Abstract
Background
Alzheimer’s disease (AD) is characterized by amyloid beta (Aβ) plaques, neurofibrillary tangles (NFTs) and cognitive impairment. Literature cites the role of advanced glycation end products (AGEs) in AD due to increased cytotoxicity via oxidative stress. d-galactose (d-gal) induced amnesia stimulates Aβ overproduction via increased oxidative stress and AGEs. Trigonelline (TRG), a naturally occurring alkaloid has been reported to have neuroprotective and antidiabetic properties.
Methods
Present study assessed the protective effect of TRG against in vitro AGEs formation. Since chronic administration of d-gal increases AGEs, we subsequently investigated the neuroprotective role of TRG (50 and 100 mg/kg as per body weight) against d-gal induced amnesia. Mice were subcutaneously (sc) injected with d-gal (150 mg/kg) for 6 weeks. Behavioral assessments in Morris water maze (MWM) and Y-maze were performed, followed by biochemical estimations to deduce the probable mechanism of action.
Results
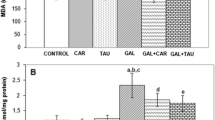
In vitro experiments demonstrated that TRG stalled early and late AGEs formation. Chronic d-gal administration significantly impaired cognitive performance in MWM and Y maze, caused marked oxidative damage, elevated the AGEs levels and significantly increased the acetylcholinesterase levels as compared to sham group. TRG (50 and 100 mg/kg) treatment significantly ameliorated cognitive performance, reversed the oxidative damage, decreased AGE levels and caused significant decline in acetylcholine esterase levels as compared to d-gal group.
Conclusion
Present study highlights the neuroprotective role of TRG against d-gal induced amnesia due to the antioxidant, antiglycative and anticholinesterase properties.
Similar content being viewed by others
Abbreviations
- Aβ:
-
amyloid beta
- AChE:
-
acetyl cholinesterase
- AD:
-
Alzheimer’s disease
- AG:
-
aminoguanidine
- AGEs:
-
advanced glycation end products
- ANOVA:
-
analysis of variance
- AU:
-
arbitrary units
- BSA:
-
bovine serum albumin
- d-gal:
-
d-galactose
- DNP:
-
Donepezil
- GSH:
-
reduced glutathione
- MDA:
-
malonaldehyde
- MWM:
-
Morris water maze
- NADPH:
-
nicotinamide adenine dinucleotide phosphate oxidase
- NFTs:
-
neurofibrillary tangles
- PBS:
-
phosphate buffer saline
- ROS:
-
reactive oxygen species
- RFU:
-
relative fluorescence units
- SAB:
-
spontaneous alteration behaviour
- SD:
-
standard deviation
- SOD:
-
superoxide dismutase
- SEM:
-
standard error of the mean
- TRG:
-
trigonelline
References
Association As. Alzheimer’s disease facts and figures. Alzheimers Dement 2016;12(4):459–509.
Behl C. Alzheimer’s disease and oxidative stress: implications for novel therapeutic approaches. Prog Neurobiol 1999;57(3):301–23.
Sasaki N, Fukatsu R, Tsuzuki K, Hayashi Y, Yoshida T, Fujii N, et al. Advanced glycation end products in Alzheimer’s disease and other neurodegenerative diseases. Am J Pathol 1998;153(4):1149–55.
Sato T, Shimogaito N, Wu X, Kikuchi S, Yamagishi SI, Takeuchi M. Toxic advanced glycation end products (TAGE) theory in Alzheimer’s disease. Am J Alzheimers Dis Other Dement 2006;21(3):197–208.
Li J, Liu D, Sun L, Lu Y, Zhang Z. Advanced glycation end products and neurodegenerative diseases: mechanisms and perspective. J Neurol Sci 2012;317(1):1–5.
Dukic-Stefanovic S, Schinzel R, Riederer P, Münch G. AGES in brain ageing: AGE-inhibitors as neuroprotective and anti-dementia drugs? Biogerontology 2001;2(1):19–34.
Loske C, Neumann A, Cunningham A, Nichol K, Schinzel R, Riederer P, et al. Cytotoxicity of advanced glycation endproducts is mediated by oxidative stress. J Neural Transm 1998;105(8–9):1005–15.
Cui X, Zuo P, Zhang Q, Li X, Hu Y, Long J, et al. Chronic systemic D-galactose exposure induces memory loss, neurodegeneration, and oxidative damage in mice: protective effects of R-α-lipoic acid. J Neurosci Res 2006;83(8):1584–90.
Kumar A, Prakash A, Dogra S. Naringin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress induced by D-galactose in mice. Food Chem Toxicol 2010;48(2):626–32.
Wu DM, Lu J, Zhe YL, Zhou Z, Shan Q, Ma DF. Purple sweet potato color repairs D-galactose-induced spatial learning and memory impairment by regulating the expression of synaptic proteins. Neurobiol Learn Mem 2008;90(1):19–27.
Song X, Bao M, Li D, Li YM. Advanced glycation in D-galactose induced mouse aging model. Mech Ageing Dev 1999;108(3):239–51.
Sung JS, Go EB, Shin HS. Cytoprotective activity of extract of roasted coffee residue on mouse embryonic fibroblasts cells against apoptosis induced by oxidative stress. Food Sci Biotechnol 2012;21(1):137–43.
