Abstract
Tumor necrosis factor α (TNF-α) induces the activation of transcription factor nuclear factor κB (NF-κB), which upregulates a variety of genes, including the gene encoding intercellular adhesion molecule-1 (ICAM-1). Allantopyrone A, a recently identified α-pyrone metabolite from an endophytic fungus, was found to inhibit the TNF-α-induced expression of ICAM-1 in human lung carcinoma A549 cells. Allantopyrone A also inhibited the TNF-α-induced luciferase expression of an NF-κB-responsive reporter. In the NF-κB signaling pathway, allantopyrone A inhibited the nuclear translocation of NF-κB subunits as well as the phosphorylation and subsequent degradation of the inhibitor of NF-κB (IκB) α proteins. By contrast, allantopyrone A did not directly affect the catalytic activity of active IκB kinase β. These findings indicate that allantopyrone A inhibits the NF-κB signaling pathway at a step upstream of IκBα phosphorylation.
Similar content being viewed by others
Introduction
Proinflammatory cytokines, such as tumor necrosis factor α (TNF-α), induce intracellular signaling pathways, one of which leads to the activation of transcription factor nuclear factor κB (NF-κB).1 NF-κB plays a role in the expression of many genes essential for inflammation and tumor promotion.2 In response to TNF-α stimulation, TNF receptor 1 recruits the TNF receptor-associated death domain (TRADD), followed by the recruitment of other adaptor proteins, including receptor-interacting protein 1 (RIP1).3, 4 RIP1 is then ubiquitinated and thereby recruits the TGF-β-activated kinase 1 (TAK1) complex and the inhibitor of the NF-κB (IκB) kinase complex in its polyubiquitin chains.5 The IκB kinase complex is subsequently activated and the activated complex phosphorylates N-terminal serine residues of IκB, which promotes IκB polyubiquitination and proteolysis by proteasomes.6, 7 NF-κB heterodimers, such as p65/p50, are associated with IκB in the cytoplasm, and following IκB degradation they are released and translocate to the nucleus.8 NF-κB heterodimers activate the transcription of target genes, including the gene encoding intercellular adhesion molecule-1 (ICAM-1).9
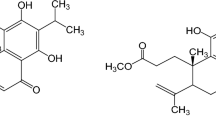
We have recently isolated a new α-pyrone metabolite designated allantopyrone A (Figure 1) from an endophytic fungus, Allantophomopsis lycopodina KS-97.10 Allantopyrone A was found to have high cytotoxicity and induce DNA fragmentation in human leukemia HL-60 cells.10 Thus far, the biological activities of allantopyrone A have been poorly understood, except that allantopyrone A possesses an α,β-unsaturated carbonyl moiety, which reacts with cysteinyl thiol groups. It has been reported that natural and synthetic compounds possessing an α,β-unsaturated carbonyl moiety inhibit multiple steps of the NF-κB signaling pathway.11 We have found that the inhibitory effect of allantopyrone A on the TNF-α-induced expression of ICAM-1 is stronger than its cytotoxic effect on human lung carcinoma A549 cells. In this study, we further investigated the molecular mechanism underlying the inhibition of ICAM-1 expression by allantopyrone A.
Materials and methods
Cell culture
Human lung carcinoma A549 cells (JCRB0076) were provided by the National Institute of Biomedical Innovation JCRB Cell Bank (Osaka, Japan). A549 cells were maintained in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% heat-inactivated fetal calf serum (Nichirei Biosciences, Tokyo, Japan) and a penicillin-streptomycin mixed solution (Nacalai Tesque, Kyoto, Japan). Human embryonic kidney (HEK) 293C8L cells were maintained in Dulbecco’s modified Eagle Medium supplemented with 10% heat-inactivated fetal calf serum and the penicillin-streptomycin mixed solution.12
Reagents
Allantopyrone A was prepared as described previously.10 15-deoxy-Δ12,14-prostaglandin J2 (15d-Δ12,14-PGJ2) was obtained from Cayman Chemical (Ann Arbor, MI, USA).
