Abstract
Background
Dexamethasone (Dex), a synthetic glucocorticoid, is strictly controlled for use due to its serious side effects, including immune suppression. Betulinic acid (BA), an antioxidant prepared from the white birch, exhibits immunomodulation properties. To assess the implications and investigate the mechanisms of BA-elicited immunomodulation, we hypothesized that Dex induced thymocyte apoptosis via oxidative stress could be lessened by BA.
Methods
Mice were given oral doses of BA (0.25, 0.5, and 1.0 mg/kg) daily for 14 days, and induced oxidative stress by giving a single dose of Dex intraperitoneal at the dosage of 25 mg/kg body weight 8 h after the last administration of BA.
Results
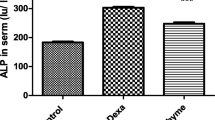
Dex administration alone significantly decreased antioxidant enzyme activities, while significantly increased reactive oxygen species (ROS) production, lipid peroxidation, mitochondrial dysfunctions, caspase-3 activation and cellular apoptosis. However, pretreatment with BA dose-dependently mitigated Dex-induced oxidative damage after 14 days of feeding. In addition to ROS scavenging activity in Dex-induced thymocyte, BA administration decreased lipid peroxidation, up-regulated antioxidant enzymes, restored mitochondrial function, increased Bcl-2 expression but reduced Bax expression, inhibited caspase-3 activation, and improved cell survival.
Conclusions
These findings reveal a protective capability of BA against Dex-induced cell death by reducing oxidative stress via mitochondrial mediated signal pathway which could be the potential mechanism underlying BA-elicited immunomodulation.
Similar content being viewed by others
References
Ortiz R, Cortes L, Gonzales-Marquez H, Comez JL, Gonzales C, Cortes E. Flow cytometric analysis of spontaneous and dexamethasone induced apoptosis in thymocytes from severely malnourished rats. Br J Nutr 2001;86:545–8.
Herold MJ, McPherson KG, Reichardt HM. Glucocorticoids in T cell apoptosis and function. Cell Mol Life Sci 2006;63:60–72.
Inkielewicz-Stepniak I, Radomski MW, Wozniak M. Fisetin prevents fluoride- and dexamethasone-induced oxidative damage in osteoblast and hippocampal cells. Food Chem Toxicol 2012;50:583–9.
You JM, Yun SJ, Nam KN, Kang C, Won R, Lee EH. Mechanism of glucocorticoid-induced oxidative stress in rat hippocampal slice cultures. Can J Physiol Pharm 2009;87:440–7.
Maio R, Perticone M, Sciacqua A, Tassone EJ, Naccarato P, Bagnato C, et al. Oxidative stress impairs endothelial function in nondipper hypertensive patients. Cardiovasc Ther 2010;30:85–92.
Qiao S, Okret S, Jondal M. Thymocyte-synthesized glucocorticoids play a role in thymocyte homeostasis and are down-regulated by adrenocorticotropic hormone. Endocrinology 2009;150:4163–9.
Feng YL, Tang XL. Effect of glucocorticoid-induced oxidative stress on the expression of Cbfa1. Chem Biol Interact 2014;207:26–31.
Kumari A, Kakkar P. Lupeol prevents acetaminophen induced in vivo hepatotoxicity by altering the Bax/Bcl-2 and oxidative stress mediated mitochondrial signaling cascade. Life Sci 2012;90:561–70.
Tzakos AG, Kontogianni VG, Tsoumani M, Kyriakou E, Hwa J, Rodrigues FA, et al. Exploration of the antiplatelet activity profile of betulinic acid on human platelets. J Agric Food Chem 2012;60:6977–83.
Hu TJ, Shuai XH, Chen JR, Wei YY, Zheng RL. Protective effect of a Potentilla anserine polysaccharide on oxidative damages in mice. Int J Biol Macromol 2009;45:279–83.
Chen ZT, Chu HL, Chyau CC, Chu CC, Duh PD. Protective effects of sweet orange (citrus sinensis) peel and their bioactive compounds on oxidative stress. Food Chem 2012;135:2119–27.
Pisha E, Chai H, Lee IS, Chagwedera TE, Farnsworth NR, Cordell GA, et al. Discovery of betulinic acid as a selective inhibitor of human melanoma that functions by induction of apoptosis. Nat Med 1995;1:1046–51.
Yoon JJ, Lee YJ, Kim JS, Kang DG, Lee HS. Protective role of betulinic acid on TNF-alpha-induced cell adhesion molecules in vascular endothelial cells. Biochem Biophys Res Commun 2010;391:96–101.
Szuster-Ciesielska A, Plewka K, Daniluk J, Kandefer-Szerszeń M. Betulin and betulinic acid attenuate ethanol-induced liver stellate cell activation by inhibiting reactive oxygen species (ROS), cytokine (TNF-α, TGF-β) production and by influencing intracellular signaling. Toxicology 2011;280:152–63.