Mayakrishnan T, Nakkala JR, Jeepipalli SPK, Raja K, Chandra VK, Mohan VK, et al. Fenugreek seed extract and its phytocompounds-trigonelline and diosgenin arbitrate their hepatoprotective effects through attenuation of endoplasmic reticulum stress and oxidative stress in type 2 diabetic rats. Eur Food Res Technol 2015;240(1):223–32.
Tohda C, Kuboyama T, Komatsu K. Search for natural products related to regeneration of the neuronal network. Neurosignals 2005;14(1–2):34–45.
Yoshinari O, Takenake A, Igarashi K. Trigonelline ameliorates oxidative stress in type 2 diabetic Goto-Kakizaki rats. J Med Food 2013;16(1):34–41.
Yoshinari O, Sato H, Igarashi K. Anti-diabetic effects of pumpkin and its components, trigonelline and nicotinic acid, on Goto-Kakizaki rats. Biosci Biotechnol Biochem 2009;73(5):1033–41.
Kamble HV, Bodhankar SL. Antihyperglycemic activity of trigonelline and sitagliptin in nicotinamide-streptozotocin induced diabetes in wistar rats. Biomed Aging Pathol 2013;3(3):125–30.
Ghule AE, Jadhav SS, Bodhankar SL. Trigonelline ameliorates diabetic hypertensive nephropathy by suppression of oxidative stress in kidney and reduction in renal cell apoptosis and fibrosis in streptozotocin induced neonatal diabetic (nSTZ) rats. Int Immunopharmacol 2012;14(4):740–8.
Zhou JY, Zhou SW. Protection of trigonelline on experimental diabetic peripheral neuropathy. Evid Based Complement Alternat Med 2012164219.
Sakai M, Oimori M, Kasuga M. Experimental studies on the role of fructose in the development of diabetes complications. KobeJ Med Sci 2002;48(5):125–36.
Islam MN, Ishita IJ, Jung HA, Choi JS. Vicenin 2 isolated from Artemisia capillaris exhibited potent anti-glycation properties. Food Chem Toxicol 2014;69:55–62.
Joshi R, Garabadu D, Teja GR, Krishnamurthy S. Silibinin ameliorates LPS-induced memory deficits in experimental animals. Neurobiol Learn Mem 2014;116:117–31.
Ellman GL, Courtney KD, Andres V, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 1961;7(2):88–95.
Nandi A, Chatterjee I. Assay of superoxide dismutase activity in animal tissues. J Biosci 1988;13(3):305–15.
Smith IK, Vierheller TL, Thorne CA. Assay of glutathione reductase in crude tissue homogenates using 5, 5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 1988;175(2):408–13.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979;95(2):351–8.
Gornall AG, Bardawill CJ, David MM. Determination of serum proteins by means of the biuret reaction. J Biol Chem 1949;177(2):751–66.
Jagtap A, Patil P. Antihyperglycemic activity and inhibition of advanced glycation end product formation by Cuminum cyminum in streptozotocin induced diabetic rats. Food Chem Toxicol 2010;48(8):2030–6.
Münch G, Mayer S, Michaelis J, Hipkiss AR, Riederer P, Müller R, et al. Influence of advanced glycation end-products and AGE-inhibitors on nucleation-dependent polymerization of β-amyloid peptide. Biochim Biophys Acta 1997;1360(1):17–29.
Ko SY, Ko HA, Chu KH, Shieh TM, Chi TC, Chen HI, et al. The possible mechanism of advanced glycation end products (AGEs) for Alzheimer’s disease. PLoS One 2015;10(11):e0143345.
Smith MA, Taneda S, Richey PL, Miyata S, Yan SD, Stern D, et al. Advanced maillard reaction end products are associated with Alzheimer disease pathology. Proc Natl Acad Sci U S A 1994;91(12):5710–4.
Lu J, Wu DM, Hu B, Cheng W, Zheng YL, Zhang ZF, et al. Chronic administration of troxerutin protects mouse brain against D-galactose-induced impairment of cholinergic system. Neurobiol Learn Mem 2010;93(2):157–64.
Xian YF, Su ZR, Chen JN, Lai XP, Mao QQ, Cheng CH, et al. Isorhynchophylline improves learning and memory impairments induced by D-galactose in mice. Neurochem Int 2014;76:42–9.
Kim DH, Hung TM, Bae KH, Jung JW, Lee S, Yoon BH, et al. Gomisin A improves scopolamine-induced memory impairment in mice. Eur J Pharmacol 2006;542(1):129–35.
Singh M, Kaur M, Kukreja H, Chugh R, Silakari O, Singh D. Acetylcholinesterase inhibitors as Alzheimer therapy: from nerve toxins to neuroprotection. Eur J Med Chem 2013;70:165–88.
Satheeshkumar N, Mukherjee PK, Bhadra S, Saha B. Acetylcholinesterase enzyme inhibitory potential of standardized extract of Trigonella foenum graecum L and its constituents. Phytomedicine 2010;17(3):292–5.
Panda S, Biswas S, Kar A. Trigonelline isolated from fenugreek seed protects against isoproterenol-induced myocardial injury through down-regulation of Hsp27 and αB-crystallin. Nutrition 2013;29(11):1395–403.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chowdhury, A.A., Gawali, N.B., Bulani, V.D. et al. In vitro antiglycating effect and in vivo neuroprotective activity of Trigonelline in d-galactose induced cognitive impairment. Pharmacol. Rep 70, 372–377 (2018). https://doi.org/10.1016/j.pharep.2017.09.006
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2017.09.006