Cell-enzyme-linked immunosorbent assay
The cell-surface expression level of ICAM-1 was measured by cell-enzyme-linked immunosorbent assay as described previously.13, 14 The expression level of ICAM-1 (%) was calculated using the following formula: (test samples−background)/(positive control−background) × 100.
Assay of cell viability
Cell viability was measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay as described previously,13, 14 except that A549 cells were pulsed with MTT (500 μg ml–1) for 2 h.
Reporter assay
The NF-κB-responsive firefly luciferase gene was controlled by two copies of κB sequences from the Igκ enhancer.15 A549 cells were transfected with reporter vectors encoding the NF-κB-responsive firefly luciferase gene and a CMV-promoter-driven Renilla luciferase gene using HilyMax transfection reagent (Dojindo Laboratories, Kumamoto, Japan) for 8 h. The transfected A549 cells were seeded in 24-well culture plates and incubated overnight. The reporter assay of the cells was performed as described previously.12
Western blotting
Cell lysates were prepared and western blotting was performed as described previously.16 Antibodies to β-actin (AC-15; Sigma-Aldrich, St Louis, MO, USA), IκBα (clone 25; BD Biosciences, Franklin Lakes, NJ, USA), IκB kinase β (10A9B6; BD Biosciences), NF-κB p50 (H-119; Santa Cruz Biotechnology, Santa Cruz, CA, USA), NF-κB p65 (C-20; Santa Cruz Biotechnology), phospho-IκBα(Ser32/36) (5A5; Cell Signaling Technology, Danvers, MA, USA) and poly(ADP-ribose) polymerase (C-2-10; Sigma-Aldrich) were used for western blotting. The protein bands were analyzed using an ImageQuant LAS 4000 Mini (GE Healthcare, Piscataway, NJ, USA).
IκB kinase assay
IκB kinase assay was performed as described previously.17 HEK293C8L cells were transfected with the pCR3 expression vector encoding FLAG-tagged human IκB kinase β by the calcium phosphate method. Cell lysates were prepared using NP-40 lysis buffer and immunoprecipitated with anti-FLAG antibody-conjugated agarose beads (Sigma-Aldrich). The obtained immunoprecipitates were incubated with glutathione S-transferase (GST)-tagged IκBα(1–54) (1.5 μg) at 37 °C for 30 min in the presence or absence of ATP (10 μM). Reaction mixtures were analyzed by western blotting.
Statistical analysis
Statistical significance was assessed by one-way analysis of variance followed by Tukey’s test for multiple comparisons. Differences with P<0.05 were considered to be statistically significant.
Results
Allantopyrone A inhibits TNF-α-induced expression of ICAM-1
TNF-α stimulation markedly induces the cell-surface expression of ICAM-1 in an NF-κB-dependent manner in A549 cells.18 A549 cells were preincubated with allantopyrone A for 1 h and then incubated with TNF-α for 6 h in the presence of allantopyrone A. Allantopyrone A at concentrations higher than 32 μM inhibited the TNF-α-induced ICAM-1 expression almost completely (Figure 2a). For the same incubation period, allantopyrone A at 100 μM decreased cell viability by ∼30% (Figure 2b), indicating that allantopyrone A exerts a moderately strong cytotoxic effect within 7 h on A549 cells.
Allantopyrone A inhibits TNF-α-induced ICAM-1 expression. (a) A549 cells were preincubated with allantopyrone A at various concentrations for 1 h and then incubated with (filled circles) or without (open circles) TNF-α (2.5 ng ml–1) for 6 h in the presence of allantopyrone A. ICAM-1 expression (%) is shown as the mean±s.e. of three independent experiments. **P<0.01, compared with control. (b) A549 cells were incubated with allantopyrone A at various concentrations for 7 h. Cell viability (%) is shown as the mean±s.e. of three independent experiments. **P<0.01, compared with control.
