Abstract
Objective:
To investigate whether early treatment with inhaled nitric oxide (iNO) could prevent bronchopulmonary dysplasia (BPD) in very preterm infants.
Study Design:
A non-randomized, controlled trial was conducted prospectively in 27 neonatal intensive care units over 12 months. Preterm infants with gestational age <34 weeks and after 7 days of life, who received invasive mechanical ventilation (MV) or nasal continuous positive airway pressure for >2 days, were treated either with low-dose iNO (from 5 as initial dose to 2 parts per million as maintenance dose for ⩾7 days, n=162) or as non-placebo control (n=240). Primary outcome was the incidence of moderate-to-severe BPD at 36 weeks postmenstrual age and/or death before discharge. Secondary outcomes were major complications.
Results:
iNO was started on average on day 19 of life (median duration 18 days, range 7 to 55 days). Rate of survival without BPD was significantly lower in the iNO than in the control group, whereas overall rates of BPD, death and major complications were similar between the two groups. Infants who started MV and iNO on postnatal days 15 to 21 had significantly increased survival without BPD (47.6% vs 17.1%, P=0.03, relative risk 2.7, 95% confidence interval 1.1 to 6.5). Additionally, pooled data from both groups showed that rates of perinatal co-morbidities and postnatal complications were higher in BPD infants than in non-BPD infants. The overall incidence of BPD was 55.6% and 75.9% for birth weight <1500 and <1000 g, respectively, or 1.6% for the total population <34 weeks of gestation admitted through the network.
Conclusion:
Treatment with low-dose iNO did not decrease the overall risk of BPD and death nor showed adverse effects in short-term morbidities among very preterm infants. The benefit of delayed iNO treatment on BPD warrants further studies.
Similar content being viewed by others
Introduction
With the establishment and dramatic expansion of neonatal special care and a universal health insurance policy for rural families, there have been advances in perinatal–neonatal care across China. These advances have resulted in significant reductions in maternal and neonatal mortality and improved survival of very preterm infants.1, 2, 3 Bronchopulmonary dysplasia (BPD), or chronic lung disease, and other chronic conditions related to prematurity are more frequently encountered in neonatal intensive care units (NICUs) in the postsurfactant era as a result of improved survival of very preterm infants.4 In the developed countries, BPD occurs in 20 to 30% of newborns with birth weight (BW) <1500 g and gestational age (GA) <30 weeks.5, 6, 7 So far, there is no report of the incidence of BPD based on either NICU admissions or the total birth population in China. Prenatal risks, lung immaturity, invasive mechanical ventilation (MV), oxygen toxicity, persistent infection and inflammation have been reported to be involved in the etiology of BPD.8, 9, 10 Existing interventions such as non-invasive ventilation, surfactant, postnatal corticosteroids, fluid restriction, caffeine and nutrition have shown inconsistent or limited effects, often with controversies over their values.8, 9, 10, 11, 12, 13 It remains unknown whether the main etiological factors of BPD and the responses of BPD patients to various interventions are the same across different regions of the world.
Inhaled nitric oxide (iNO), a selective pulmonary vasodilator, is routinely used for the treatment of term and late preterm newborns with hypoxemic respiratory failure and persistent pulmonary hypertension of the newborn.14 The use of iNO has been extended to very preterm neonates, albeit off-label, for both hypoxemic respiratory failure/persistent pulmonary hypertension of the newborn and those at risk of BPD.15, 16, 17, 18, 19, 20, 21, 22, 23 The prevalence of BPD and the efficacy of iNO to prevent BPD may be associated with ethnicity and timing of the intervention.15, 16, 17, 18, 19, 20 Some of the benefits of iNO in preterm neonates may be associated with alveolarization24, 25 and suppression of inflammation.26 However, randomized controlled trials investigating the efficacy of iNO to reduce the risk of BPD and death rate showed no significant reduction in BPD and/or death.15, 16, 17, 18, 19, 20, 21, 22, 23
The purpose of this non-randomized, controlled study was to evaluate the efficacy and safety of iNO among very preterm infants with evolving BPD in the background of a growing network of neonatal units in China. Thereby we also sought to assess the quality of current respiratory care, with primary outcomes being the survival in the absence of BPD as well as overall incidence and outcome of BPD in very preterm infants. We hypothesized that iNO given in a low-dose regimen after 7 days of postnatal life may reduce the overall incidence and severity of BPD without causing major complications.
Materials and Methods
Characteristics of participating NICUs
Twenty-seven NICUs across the country participated in this study, including 13 maternity hospitals, 8 children’s hospitals and 6 general hospitals, all of which were tertiary referral centers. Coordination was carried out by Children’s Hospital of Fudan University, the ethics committee of which approved the study protocols and ensured that the protocol was adopted by all participating centers according to Chinese regulations for clinical investigation.
