Abstract
Objectives
To investigate the cytotoxicity of FM-Nov17 against chronic myeloid leukemia (CML) cells, we explored its underlying mechanisms mediating the induction of DNA damage and apoptotic cell death by reactive oxygen species (ROS).
Methods
MTT assays were used to measure the proliferation-inhibition ratio of K562 and K562/G01 cells. Flow cytometry (FCM) was used to test the level of extracellular ROS, DNA damage, cell cycle progression and apoptosis. Western blotting was used to verify the amount of protein.
Results
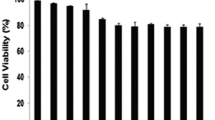
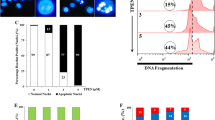
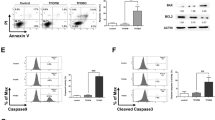
FM-Nov17 significantly inhibited the proliferation of K562 cells, with an IC50 of 58.28 ± 0.304 μM, and K562/G01 cells, with an IC50 of 62.36 ± 0.136 μM. FM-Nov17 significantly stimulated the generation of intracellular ROS, followed by the induction of DNA damage and the activation of the ATM–p53–r-H2AX pathway and checkpoint-related signals Chk1/Chk2, which led to increased numbers of cells in the S and G2/M phases of the cell cycle. Furthermore, FM-Nov17 induced apoptotic cell death by decreasing mitochondrial membrane potential and activating caspase-3 and PARP. The above effects were all prevented by the ROS scavenger N-acetylcysteine.
Conclusions
FM-Nov17-induces DNA damage and mitochondria-dependent cellular apoptosis in CML cells. The process is mediated by the generation of ROS.
Similar content being viewed by others
References
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM, et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001;344:1031–7.
Gorre ME, Mohammed M, Ellwood K, Hsu N, Paquette R, Rao PN, et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 2001;293:876–80.
Von Bubnoff N, Schneller F, Peschel C, Duyster J. BCR-ABL gene mutations in relation to clinical resistance of Philadelphia-chromosome-positive leukaemia to STI571: a prospective study. Lancet 2002;359:487–91.
Perl A, Carroll M. BCR-ABL kinase is dead; long live the CML stem cell. J Clin Invest 2011;121:22–5.
Corbin AS, Agarwal A, Loriaux M, Cortes J, Deininger MW, Druker BJ. Human chronic myeloid leukemia stem cells are insensitive to imatinib despite inhibition of BCR-ABL activity. J Clin Invest 2011;121:396–409.
Wang J, Yi J. Cancer cell killing via ROS: to increase or decrease, that is the question. Cancer Biol Ther 2008;7:1875–84.
Trachootham D, Zhou Y, Zhang H, Demizu Y, Chen Z, Pelicano H, et al. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by beta-phenylethyl isothiocyanate. Cancer Cell 2006;10:241–52.
Wang S, Konorev EA, Kotamraju S, Joseph J, Kalivendi S, Kalyanaraman B. Doxorubicin induces apoptosis in normal and tumor cells via distinctly different mechanisms: intermediacy of H2O2- and p53-dependent pathways. J Bio Chem 2004;279:25535–43.
Brem R, Li F, Montaner B, Reelfs O, Karran P. DNA breakage and cell cycle checkpoint abrogation induced by a therapeutic thiopurine and UVA radiation. Oncogene 2010;29:3953–63.
Mroz RM, Schins RP, Li H, Jimenez LA, Drost EM, Holownia A, et al. Nanoparticle-driven DNA damage mimics irradiation-related carcinogenesis pathways. Eur Respir J 2008;31:241–51.
Ko CH, Shen SC, Hsu CS, Chen YC. Mitochondrial-dependent, reactive oxygen species-independent apoptosis by myricetin: roles of protein kinase C, cytochrome c, and caspase cascade. Biochem Pharmacol 2005;69:913–27.
Lu HF, Hsueh SC, Ho YT, Kao MC, Yang JS, Chiu TH, et al. ROS mediates baicalin-induced apoptosis in human promyelocytic leukemia HL-60 cells through the expression of the Gadd153 and mitochondrial-dependent pathway. Anticancer Res 2007;27:117–25.
Wu LX, Xu JH, Zhang KZ, Lin Q, Huang XW, Wen CX, Chen YZ. Disruption of the Bcr-Abl/Hsp90 protein complex: a possible mechanism to inhibit Bcr-Abl-positive human leukemic blasts by novobiocin. Leukemia 2008;22:1402–9.
Rowley R, Kort L. Novobiocin, nalidixic acid, etoposide, and 4′-(9-acridinylamino) methanesulfon-m-anisidide effects on G2 and mitotic Chinese hamster ovary cell progression. Cancer Res 1989;49:4752–7.
Smith PJ, Bell SM. A DNA topoisomerase II-independent route. Cancer Chemother Pharmacol 1990;26:257–62.
Duan P, You G. Novobiocin is a potent inhibitor for human organic anion transporters. Drug Metab Dispos 2009;37:1203–10.