Allantopyrone A inhibits TNF-α-induced NF-κB-dependent luciferase activity
To minimize the cytotoxic effect of allantopyrone A, A549 cells were exposed to allantopyrone A for a shorter period in further experiments. A549 cells were preincubated with allantopyrone A for 1 h and then incubated with TNF-α for 2.5 h in the presence of allantopyrone A. TNF-α induced an approximately eightfold increase in NF-κB-dependent luciferase reporter activity (Figure 3a). Allantopyrone A at concentrations higher than 32 μM markedly decreased the TNF-α-induced NF-κB luciferase reporter activity (Figure 3a). By contrast, allantopyrone A did not affect cell viability at these concentrations up to 3.5 h (Figure 3b).
Allantopyrone A inhibits TNF-α-induced NF-κB-responsive reporter activity. (a) A549 cells were transfected with reporter vectors encoding an NF-κB-responsive luciferase gene and a CMV promoter-driven Renilla luciferase gene. A549 cells were preincubated with allantopyrone A at various concentrations for 1 h and then incubated with (+) or without (–) TNF-α (2.5 ng ml–1) for 2.5 h in the presence of allantopyrone A. NF-κB activation (fold) is shown as means±s.e. of three independent experiments. *P<0.05 and **P<0.01, compared with TNF-α (+). (b) A549 cells were incubated with various concentrations of allantopyrone A for 3.5 h. Cell viability (%) is shown as the mean±s.e. of three independent experiments. **P<0.01, compared with control.
Allantopyrone A inhibits TNF-α-induced nuclear translocation of NF-κB subunits
The NF-κB p65/p50 heterodimers are localized in the cytoplasm and translocate to the nucleus upon TNF-α stimulation.16, 18 A549 cells were preincubated with allantopyrone A for 1 h and then incubated with TNF-α for 30 min in the presence of allantopyrone A. TNF-α augmented the amounts of p65 and p50 in the nuclear fraction within 30 min (Figure 4). Allantopyrone A at concentrations higher than 32 μM significantly inhibited the accumulation of nuclear p65 and p50 (Figure 4).
Allantopyrone A inhibits TNF-α-induced nuclear translocation of NF-κB subunits. A549 cells were preincubated with allantopyrone A at various concentrations for 1 h and then incubated with (+) or without (–) TNF-α (2.5 ng ml–1) for 30 min in the presence of allantopyrone A. Nuclear and cytoplasmic fractions were analyzed by western blotting. The amounts of nuclear p65 and p50 were normalized with that of PARP. The amounts of nuclear p65 and p50 (%) are shown as the mean±s.e. of three independent experiments. **P<0.01, compared with TNF-α (+).
Allantopyrone A inhibits TNF-α-induced IκBα phosphorylation and degradation
TNF-α stimulation induces IκBα phosphorylation within 5 min and the subsequent IκBα proteolysis by the ubiquitin-proteasome system within 15 min in A549 cells.19 A549 cells were preincubated with allantopyrone A for 1 h and then incubated with TNF-α for 15 min in the presence of allantopyrone A. The amount of IκBα markedly decreased 15 min after TNF-α stimulation (Figure 5a). Allantopyrone A at concentrations higher than 32 μM inhibited the TNF-α-induced IκBα degradation (Figure 5a).
Allantopyrone A inhibits TNF-α-induced phosphorylation and degradation of IκBα. (a) A549 cells were preincubated with allantopyrone A at various concentrations for 1 h and then incubated with (+) or without (–) TNF-α (2.5 ng ml–1) for 15 min in the presence of allantopyrone A. Cell lysates were analyzed by western blotting. The amount of IκBα was normalized with that of β-actin. IκBα (%) is shown as the mean±s.e. of four independent experiments. **P<0.01, compared with TNF-α (+). (b) A549 cells were preincubated with (+) or without (–) allantopyrone A (100 μM) for 1 h and then incubated with TNF-α (2.5 ng ml–1) for indicated times in the presence of allantopyrone A. Cell lysates were analyzed by western blotting. The amounts of phospho-IκBα and total IκBα were normalized with that of β-actin. Phospho-IκBα (fold) and IκBα (%) are shown as the mean±s.e. of three independent experiments. *P<0.05 and **P<0.01, compared with control.