Study design and protocols
Based on our previous experience in routine treatment of high-risk premature patients,4 a randomized trial would be very difficult, if not impossible, to be carried out in the current neonatal network.27 The actual incidence of BPD in hospitalized very preterm infants in China is not known yet, we therefore conducted a prospective, non-randomized and controlled trial (Clinical trial registry number: NCT01503801 at ClinicalTrials.gov).
Definition of preterm birth with GA was based on best obstetrical estimate, prenatal ultrasound, last menstrual period and neonatal examination using modified Ballard/Dubowitz score and expressed according to the Tenth revision of International Statistical Classification of Disease and Related Health Problems (ICD-10, WHO, 1993). Based on an estimated BPD prevalence of 50% in neonates with GA 25 to 33 weeks,4, 5 combined with an intervention related BPD reduction of 10% (that is, to 40%), a type I (α) error of 0.05 and type II (β) 0.2 (power 0.8), our power calculations yielded a sample size of 382 cases for each group.28 The enrollment was continued for 12 consecutive months, followed by an interim assessment for safety and efficacy. If the interim results showed an increased trend for adverse events in the absence of potential benefits, the study would be terminated.
All maternal pathologies and pregnancy-associated complications such as prenatal medical conditions, intrapartum and early postnatal complications related to intrauterine stress, delivery mode, resuscitation and stabilization and neonatal illnesses present before enrollment were recorded according to the definitions from previous randomized trials, 15, 16, 17, 18, 19, 20 domestically enforced obstetric routines and internationally recognized definitions and recommendations.29
Patient population
From 1 July 2011 to 30 June 2012, all neonates admitted to the 27 NICUs with GA <34 weeks were enrolled if they required MV via an endotracheal tube or nasal continuous positive airway pressure (nCPAP; with supplemental oxygen at FiO2>30%) to treat hypoxemic respiratory failure for at least 48 h after 7 postnatal days. Enrollment was not dependent on respiratory support before 7 days of age. Exclusion criteria included life-threatening conditions such as complex congenital anomalies, active pulmonary hemorrhage, tension pneumothorax, preexisting bilateral grade III/IV intraventricular hemorrhage30 and thrombocytopenia <50 000 mm−3,31 as well as duration of MV or CPAP <48 h. Informed consent was obtained from parents or guardians prior to the enrollment. Patients were either enrolled into a group with inhaled NO (iNO) or a control group, based on the availability of iNO at the individual facility. The enrollment was also influenced by the attending physician’s discretion and parents’ decision.
Study monitoring and data collection
Relevant personnel of all participating NICUs were trained in order to optimize their compliance with the iNO treatment protocol, which was deemed to be the best available practice, and to enhance the quality of data collection. For all infants who met the entry criteria, on-site investigators collected information on case record forms in both groups. Management of all other conditions and complications was left to the discretion of attending physicians.
Diagnosis of BPD
BPD was diagnosed according to the previous definitions and expert recommendations.6, 15, 16, 17, 18, 19, 20, 32, 33 Mild BPD was diagnosed if a patient had oxygen therapy for >28 days but not at 36 weeks of postmenstrual age (PMA) or at discharge. On the other hand, moderate BPD was defined for those on O2 >28 days and at 36 weeks PMA or at discharge and severe BPD for those requiring >30% oxygen, CPAP or MV at 36 weeks of PMA. Those who were receiving CPAP or MV on day 28 of postnatal life but died before 36 weeks of PMA, with clinical manifestation resembling moderate-to-severe BPD, were categorized as death related to BPD.
End point and primary and secondary outcomes
The end point was death or survival at discharge from the hospital, and the primary outcome was survival without moderate-to-severe BPD. Secondary outcomes included major clinical complications such as patent ductus arteriosus, necrotizing enterocolitis, sepsis or retinopathy of prematurity. The co-morbidities identified at birth and/or at NICU admission were also followed up to the completion of the study.
iNO treatment protocol
iNO was delivered into the inspiratory arm of the ventilator circuit using an iNO delivery system in a range of 0 to 200 ml min−1.34 The NO and nitrogen dioxide (NO2) levels were continuously monitored at the end of the inspiratory limb using electrochemical techniques.34 Initial iNO dose was 5 p.p.m. for the first 24 h, then decreased to 2 p.p.m. until weaning, with a minimum duration of 7 days.15, 16, 17, 18, 19, 20 We measured blood or transcutaneous methemoglobin concentrations (Masimo Rainbow SET(R) Radical-7 Pulse CO-Oximeter, Masimo, Irvine, CA, USA) at baseline, within 24 h of the initiation of iNO and once daily thereafter.35, 36 If the concentration of NO2 exceeded 3 p.p.m. or methemoglobin exceeded 5%, the iNO concentration would be decreased or the intervention ceased to reduce the risk of severe adverse events. MV and iNO therapies were supervised by the attending neonatologists who were aware of the treatment assignment.