Huss WJ, Gray DR, Greenberg NM, Mohler JL, Smith GJ. Breast cancer resistance protein-mediated efflux of androgen in putative benign and malignant prostate stem cells. Cancer Res 2005;65:6640–50.
Degtyareva NP, Heyburn L, Sterling J, Resnick MA, Gordenin DA, Doetsch PW. Oxidative stress-induced mutagenesis in single-strand DNA occurs primarily at cytosines and is DNA polymerase zeta-dependent only for adenines and guanines. Nucleic Acids Res 2013;41:8995–9005.
Wu L, Shao L, An N, Wang J, Pazhanisamy S, Feng W, et al. IKKb regulates the repair of DNA double-strand breaks induced by ionizing radiation in MCF-7 breast cancer cells. PLoS ONE 2011;6:e18447.
Kuo LJ, Yang LX. Gamma-H2AX — a novel biomarker for DNA double-strand breaks. In Vivo 2008;22:305–9.
Qi J, Peng H, Gu ZL, Liang ZQ, Yang CZ. Establishment of an imatinib resistant cell line K562/G01 and its characterization. Chin J Hematol 2004;25:337–41.
Ermolaeva MA, Segref A, Dakhovnik A, Ou HL, Schneider JI, Utermöhlen O, et al. DNA damage in germ cells induces an innate immune response that triggers systemic stress resistance. Nature 2013;501:416–20.
Hanot M, Boivin A, Malésys C, Beuve M, Colliaux A, Foray N, et al. Glutathione depletion and carbon ion radiation potentiate clustered DNA lesions, cell death and prevent chromosomal changes in cancer cells progeny. PLoS ONE 2012;7:e44367.
Lange J, Pan J, Cole F, Thelen MP, Jasin M, Keeney S. ATM controls meiotic double-strand-break formation. Nature 2011;479:237–40.
Deavall DG, Martin EA, Horner JM, Roberts R. Drug-induced oxidative stress and toxicity. J Toxicol 2012;12:645–60.
Hepel M, Stobiecka M, Peachey J, Miller J. 1ntervention of glutathione in premutagenic catechol-mediated DNA damage in the presence of copper(II) ions. Mutat Res 2012;735:1–11.
Raj L, Ide T, Gurkar AU, Foley M, Schenone M, Li X, et al. Selective killing of cancer cells by a small molecule targeting the stress response to ROS. Nature 2011;475:231–4.
Diehn M, Cho RW, Lobo NA, Kalisky T, Dorie MJ, Kulp AN, et al. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature 2009;458:780–3.
Hu L, Kim TM, Son MY, Kim SA, Holland CL, Tateishi S, et al. Two replication fork maintenance pathways fuse inverted repeats to rearrange chromosomes. Nature 2013;501:569–72.
Müller R, Misund K, Holien T, Bachke S, Gilljam KM, Våtsveen TK, et al. Targeting proliferating cell nuclear antigen and its protein interactions induces apoptosis in multiple myeloma cells. PLOS ONE 2013;8:e70430.
Floyd SR, Pacold ME, Huang Q, Clarke SM, Lam FC, Cannell IG, et al. The bromodomain protein Brd4 insulates chromatin from DNA damage signalling. Nature 2013;498:246–50.
Martin OA, Ivashkevich A, Choo S, Woodbine L, Jeggo PA, Martin RF, et al. Statistical analysis of kinetics, distribution and co-localisation of DNA repair foci in irradiated cells: Cell cycle effect and implications for prediction of radiosensitivity. DNA Repair 2013;12:844–55.
Tu WZ, Li B, Huang B, Wang Y, Liu XD, Guan H, et al. γH2AX foci formation in the absence of DNA damage: mitotic H2AX phosphorylation is mediated by the DNA-PKcs/CHK2 pathway. FEBS Lett 2013;587:3437–43.
Zhao H, Halicka HD, Li J, Biela E, Berniak K, Dobrucki J, et al. DNA damage signaling, impairment of cell cycle progression, and apoptosis triggered by 5-ethynyl-2′-deoxyuridine incorporated into DNA. Cytometry A 2013;83:979–88.
Bekker-Jensen S, Mailand N. Assembly and function of DNA double-strand break repair foci in mammalian cells. DNA Repair (Amst) 2010;9:1219–28.
Peterson CL, Almouzni G. Nucleosome dynamics as modular systems that integrate DNA damage and repair. Cold Spring Harb Perspect Biol 2013;5(9).
Gospodinov A, Herceg Z. Chromatin structure in double strand break repair. DNA Repair (Amst) 2013;12:800–10.
Heijink AM, Krajewska M, van Vugt MA. The DNA damage response during mitosis. Mutat Res 2013;750:45–55.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, N., Huang, L., Tian, J. et al. A novel synthetic novobiocin analog, FM-Nov17, induces DNA damage in CML cells through generation of reactive oxygen species. Pharmacol. Rep 68, 423–428 (2016). https://doi.org/10.1016/j.pharep.2015.11.002
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.11.002