The amount of the phosphorylated form of IκBα transiently increased 5 min following TNF-α stimulation (Figure 5b). Allantopyrone A at 100 μM inhibited the phosphorylation and subsequent degradation of IκBα almost completely (Figure 5b). These findings suggest that allantopyrone A blocks the NF-κB signaling pathway at a step upstream of IκBα phosphorylation.
Allantopyrone A does not directly inhibit the catalytic activity of IκB kinase β
FLAG-tagged IκB kinase β was transiently overexpressed in HEK293C8L cells and immunoprecipitated with anti-FLAG-antibody-conjugated beads. The immunoprecipitates containing IκB kinase β were capable of phosphorylating GST-tagged IκBα(1–54) in the presence of ATP. As reported previously,20, 21 15d-Δ12,14-PGJ2 markedly inhibited the phosphorylation of IκBα(1–54) (Figure 6). The MW of IκB kinase β treated with 15d-Δ12,14-PGJ2 slightly increased (Figure 6), suggesting that 15d-Δ12,14-PGJ2 binds covalently to IκB kinase β. By contrast, allantopyrone A at concentrations up to 100 μM did not inhibit the phosphorylation of IκBα(1–54) (Figure 6). These findings suggest that allantopyrone A does not directly inhibit the catalytic activity of active IκB kinase β.
Allantopyrone A does not directly affect IκB kinase β. HEK293C8L cells were transfected with an expression vector encoding FLAG-tagged IκB kinase β for 30 h. Cell lysates were immunoprecipitated with anti-FLAG antibody-conjugated agarose beads. Immunoprecipitates were incubated with GST-tagged IκBα(1–54) in the presence (+) or absence (−) of ATP, allantopyrone A (at indicated concentrations) or 15d-Δ12,14-PGJ2 (50 μM) for 30 min. The amount of phospho-IκBα(1–54) (GST-tagged) was normalized with that of FLAG-tagged IκB kinase β. Phosphorylation (%) is shown as the mean±s.e. of three independent experiments. **P<0.01, compared with control.
Discussion
We have recently isolated allantopyrone A from A. lycopodina KS-97.10 Allantopyrone A has been shown to have high cytotoxicity and induce DNA fragmentation in HL-60 cells.10 Thus far, the biological activities of allantopyrone A have been poorly characterized. In line with our previous finding,10 allantopyrone A decreased the viability of A549 cells by ∼30% following the 7-h incubation. More importantly, we found the novel biological activity that allantopyrone A selectively inhibits the NF-κB signaling pathway in A549 cells under conditions that do not affect cell viability.
As compounds structurally related to allantopyrone A, islandic acid-I and islandic acid-II as well as their methyl esters were previously isolated from Penicillium islandicum Sopp.22, 23 The islandic acid-I and islandic acid-I methyl esters showed a 100% inhibitory activity against the growth of Yoshida sarcoma cells (rat tumor cells) at concentrations of 100 μg ml−l (286 μM) and 1 μg ml−1 (2.7 μM), respectively.23 We previously showed that allantopyrone A and islandic acid-II methyl ester exert the cytotoxic effect on HL-60 cells at IC50 values of 0.32 and 6.55 μM, respectively.10 Thus, it seems that allantopyrone A is one of most potent compounds with antitumor activity among the α-pyrone metabolites.