Estimation of incidence of BPD and associated risk factors
In order to calculate the incidence of BPD for premature infants admitted to the NICUs in our network, we divided the number of cases by the total number of admissions under 34 weeks’ gestation. To identify the risk factors for BPD, we merged the iNO and control groups and calculated the relative risks or odds ratios of BPD for infants with and without certain risk factors. We performed logistic regression of selected risk factors for BPD and death, accounting for GA, BW, SNAPPE-II (Score for Neonatal Acute Physiology with Perinatal Extension-II) and major complications to determine the independent predictive power of each factor.
Statistical analysis
Statistical analysis was performed using the SPSS software (version. 21.0; IBM, New York, NY, USA). Continuous variables were summarized as median with interquartile range or range or as means and s.d., and differences were compared using Mann–Whitney test. Categorical variables were presented as numbers and proportions in percentage, and differences were compared using Chi square test or Fisher’s exact test. A P-value <0.05 was considered statistically significant. Assessment of individual clinical risk factors associated with mortality and BPD was performed by univariate or bivariate logistic regression, and variables detected with P<0.1 in univariate logistic regression and closely related to mortality and BPD were further tested in binary multivariate logistic regression (forward). The goodness of fit of the model was examined by Hosmer–Lemeshow test. Results were reported as odds ratio or relative risk and 95% confidence interval (CI).
Results
As preliminary statistic analysis failed to show clear benefits of iNO with regard to major outcomes, the study was terminated prematurely (see below for details in Tables 1 and 2).
General characteristics of the two groups at study entry
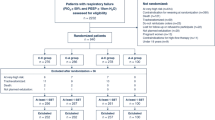
This study was carried out in strict compliance with the protocol from the start but terminated at interim assessment with 433 cases enrolled, of which 402 fulfilled the criteria for statistical analysis (Figure 1). Supplementary Table S1 shows the patients’ perinatal and baseline demographic characteristics, with no significant between-group differences in gender, GA and BW (and strata) and maternal complications except hypertension. Baseline co-morbidities were similar between the two groups, and 75% of the total population received surfactant. Postnatal steroids were more frequently used in the iNO than in the control group. More infants in the iNO group had a history of previous pulmonary hemorrhage, which was inactive at entry. There were no significant differences between the two groups in terms of other preexisting co-morbidities and conditions.
The median (interquartile range) age at entry was 19 (13, 28) days in the iNO group, 4 days later than the control group (15 (12, 19)), with an average duration of 18 (range 7, 55) days of iNO. More patients in the iNO group were born in non-tertiary hospitals, but the median oxygenation index for the two groups was similar at study entry. More infants in the control group received nCPAP at study entry.
Major outcome and risks measurement
Fewer patients in the iNO group survived without BPD to discharge as compared with the control group (Table 1). Although the individual risk of BPD or death was similar in both groups, the combined rate of BPD and death before discharge was significantly higher in the iNO than in the control group. Overall rates of BPD and death were similar between the two groups. However, when cases with pulmonary hemorrhage prior to the entry were excluded, the differences of survival without BPD between the two groups (n=137 vs n=227) were not statistically significant (48 (35.0%) vs 101 (44.5%), relative risk 0.8 (95% CI 0.6 to 1.0), P=0.075). The same was also observed for the outcome of death or survival with BPD (89 (65.0%) vs 126 (55.5%), relative risk 1.2 (1.1 to 1.4), P=0.075, respectively).
Ventilation mode at study entry categorized under BW strata and the disease severity (Table 1) showed no significant differences between the two groups. The disease severity as well as the combined risk of death and BPD were less in those with nCPAP than in those with MV (conventional MV (CMV), high-frequency oscillation ventilation (HFOV)). The median (and range) duration of iNO in those with nCPAP, CMV and HFOV was 13 (7 to 35), 16 (7 to 55) and 20 (7 to 54) days, respectively. There were no significant differences in the incidence of BPD between the two groups by different ventilation modes.
