILRα. Proc Natl Acad Sci USA 2014; 111: 8877–8882.
Weiss CN, Ito K . DNA damage: a sensible mediator of the differentiation decision in hematopoietic stem cells and in leukemia. Int J Mol Sci 2015; 16: 6183–6201.
Naka K, Hirao A . Maintenance of genomic integrity in hematopoietic stem cells. Int J Hematol 2011; 93: 434–439.
Clarke MF, Fuller M . Stem cells and cancer: two faces of eve. Cell 2006; 124: 1111–1115.
Jamieson CH, Weissman IL, Passegué E . Chronic versus acute myelogenous leukemia: a question of self-renewal. Cancer Cell 2004; 6: 531–533.
Du W, Erden O, Pang Q . TNF-α signaling in Fanconi anemia. Blood Cells Mol Dis 2014; 52: 2–11.
Mandal PK, Blanpain C, Rossi DJ . DNA damage response in adult stem cells: pathways and consequences. Nat Rev Mol Cell Biol 2011; 12: 198–202.
Niedernhofer LJ . DNA repair is crucial for maintaining hematopoietic stem cell function. DNA Repair 2008; 7: 523–529.
Jan M, Snyder TM, Corces-Zimmerman MR, Vyas P, Weissman IL, Quake SR et al. Clonal evolution of preleukemic hematopoietic stem cells precedes human acute myeloid leukemia. Sci Transl Med 2012; 4: 149ra118.
Welch JS, Ley TJ, Link DC, Miller CA, Larson DE, Koboldt DC et al. The origin and evolution of mutations in acute myeloid leukemia. Cell 2012; 150: 264–278.
Santos MA, John S, Nussenzweig A . Tumor promoting role of the DNA damage response. Cell Cycle 2014; 13: 2807–2808.
Rossi DJ, Bryder D, Seita J, Nussenzweig A, Hoeijmakers J, Weissman IL . Deficiencies in DNA damage repair limit the function of haematopoietic stem cells with age. Nature 2007; 447: 725–729.
Zhang S, Yajima H, Huynh H, Zheng J, Callen E, Chen HT et al. Congenital bone marrow failure in DNA-PKcs mutant mice associated with deficiencies in DNA repair. J Cell Biol 2011; 193: 295–305.
Milyavsky M, Gan OI, Trottier M, Komosa M, Tabach O, Notta F et al. A distinctive DNA damage response in human hematopoietic stem cells reveals an apoptosis-independent role for p53 in self-renewal. Cell Stem Cell 2010; 7: 186–197.
Gilliland DG, Tallman MS . Focus on acute leukemias. Cancer Cell 2002; 1: 417–420.
Pateras IS, Havaki S, Nikitopoulou X, Vougas K, Townsend PA, Panayiotidis MI et al. The DNA damage response and immune signaling alliance: is it good or bad? Nature decides when and where. Pharmacol Ther 2015; 154: 36–56.
Fontes FL, Pinheiro DM, Oliveira AH, Oliveira RK, Lajus TB, Agnez-Lima LF . Role of DNA repair in host immune response and inflammation. Mutat Res Rev Mutat Res 2015; 763: 246–257.
Günther C, Kind B, Reijns MA, Berndt N, Martinez-Bueno M, Wolf C et al. Defective removal of ribonucleotides from DNA promotes systemic autoimmunity. J Clin Invest 2015; 125: 413–424.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Tang ML, Gasser S . ATM activation mediates anticancer immunosurveillance by natural killer and T cells. Oncoimmunology 2013; 2: e24438.
Motz GT, Santoro SP, Wang LP, Garrabrant T, Lastra RR, Hagemann IS et al. Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat Med 2014; 20: 607–615.
Breccia M, Molica M, Alimena G . How tyrosine kinase inhibitors impair metabolism and endocrine system function: A systematic updated review. Leuk Res 2014; 38: 1392–1398.
Acknowledgements
We thank Dr Madeleine Carreau (Laval University) for Fanca+/− mice, Dr Manuel Buchwald (University of Toronto) for Fancc+/− mice, Dr Lenhand Rudolph (Institute of Molecular Medicine and Max-Planck-Research, Germany) for SF-LV-shRNA-EGFP vector, Dr Punam Malik (Cincinnati Children’s Hospital Medical Center) for the pRRL-SIN-cPPTMNDU3-MCS-IVW (TMND-IRES-Venus) vector, the Viral Vector Core of Cincinnati Children’s Research Foundation (Cincinnati Children’s Hospital Medical Center) for the preparation of viruses, and the Comprehensive Mouse and Cancer Core of the Cincinnati Children’s Research Foundation (Cincinnati Children’s Hospital Medical Center) for BM transplantation service. This investigation was supported by National Institutes of Health grants R01 HL076712, R01 CA157537 and T32 HL091805. QP is supported by a Leukemia and Lymphoma Scholar award. WD is supported by National Natural Science Fundation of China 81470288.
Author contributions
WD designed the research, performed the research, analyzed the data and wrote the paper; SA performed the research and analyzed the data; AFW performed the research; QP designed the research and wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Du, W., Amarachintha, S., Wilson, A. et al. The immune receptor Trem1 cooperates with diminished DNA damage response to induce preleukemic stem cell expansion. Leukemia 31, 423–433 (2017). https://doi.org/10.1038/leu.2016.242
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.242
- Springer Nature Limited
This article is cited by
-
Quinacrine-CASIN combination overcomes chemoresistance in human acute lymphoid leukemia
Nature Communications (2021)