Allantopyrone A possesses an α,β-unsaturated carbonyl moiety that is reactive to cysteinyl thiol groups by the Michael addition. It has been thus far reported that natural and synthetic compounds possessing an α,β-unsaturated carbonyl moiety inhibit multiple steps in the NF-κB signaling pathway.11 IκB kinase β contains a Cys-179 residue within its activation loop in which two serine residues (Ser-177 and Ser-181) are phosphorylated for kinase activation.20, 24 It has been reported that 15d-Δ12,14-PGJ2 targets Cys-179 of IκB kinase β and inhibits its catalytic activity.20, 21 In contrast to 15d-Δ12,14-PGJ2, allantopyrone A did not directly inhibit IκB kinase β activity. However, allantopyrone A inhibited the TNF-α-induced IκBα phosphorylation at the cellular level. These findings suggest that allantopyrone A inhibits a step upstream of the activation of IκB kinase in the NF-κB signaling pathway.
We previously showed that peperomin E and 2,6-didehydropeperomin B that possess an α-methylene-γ-lactone moiety reactive to thiol residues, but not peperomin A and peperomin B that possess an α-methyl-γ-lactone moiety unreactive to thiol residues, inhibit the TNF-α-induced activation of IκB kinase in A549 cells.17 However, similar to allantopyrone A, peperomin E and 2,6-didehydropeperomin B were not capable of inhibiting IκB kinase β catalytic activity directly.17 These findings collectively imply that certain protein(s) containing cysteine residues critical for their biological activities are required for the signaling process upstream of the activation of IκB kinase. TAK1 might be one of the candidate proteins, because it has been reported that epoxyquinol B inhibits NF-κB signaling by crosslinking TAK1 through cysteine residues.25
In conclusion, we have shown that allantopyrone A inhibits the TNF-α-induced NF-κB signaling pathway at a step upstream of IκBα phosphorylation. NF-κB targets many genes required for cancer development and progression.26 Thus, agents that block the NF-κB signaling pathway are expected to have high anticancer activity. It is currently unclear whether the inhibition of NF-κB activation by allantopyrone A is related to the apoptosis-inducing activity of allantopyrone A, as observed in HL-60 cells. Further experiments will be necessary to clarify the molecular mechanism by which allantopyrone A inhibits the NF-κB signaling pathway or induces apoptosis.
References
Aggarwal, B. B. Signalling pathways of the TNF superfamily: a double-edged sword. Nat. Rev. Immunol. 3, 745–756 (2003).
Karin, M. & Greten, F. R. NF-κB: linking inflammation and immunity to cancer development and progression. Nat. Rev. Immunol. 5, 749–759 (2005).
Hayden, M. S. & Ghosh, S. Shared principles in NF-κB signaling. Cell 132, 344–362 (2008).
Wajant, H. & Scheurich, P. TNFR1-induced activation of the classical NF-κB pathway. FEBS J. 278, 862–876 (2011).
Bhoj, V. G. & Chen, Z. J. Ubiquitylation in innate and adaptive immunity. Nature 458, 430–437 (2009).
Perkins, N. D. Post-translational modifications regulating the activity and function of the nuclear factor kappa B pathway. Oncogene 25, 6717–6730 (2006).
Scheidereit, C. IκB kinase complexes: gateways to NF-κB activation and transcription. Oncogene 25, 6685–6705 (2006).
Ghosh, S. & Hayden, M. S. New regulators of NF-κB in inflammation. Nat. Rev. Immunol. 8, 837–848 (2008).
Roebuck, K. A. & Finnegan, A. Regulation of intercellular adhesion molecule-1 (CD54) gene expression. J. Leukoc. Biol. 66, 876–888 (1999).
Shiono, Y. et al. Allantopyrone A, a new α-pyrone metabolite with potent cytotoxicity from an endophytic fungus, Allantophomopsis lycopodina KS-97. J. Antibiot. 63, 251–253 (2010).
Kataoka, T. Chemical biology of inflammatory cytokine signaling. J. Antibiot. 62, 655–667 (2009).