Infants enrolled at 15 to 21 days of age receiving MV (CMV and/or HFOV) showed improved outcome after iNO treatment, indicating that the onset of intervention may be related with the outcome. At entry, infants on iNO were less likely to have birth asphyxia but more likely to have pulmonary hemorrhage than the control group. Moreover, no significant differences were found between both subsets in gender, GA, BW, antenatal steroids or previous surfactant, whereas the incidence of BPD and the combined risk of BPD and death before discharge were significantly lower in the iNO than in the control group. However, the rate of death was similar in both groups (Table 1). Regarding the initial ventilation mode, fewer patients in the iNO group were on nCPAP and HFOV than in the control group. There were no significant differences in each ventilation mode by BW strata in terms of incidence of death, BPD, death or survival with BPD. When cases with pulmonary hemorrhage prior to the entry were excluded (n=15 (10.9%) in iNO vs n=34 (14.9%) in control), the between-group statistical difference in survival without BPD remained (8 (53.3%) vs 6 (17.6), relative risk 0.6 (0.3 to 0.9), P=0.011). The same was also observed for the outcome of death or survival with BPD (7 (46.7%) vs 28 (82.4%), relative risk 3.0 (95% CI 1.3 to 7.2), P=0.011). It is notable that this postadjustment between-group statistical difference only persisted in the subgroup with delayed iNO treatment. These differences were found only in these time window subsets.
In the iNO group, none had significantly elevated methemoglobin level. The methemoglobin concentration was 0.3 to 0.4% at baseline, 0.5 to 0.7% in 1 to 24 h, and 0.4 to 0.6% on day 2 and ensuing days. There were no significant differences between the two groups in the incidence and severity of complications or co-morbidities of prematurity resulting from intrapartum events (Table 2). The incidence of pulmonary hemorrhage was low in both groups.
Incidence of BPD from the merged data
As there were no significant differences between the two groups in the major outcome measures, data were merged for bivariate analysis of overall risks of major outcome including co-morbidities and complications (Supplementary Table S2). Results of binary logistic regression analysis of variables associated with the mortality and incidence of moderate-to-severe BPD are shown in Table 3. As compared with infants who did not develop BPD, those who developed BPD had higher scores of SNAPPE-II and were more likely to have delivery room resuscitation, complications requiring intervention, respiratory distress syndrome, MV, surfactant and postnatal corticosteroids.
The overall incidence of moderate-to-severe BPD was 1.6% (2.5% if mild BPD were included) among the total NICU admissions of those <34 weeks of gestation (10 972), with a case-specific death rate of 13.4% (54/402) (Supplementary Table S2). The incidence (95% CI) of BPD (including the deaths from BPD) at 36 weeks of PMA in those with GA <28, <32 and <34 weeks was 64.5% (54.4 to 73.6%), 55.1% (49.8 to 60.4%) and 53.0% (48.1 to 57.9%), respectively, and in those with BW <1000, <1250 and <1500 g, 75.9% (66.5 to 85.3%), 57.1% (50.7 to 63.5%) and 55.6% (50.2 to 61.0%), respectively.
Discussion
This study was our first observation in a rapidly developing NICU network through a prospective, non-randomized iNO trial for very preterm infants with evolving BPD. As compared with the control group, the combined rate of death or survival with BPD was significantly higher, and survival without BPD lower, in the iNO group. These findings were different from previous randomized trials17, 18, 19, 20 but similar to that of a meta-analysis.37 The dose and timing of iNO in the current study were close to, and the enrolled subjects relatively more mature and the main outcomes worse than, those of the previous trials or survey.18, 19, 20, 23 A subset of neonates treated with iNO at 15 to 21 days of life with MV showed better outcome despite a small sample size. No severe adverse events were reported, and some of the perinatally acquired co-morbidities of prematurity, such as severe IVH and pulmonary hemorrhage, did not aggravate during iNO treatment. Furthermore, stratified data analysis showed no significant difference in death and/or moderate-to-severe BPD when patients with prior pulmonary hemorrhage were excluded in the final outcome analysis.
