Abstract
Background
The pathogenic mechanisms of skin fibrosis are still not completely understood, unlike the profibrotic role played by inflammatory cytokines and transforming growth factor-β1 (TGF-β1). Few antifibrotic drugs are available. Nevertheless, folk medicine suggests numerous treatments of fibrotic conditions. Based on information from folk medicine and literature, the hypothesis was made that proto-berberine alkaloids could act as antifibrotic and cytoprotective agents.
Methods
The effects of berberine, dihydroberberine, canadine, stylopine, and coptisine were investigated on an in vitro model of fibrosis purposely set up. The study is based on the use of human dermal fibroblasts (HDF). The ability of the proto-berberine alkaloids investigated to modulate mitochondrial dehydrogenase activity, cell proliferation, collagen production, and inflammatory cytokine (IL-1β and IL-6) production was tested on HDF cells grown under standard growth conditions, in the presence of 100 μM H2O2, simulating oxidative stress conditions, and in the presence of 34 ng/ml TGF-β1, simulating fibrotic conditions. Antiradical activity was assayed as well, as it could contribute to cytoprotection.
Results
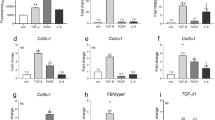
Each alkaloid tested showed peculiar effects on HDF. In particular, all of the alkaloids tested, with the exception of coptisine, inhibited TGF-β1-induced collagen production.
Conclusions
Due to its irritant effects and the lack of desired properties, coptisine has low exploitation potentialities. The other proto-berberine alkaloids investigated resulted all endowed with activities for which they can be exploited as antifibrotic and cytoprotective agents. Stylopine globally proved to be the most promising compound, being endowed with revitalizing, anti-inflammatory, antifibrotic and wound-healing promoting activities, and showing no toxic effects.
Similar content being viewed by others
References
Satish L, Kathju S. Cellular and molecular characteristics of scarless versus fibrotic wound healing. Derm Res Pract 2010. 11 pp. Article ID 790234.
Engrav LH, Garner WL, Tredget EE. Hypertrophic scar, wound contraction and hyper-hypopigmentation. J Burn Care Res 2007;28:593–7.
Zhu Z, Ding J, Shankowsky HA, Tredget EE. The molecular mechanism of hypertrophic scar. J Cell Commun Signal 2013. https://doi.org/10.1007/s12079-013-0195-5 [Epub ahead of print].
Atiyeh BS, Costagliola M, Hayek SN. Keloid or hypertrophic scar: the controversy: review of the literature. Ann Plast Surg 2005;54:676–80.
Hinchcliff M, Varga J. Systemic sclerosis/scleroderma. A treatable multisystem disease. Am Fam Physician 2008;78:961–8.
Thomay AA, Daley JM, Sabo E, Worth PJ, Shelton LJ, Harty MW, et al. Disruption of interleukin-1 signaling improves the quality of wound healing. Am J Pathol 2009;174:2129–36.
Penn JW, Grobbelaar AO, Rolfe KJ. The role of the TGF-β family in wound healing, burns and scarring: a review. Int J Burn Trauma 2012;2:18–28.
Xu Q, Norman JT, Shrivastav S, Lucio-Cazana J, Kopp JB. In vitro models of TGF-β-induced fibrosis suitable for high-throughput screening of antifibrotic agents. Am J Physiol Renal Physiol 2007;293:F631–40.
Chandran S, Sashwati R. Redox signals in wound healing. Biochim Biophys Acta 2008;1780:1348–61.
Kanta J. The role of hydrogen peroxide and other reactive oxygen species in wound healing. Acta Medica (Hradec Kralove) 2011;54:97–101.
Martinez-Hernandez A. Repair, regeneration and fibrosis. In: Rubin E, Farber JL, editors. Pathology. Philadelphia: Lippincott; 1999.
Sun X, Zhang X, Hu H, Lu Y, Chen J, Yasuda K, et al. Berberine inhibits hepatic stellate cell proliferation and prevents experimental liver fibrosis. Biol Pharm Bull 2009;32:1533–7.
Domitrović R, Jakovac H, Vasiljev Marchesi V, Blažeković B. Resolution of Liver fibrosis by isoquinoline alkaloid berberine in CCl4-intoxicated mice is mediated by suppression of oxidative stress and upregulation of MMP-2 expression. J Med Food 2013;16:518–28.
Li J, Pan Y, Kan M, Xiao X, Wang Y, Guan F, et al. Hepatoprotective effects of berberine on liver fibrosis via activation of AMP-activated protein kinase. Life Sci 2014;98:24–30.
Zhang YJ, Yang SH, Li MH, Iqbal J, Bourantas CV, Mi QY, et al. Berberine attenuates adverse left ventricular remodeling and cardiac dysfunction after acute myocardial infarction in rats: role of autophagy. Clin Exp Pharmacol Physiol 2014;12:995–1002.
Li M-H, Zhang Y-J, Yu Y-H, Yang S-H, Iqbal J, Mi Q-Y, et al. Berberine improves pressure overload-induced cardiac hypertrophy and dysfunction through enhanced autophagy. Eur J Pharmacol 2014;728:67–76.
Huang K, Liu W, Lan T, Xie X, Peng J, Huang J, et al. Berberine reduces fibronectin expression by suppressing the S1P-S1P2 receptor pathway in experimental diabetic nephropathy models. PLoS ONE 2012;7:e43874.
Wan X, Chen X, Liu L, Zhao Y, Huang W-J, Zhang Q, et al. Berberine ameliorates chronic kidney injury caused by atherosclerotic renovascular disease through the suppression of NFκB signaling pathway in rats. PLoS ONE 2013;8:e59794.
Chitra P, Saiprasad G, Manikandan R, Sudhandiran G. Berberine attenuates bleomycin induced pulmonary toxicity and fibrosis via suppressing NF-κB dependant TGF-β activation: a biphasic experimental study. Toxicol Lett 2013;219:178–93.
Rasband WS. ImageJ. Bethesda, MD: U.S. National Institutes of Health; 1997– 2012, https://doi.org/imagej.nih.gov/ij/.
Lee C-H, Chen J-C, Hsiang C-Y, Wu S-L, Wu H-C, Ho T-Y. Berberine suppresses inflammatory agents-induced interleukin-1β and tumor necrosis factor-α productions via the inhibition of IκB degradation in human lung cells. Pharm Res 2007;56:193–201.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pietra, D., Borghini, A. & Bianucci, A.M. In vitro studies of antifibrotic and cytoprotective effects elicited by proto-berberine alkaloids in human dermal fibroblasts. Pharmacol. Rep 67, 1081–1089 (2015). https://doi.org/10.1016/j.pharep.2015.04.001
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.04.001