Matsuda, I. et al. The C-terminal domain of the long form of cellular FLICE-inhibitory protein (c-FLIPL) inhibits the interaction of the caspase 8 prodomain with the receptor-interacting protein 1 (RIP1) death domain and regulates caspase-8-dependent nuclear factor κB (NF-κB) activation. J. Biol. Chem. 289, 3876–3887 (2014).
Takada, Y. et al. Odoroside A and ouabain inhibit Na+/K+-ATPase and prevent NF-κB-inducible protein expression by blocking Na+-dependent amino acid transport. Biochem. Pharmacol. 78, 1157–1166 (2009).
Yamada, Y., Tashiro, E., Taketani, S., Imoto, M. & Kataoka, T. Mycotrienin II, a translation inhibitor that prevents ICAM-1 expression induced by pro-inflammatory cytokines. J. Antibiot. 64, 361–366 (2011).
Dohrman, A. et al. Cellular FLIP (long form) regulates CD8+ T cell activation through caspase-8-dependent NF-κB activation. J. Immunol. 174, 5270–5278 (2005).
Hirano, S. & Kataoka, T. Deoxynivalenol induces ectodomain shedding of TNF receptor 1 and thereby inhibits the TNF-α-induced NF-κB signaling pathway. Eur. J. Pharmacol. 701, 144–151 (2013).
Tsutsui, C. et al. Peperomins as anti-inflammatory agents that inhibit the NF-κB signaling pathway. Bioorg. Med. Chem. Lett. 19, 4084–4087 (2009).
Tamura, R. et al. Eudesmane-type sesquiterpene lactones inhibit multiple steps in the NF-κB signaling pathway induced by inflammatory cytokines. Bioorg. Med. Chem. Lett. 22, 207–211 (2012).
Ogura, H. et al. Ectodomain shedding of TNF receptor 1 induced by protein synthesis inhibitors regulates TNF-α-mediated activation of NF-κB and caspase-8. Exp. Cell Res. 314, 1406–1414 (2008).
Rossi, A. et al. Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IκB kinase. Nature 403, 103–108 (2000).
Straus, D. S. et al. 15-Deoxy-Δ12,14-prostaglandin J2 inhibits multiple steps in the NF-κB signaling pathway. Proc. Natl. Acad. Sci. USA 97, 4844–4849 (2000).
Fujimoto, Y., Tsunoda, H., Uzawa, J. & Tatsuno, T. Structure of islandic acid, a new metabolite from Penicillium islandicum Sopp. J. Chem. Soc. Chem. Commun. 1982, 83–84 (1982).
Fujimoto, Y. et al. Studies on the biological activities of islandic acid and related compounds. Chem. Pharm. Bull. 32, 1583–1586 (1984).
Kapahi, P. et al. Inhibition of NF-κB activation by arsenite through reaction with a critical cysteine in the activation loop of IκB kinase. J. Biol. Chem. 275, 36062–36066 (2000).
Kamiyama, H. et al. Epoxyquinol B, a naturally occurring pentaketide dimer, inhibits NF-κB signaling by crosslinking TAK1. Biosci. Biotechnol. Biochem. 72, 1894–1900 (2008).
Baud, V. & Karin, M. Is NF-κB a good target for cancer therapy? Hopes and pitfalls. Nat. Rev. Drug Discov. 8, 33–40 (2009).
Acknowledgements
This work was supported in part by Grants-in-Aid for Scientific Research (KAKENHI) Grant Numbers 22380060 and 25292061 (to T.K.) from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yokoigawa, J., Morimoto, K., Shiono, Y. et al. Allantopyrone A, an α-pyrone metabolite from an endophytic fungus, inhibits the tumor necrosis factor α-induced nuclear factor κB signaling pathway. J Antibiot 68, 71–75 (2015). https://doi.org/10.1038/ja.2014.103
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2014.103
- Springer Japan KK