The study protocol had both advantages and disadvantages. First, despite a non-randomized design, this study ensured the enrollment of a large sample of eligible cases. The vast majority of the baseline variables except gestational hypertension and entry time were similarly distributed between the two groups (Supplementary Table S1), indicating a low risk of heterogeneity. Discrepancy was found in the initial ventilation mode as neonates in the iNO group were more frequently exposed to MV, had a longer MV duration of >7 days and higher proportion of postnatal corticosteroid use than the control group (Supplementary Table S1). This is probably due to more pulmonary hemorrhage and sepsis in the iNO group at entry, suggesting an early and persistent pulmonary dysfunction23, 38 prior to and during the enrollment for iNO treatment. Nevertheless, SNAPPE-II, oxygenation index levels and prior surfactant use, reflecting the initial baseline perinatal status and underlying diseases of those enrolled, were similar between the two groups. Second, all the variables for assessment of the primary and secondary end points, as well as perinatal co-morbidities and postnatal complications/severe adverse events, were obtained, reflecting the comprehensiveness and validity of the study. Given the fact that the overall incidence of BPD and its related outcomes were evenly distributed between both groups, patients of the two groups at entry should have been derived from the population with very similar risks of developing BPD. This, in combination with an identical procedure carried out throughout the trial, may have counterbalanced, at least in part, the above-mentioned discrepancies. As observational or controlled trials of a larger sample size as compared with ours are likely to be achieved in regions where the perinatal and neonatal care is emerging or well advanced,21, 39, 40, 41, 42, 43 the non-randomized design may be an approach of more value to reflect the actual NICU care standard than randomization-based procedures, given the limitation of our current health-care system and validity of results demonstrated by our study as compared with similar randomized, controlled trials. Other alternative approaches, such as specific case registries, were used to generally assess the efficacy of iNO therapy and BPD outcome in the total NICU admissions or birth population profiles by data integration and sample adjustment.21, 22, 23, 40
Our iNO treatment protocol concur with the clinical experience and trial by Ballard et al.19 in that the improvement of survival without BPD by iNO was associated with specified timing and dosing, with the risk of bleeding and methemoglobinemia accounted for.31, 35, 36 Similar as the EUNO trial, we enrolled those with CPAP as initial ventilation mode.20 Our results did not support the hypothesis that iNO could prevent BPD; however, the outcome analysis did help in understanding the efficacy of early therapeutic iNO and the risks of developing BPD and respiratory failure in very preterm infants. Notably, this iNO regimen did show benefit for a subgroup with delayed iNO and MV at 15 to 21 days, suggesting a potential effect of iNO on the lungs that were more maturely developed. This contrasts the overall efficacy with regard to MV strength and MV >7 days at study entry and requires further large-sample study to verify. The CPAP subgroups did not show any benefit from iNO, which was consistent with the results from EUNO trial20 and a recent trial by Kinsella et al.44
Our study showed that BPD occurred in 55.6% of very low BW and 75.9% of extremely low BW based on the merged data, though the overall incidence of BPD in very preterm infants was low. By comparing with similar iNO trials15, 16, 17, 18, 19, 20 based on population or NICU admission data,43 our BPD rates were much higher, indicating that our standard of care may have lagged behind North America and Nordic countries by nearly two decades.5, 7 From our previous complete birth-population-based regional survey, those with GA<34 weeks and BW<2000 g accounted for 0.8 to 1% of all births and had a survival rate of 80%. Among all births, 1 to 1.5% required NICU admission after birth.45 Current data show that the national annual births are 16.5 million, of which 5% are preterm and 1% are with a GA<34 weeks.2, 45, 46 In view of this, and with reference to the current BPD incidence and outcome data, there should be 2 00 000 infants requiring NICU admission after birth. No <2%, or rather 4000 cases annually, may develop moderate-to-severe BPD or BPD-related death. In fact, no >50% of the total birth population are accessible to medical care of a standard equivalent to the current NICU network standard, and the proportion would be up to 60 to 70% in the next decade to come. This will be a definitive challenge and burden to the entire emerging perinatal–neonatal care system. However, this speculation awaits further large-scaled study to confirm.5, 7, 21, 39, 40, 41, 42, 43
In conclusion, the prolonged, low-dose iNO treatment conferred no benefits nor inflicted any adverse effects on preterm infants <34 weeks with evolving BPD. Notably, a lower rate of survival without BPD was found in the iNO group. To accurately assess the risk and incidence of BPD, further studies of iNO in this population should be more specified on time window and ventilation mode. Complete GA and BW stratified outcome measurement as well as long-term follow-up are also needed. The advantage and limitation of the trial methodology warrant further investigation.
References
Rajaratnam JK, Marcus JR, Flaxman AD, Wang H, Levin-Rector A, Dwyer L et al. Neonatal, post neonatal, childhood, and under-5 mortality for 187 countries, 1970-2010: a systematic analysis of progress towards Millennium Development Goal 4. Lancet 2010; 375 (9730): 1988–2008.
Sun B, Shao X, Cao Y, Xia S, Yue H . Neonatal-perinatal medicine in a transitional period in China. Arch Dis Fetal Neonat Ed 2013; 98 (5): 440–444.
Sun B, Ma L, Liu X, Gao X, Ni L . Development of neonatal respiratory and intensive care: Chinese perspectives. Neonatology 2012; 101 (2): 77–82.
Wang H, Gao X, Liu C, Yan C, Lin X, Yang C et al. Morbidity and mortality of neonatal respiratory failure in China: surfactant treatment in very immature infants. Pediatrics 2012; 129 (3): 731–740.
Marshall DD, Kotelchuck M, Young TE, Bose CL, Kruyer L, O’Shea TM . Risk factors for chronic lung disease in the surfactant era: a North Carolina population-based study of very low birth weight infants. Pediatrics 1999; 104 (6): 1345–1350.
Walsh MC, Szefler S, Davis J, Allen M, Van Marter L, Abman S et al. Summary proceedings from the bronchopulmonary dysplasia group. Pediatrics 2006; 117 (3 Pt 2): S52–S56.