Yi J, Xia W, Wu J, Yuan L, Wu J, Tu D, et al. Betulinic acid prevents alcohol-induced liver damage by improving the antioxidant system in mice. J Vet Sci 2014;15:141–8.
Zheng ZW, Song SZ, Wu YL, Lian LH, Wan Y, Nan JX. Betulinic acid prevention of d-galactosamine/lipopolysaccharide liver toxicity is triggered by activation of Bcl-2 and antioxidant mechanisms. J Pharm Pharmacol 2011;63:572–8.
Yi JE, Obminska-Mrukowicz B, Yuan LY, Yuan H. Immunomodulatory effects of betulinic acid from the bark of white birch on mice. J Vet Sci 2010;11:305–13.
Yi J, Lis M, Szczypka M, Obminska-Mrukowicz B. Influence of betulinic acid on lymphocyte subsets and humoral immune response in mice. Pol J Vet Sci 2012;15:305–13.
Yi J, Zhu R, Wu J, Wu J, Tan Z. Ameliorative effect of betulinic acid on oxidative damage and apoptosis in the splenocytes of dexamethasone treated mice. Int Immunopharmacol 2015;27:85–94.
Zhu L, Yuan H, Guo C, Lu Y, Deng S, Yang Y, et al. Zearalenone induces apoptosis and necrosis in porcine granulosa cells via a caspase-3- and caspase-9-dependent mitochondrial signaling pathway. J Cell Physiol 2012;227:1814–20.
Laszczyk MN. Pentacyclic triterpenes of the lupane, oleanane and ursane group as tools in cancer therapy. Planta Med 2009;75:1549–60.
Malini MM, Lenin M, Varalakshmi P. Protective effect of triterpenes on calcium oxalate crystal-induced peroxidative changes in experimental urolithiasis. Pharmacol Res 2000;41:413–8.
Yoon JJ, Lee YJ, Kim JS, Kang DG, Lee HS. Betulinic acid inhibits high glucose-induced vascular smooth muscle cells proliferation and migration. J Cell Biochem 2010;111:1501–11.
Viji V, Shobha B, Kavitha SK, Ratheesh M, Kripa K, Helen A. Betulinic acid isolated from Bacopa monniera (L.) Wettst suppresses lipopolysaccharide stimulated interleukin-6 production through modulation of nuclear factor-kappaB in peripheral blood mononuclear cells. Int Immunopharmacol 2010;10:843–9.
Almeida M, Han L, Ambrogini E, Weinstein RS, Manolagas SC. Glucocorticoids and tumor necrosis factor an increase oxidative stress and suppress Wnt protein signaling in osteoblasts. J Biol Chem 2011;286:44326–35.
Fu JY, Qian LB, Zhu LG, Liang HT, Tan YN, Lu HT, et al. Betulinic acid ameliorates endothelium-dependent relaxation in L-NAME-induced hypertensive rats by reducing oxidative stress. Eur J Pharm Sci 2011;44:385–91.
Circu ML, Aw TY. Reactive oxygen species, cellularredoxsystemand apoptosis. Free Radic Biol Med 2010;48:749–62.
Pathak N, Khandelwal S. Influence of cadmium on murine thymocytes: potentiation of apoptosis and oxidative stress. Toxicol Lett 2006;165:121–32.
Suwanjang W, Abramov AY, Govitrapong P, Chetsawang B. Melatonin attenuates dexamethasone toxicity-induced oxidative stress, calpain and caspase activation in human neuroblastoma SH-SY5Y cells. JSteroid Biochem Mol Biol 2013;138:116–22.
Filipović D, Zlatković J, Inta D, Bjelobaba I, Stojiljkovic M, Gass P. Chronic isolation stress predisposes frontal cortex but not the hippocampus to the potentially detrimental release of cytochrome c from mitochondria and the activation of caspase-3. J Neurosci Res 2011;89:1461–70.
Psarra AM, Solakidi S, Sekeris CE. The mitochondrion as a primary site of action of steroid and thyroid hormones: presence and action of steroid and thyroid hormone receptors in mitochondria of animal cells. Mol Cell Endocrinol 2006;246:21–33.
Tome ME, Lee K, Jaramillo MC, Briehl MM. Mitochondria are the primary source of the H2O2 signal for glucocorticoid-induced apoptosis of lymphoma cells. Exp Ther Med 2012;4:237–42.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yi, J., Zhu, R., Wu, J. et al. In vivo protective effect of betulinic acid on dexamethasone induced thymocyte apoptosis by reducing oxidative stress. Pharmacol. Rep 68, 95–100 (2016). https://doi.org/10.1016/j.pharep.2015.07.003
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.07.003