Farstad T, Bratlid D, Medbø S, Markestad T ; Norwegian Extreme Prematurity Study Group. Bronchopulmonary dysplasia-prevalence, severity and predictive factors in a national cohort of extremely premature infants. Acta Paediatr 2011; 100 (1): 53–58.
Van Marter LJ, Allred EN, Pagano M, Sanocka U, Parad R, Moore M et al. Do clinical markers of barotrauma and oxygen toxicity explain interhospital variation in rates of chronic lung disease? The Neonatology Committee for the Developmental Network. Pediatrics 2000; 105 (6): 1194–1201.
Vanpee M, Walfridsson-Schultz U, Kats-Salamon M, Zupancic JA, Pursley D, Jonsson B . Resuscitation and ventilation strategies for extremely preterm infants: a comparison study between two neonatal centers in Boston and Stockholm. Acta Paediatr 2007; 96 (1): 10–16.
Bhandari A, Bhandari V . Pitfalls, problems, and progress in bronchopulmonary dysplasia. Pediatrics 2009; 123 (6): 1562–1573.
Van Marter LJ, Allred EN, Leviton A, Pagano M, Parad R, Moore M et al. Antenatal glucocorticoid treatment does not reduce chronic lung disease among surviving preterm infants. J Pediatr 2001; 138 (2): 198–204.
Van Marter LJ, Dammann O, Allred EN, Leviton A, Pagano M, Moore M et al. Chorioamnionitis, mechanical ventilation, and postnatal sepsis as modulators of chronic lung disease in preterm infants. J Pediatr 2002; 140 (2): 171–176.
Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A et al. Caffeine therapy for apnea of prematurity. N Engl J Med 2006; 354 (20): 2112–2121.
Neonatal Inhaled Nitric Oxide Group. Inhaled nitric oxide in full-term and nearly full-term infants with hypoxic respiratory failure. N Engl J Med 1997; 336 (9): 597–604.
Van Meurs KP, Wright LL, Ehrenkranz RA, Lemons JA, Ball MB, Poole WK et al. Inhaled nitric oxide for premature infants with severe respiratory failure. N Engl J Med 2005; 353 (1): 13–22.
Kinsella JP, Walsh WF, Bose CL, Gerstmann DR, Labella JJ, Sardesai S et al. Inhaled nitric oxide in premature neonates with severe hypoxaemic respiratory failure: a randomised controlled trial. Lancet 1999; 354 (9184): 1061–1065.
Schreiber MD, Gin-Mestan K, Marks JD, Huo D, Lee G, Srisuparp P . Inhaled nitric oxide in premature infants with the respiratory distress syndrome. N Engl J Med 2003; 349 (22): 2099–2107.
Kinsella JP, Cutter GR, Walsh WE, Gerstmann DR, Bose CL, Hart C et al. Early inhaled nitric oxide therapy in premature newborns with respiratory failure. N Engl J Med 2006; 355 (4): 354–364.
Ballard RA, Truog WE, Cnaan A, Martin RJ, Ballard PL, Merrill JD et al. Inhaled nitric oxide in preterm infants undergoing mechanical ventilation. N Engl J Med 2006; 355 (4): 343–353.
Mercier JC, Hummler H, Durrmeyer X, Sanchez-Luna M, Carnielli V, Field D et al. Inhaled nitric oxide for prevention of bronchopulmonary dysplasia in premature babies (EUNO): a randomised controlled trial. Lancet 2010; 376 (9738): 346–354.
Clark RH, Ursprung RL, Walker MW, Ellsbury DL, Spitzer AR . The changing pattern of inhaled nitric oxide use in the neonatal intensive care unit. J Perinatol 2010; 30 (12): 800–804.
Dewhurst C, Ibrahim H, Göthberg S, Jonsson B, Subhedar N . European Inhaled Nitric Oxide Registry. Use of inhaled nitric oxide in the new born period: results from the European Inhaled Nitric Oxide Registry. Acta Paediatr 2010; 99 (6): 854–860.
Stenger MR, Slaughter JL, Kelleher K, Shepherd EG, Klebanoff MA, Reagan P . Hospital variation in nitric oxide use for premature infants. Pediatrics 2012; 129 (4): 945–951.
Bland RD, Albertine KH, Carlton DP, MacRitchie AJ . Inhaled nitric oxide effects on lung structure and function in chronically ventilated preterm lambs. Am J Respir Crit Care Med 2005; 172 (7): 899–906.
McCurnin DC, Pierce RA, Chang LY, Gibson LL, Osborne-Lawrence S, Yoder BA et al. Inhaled NO improves early pulmonary function and modifies lung growth and elastin deposition in a baboon model of neonatal chronic lung disease. Am J Physiol Lung Cell Mol Physiol 2005; 288 (3): 450–459.
Kinsella JP, Parker TA, Galan H, Sheridan BC, Halbower AC, Abman SH . Effects of inhaled nitric oxide on pulmonary edema and lung neutrophil accumulation in severe experimental hyaline membrane disease. Pediatr Res 1997; 41 (4 Pt 1): 457–463.
Chinese Collaborative Study Group for Neonatal Respiratory Disease. Treatment of severe meconium aspiration syndrome with porcine surfactant: a multicentre, randomized, controlled trial. Acta Paediatr 2005; 94 (7): 896–902.
Peacock JL, Peacock PJ Chapter 1. Research desgin. In: Oxford Handbook of Medical Statistics. Oxford University Press: New York, USA, 2011, pp 1–71.
Cunningham FG, MacDonald PC, Gant NF, Leveno KJ, Gilstrap LC III, Hankins GDV et al. William’s Obstetrics. McGraw-Hill Medical Press: New York, USA, 2001.
Papile LA, Burstein J, Burstein R, Koffler H . Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr 1978; 92 (4): 529–534.
George TN, Johnson KJ, Bates JN, Segar JL . The effect of inhaled nitric oxide therapy on bleeding time and platelet aggregation in neonates. J Pediatr 1998; 132 (4): 731–734.
Jobe AH, Bancalari E . Bronchopulmonary dysplasia. Am J Respir Crit Care Med 2001; 163 (7): 1723–1729.
Ehrenkranz RA, Walsh MC, Vohr BR, Jobe AH, Wright LL, Fanaroff AA et al. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics 2005; 116 (6): 1353–1360.
Wang YF, Liu CQ, Gao XR, Yang CY, Shan RB, Zhuang DY et al. Effects of inhaled nitric oxide in neonatal hypoxemic respiratory failure from a multicenter controlled trial. Chin Med J (Engl) 2011; 124 (8): 1156–1163.
Davidson D, Barefield ES, Kattwinkel J, Dudell G, Damask M, Straube R et al. Inhaled nitric oxide for the early treatment of persistent pulmonary hypertension of the term newborns: a randomized, double-masked, placebo-controlled, dose-response, multicenter study. Pediatrics 1998; 101 (3): 325–334.
Hamon I, Gauthier-Moulinier H, Grelet-Dessioux E, Storme L, Fresson J, Hascoet JM . Methaemoglobinaemia risk factors with inhaled nitric oxide therapy in newborn infants. Acta Paediatr 2010; 99 (10): 1467–1473.
Askie LM, Ballard RA, Cutter GR, Dani C, Elbourne D, Field D et al. Inhaled nitric oxide in preterm infants: an individual-patient data meta-analysis in randomized trials. Pediatrics 2011; 128 (4): 729–739.
Laughon M, Allred EN, Bose C, O’Shea TM, Van Marter LJ, Ehrenkranz RA et al. Patterns of respiratory disease during the first 2 postnatal weeks in extremely premature infants. Pediatrics 2009; 123 (4): 1124–1131.
Tarnow-Mordi W, Kumar P, Kler N . Neonatal trials need thousands, not hundreds, to change global practice. Acta Paediatr 2011; 100 (3): 330–333.
Ambalavanan N, Walsh M, Bobashev G, Das A, Levine B, Carlo WA et al. Intercenter differences of bronchopulmonary dysplasia and death among very low birth weight infants. Pediatrics 2011; 127 (1): e107–e116.
Lapcharoensap W, Gage SC, Kan P, Profit J, Shaw GM, Gould JB et al. Hospital variation and risk factors for bronchopulmonary dysplasia in a population-based cohort. JAMA Pediatrics 2015; 169 (2): e143676.
Van Rossem MC, Van de Loo M, Laan BJ, de Sonnaville ESV, Tamminga P, van Kaam AH et al. Accuracy of the diagnosis of bronchopulmonary dysplasia in a referral-based health care system. J Pediatr 2015; 167 (3): 540–544.
Ellsworth MA, Harris MN, Carey WA, Spitzer AR, Clark RH . Off-label use of inhaled nitric oxide after release of NIH consensus statement. Pediatrics 2015; 135 (4): 643–648.
Kinsella JP, Cutter GR, Steinhorn RH, Nelin LD, Walsh WF, Finer NN et al. Noninvasive inhaled nitric oxide does not prevent bronchopulmonary dysplasia in premature newborns. J Pediatr 2014; 165 (6): 1104–1108.
Sun L, Yue H, Sun B, Han L, Qi M, Tian Z et al. Estimation of birth population-based perinatal-neonatal mortality and preterm rate in China from a regional survey in 2010. J Matern Fetal Neonat Med 2013; 26 (16): 1641–1648.
National Bureau of Statistics of China Tabulations on the 2010 Population Census of the People’s Republic of China. China Statistics Press: Beijing, China, 2012, www.stats.gov.cn.
Acknowledgements
We thank Dr Richard David, Department of Pediatrics, University of Illinois College of Medicine at Stroger Hospital of Cook County for his critical reading and constructive comments on this manuscript in revision and Dr Weili Yan, Unit of Clinical Epidemiology, Children’s Hospital of Fudan University for her advice in study design, data presentation and statistical analysis of the manuscript. This study was supported by grants for Excellence in Neonatal Medicine, National Commission of Health and Family Planning of China, the Project 211 (phase III), Ministry of Education and Shanghai Municipal Department of Health (No. LJ06038).
List of institutes and investigators
The following institutes (city, province or national special municipality) and investigators participated in the Chinese Collaborative Study Group for Neonatal Respiratory Disease (NO-BPD Project): Children’s Hospital of Fudan University, Shanghai (Bo Sun (study director), Qian Jiang (coordinator), Chao Chen, Yun Cao); Hunan Provincial Children’s Hospital, Changsha, Hunan (Xirong Gao, Rong Zhang); Hebei Provincial Children’s Hospital, Shijiazhuang, Hebei (Cuiqing Liu, Yanchun Cui); Quanzhou Children’s Hospital, Quanzhou, Fujian (Dongmei Chen, Guofeng Lei); Xiamen Maternity Hospital, Xiamen, Fujian (Xinzhu Lin, Jie Huang); Hubei Provincial Maternity Hospital, Wuhan, Hubei (Shiwen Xia); First Hospital of Xiamen University, Xiamen, Fujian (Deyi Zhuang, Ying Huang); Shenzhen Maternity and Child Health Care Hospital (Chuanzhong Yang); Chenzhou Children’s Hospital, Chenzhou, Hunan (Wenjun Zhu, Xiaoqin Gong); Guiyang Maternity Hospital, Guiyang, Guizhou (Ling Liu, Wei Wang); Guangxi Autonomous Regional Maternity Hospital, Nanning, Guangxi (Qiufen Wei); Shenzhen Children’s Hospital, Shenzhen, Guangdong (Xiaohong Liu); Guangdong General Hospital, Guangzhou, Guangdong (Shaoru He, Yifei Wang); Gansu Provincial Maternity Hospital, Lanzhou, Gansu (Bin Yi, Yue Zhang); Tianjin Central Hospital of Obstetrics and Gynecology, Tianjin (Jun Zheng, Xiuying Tian); Tai’an Maternity Hospital, Tai’an, Shandong (Zhenying Yang, Ji Qi); Tai’an Central Hospital, Tai’an, Shandong (Guo Yao, Yan Wang); Shenzhen Bao’an Maternity Hospital, Shenzhen, (Guangjin Lu, Zhijun Wu, Shanqiu Xiao); Zhengzhou Children’s Hospital, Zhengzhou, Henan (Wenqing Kang); Qingdao Women and Children’s Hospital, Qingdao, Shandong (Ruobing Shan, Shaoqing Guo); Anhui Provincial Children’s Hospital, Hefei, Anhui (Yanna Fu, Liying Dai); Nanjing Maternity and Child Health Care Hospital, Nanjing, Jiangsu (Shuping Han, Xiaoyue Dong); Shaanxi Provincial General Hospital, Xi’an, Shaanxi(Qin Zhang); Huai’an Maternity Hospital, Huai’an, Jiangsu (Hongni Yue, Liangrong Han); Shenzhen Nanshan Hospital, Shenzhen (Xiaodong Li); The Fourth Hospital of Xi’An, Xi’An, Shaanxi (Liming Ni); and Handan Maternity Hospital (Hongyan Lv).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Perinatology website
Supplementary information
Rights and permissions
About this article
Cite this article
Jiang, Q., Gao, X., Liu, C. et al. Early inhaled nitric oxide in preterm infants <34 weeks with evolving bronchopulmonary dysplasia. J Perinatol 36, 883–889 (2016). https://doi.org/10.1038/jp.2016.112
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2016.112
- Springer Nature America, Inc.
This article is cited by
-
Response categorization and outcomes in extremely premature infants born at 22–26 weeks gestation that received inhaled nitric oxide for hypoxic respiratory failure
Journal of Perinatology (2023)
-
Use of inhaled nitric oxide in preterm vs term/near-term neonates with pulmonary hypertension: results of the PaTTerN registry study
Journal of Perinatology (2022)
-
Outcome of neonatal hypoxemic respiratory failure: a livebirth population-based retrospective survey
BMC Pediatrics (2022)