Abstract
The Italian hare is endemic to central and southern Italy and currently designated as vulnerable by the International Union for the Conservation of Nature. Despite the worrying status, few studies have explored its ecology. In this scenario, improving knowledge of food preferences is of primary importance. Here, we present a non-invasive pilot study set to analyse the diet composition of the Italian hare from southern Italy, starting from fecal pellets, and using, for the first time on this species, DNA metabarcoding and next generation sequencing.
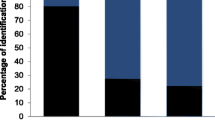
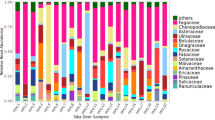
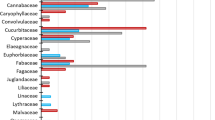
The findings indicated that this approach provides reliable qualitative and semi-quantitative information, allowing the characterization of the hare diet and its seasonal variation using 22 fecal samples. In a single experiment, through time and cost-effective screening of multiple DNA metabarcodes, we detected a broad diversity of plants (99 taxa). Unlike traditional methods, this approach can identify items that leave no solid remains or that simply are lacking in diagnostic taxonomic features. Moreover, it starts from small amounts of input fecal material, useful in investigation on elusive and vulnerable species. The major guideline for future applications is to use a barcode short enough to allow amplification of environmental degraded DNA and the selection of the universal primers for PCR amplification.
The results should be useful to define management actions for conservation of endemic Italian hare.
Similar content being viewed by others
References
Aceto, S., Caputo, S., Cozzolino, S., Gaudio, L., Moretti, A., 1999. Phylogeny and evolution of orchis and allied genera based on ITS DNA variation: morphological gaps and molecu-lar continuity. Mol. Phylogen. Evol. 13, 67–76.
Alvarez, I., Wendel, J.F., 2003. Ribosomal ITS sequences and plant phylogenetic inference. Mol. Phylogen. Evol. 29, 417–434.
Ando, H., Setsuko, S., Horikoshi, K., Suzuki, H., Umehara, S., Inoue-Murayama, M., et al., 2013. Diet analysis by next-generation sequencing indicates the frequent consumption of in-troduced plants by the critically endangered red-headed wood pigeon (Columba janthina nitens) in oceanic island habitats. Ecol. Evol. 3, 4057–4069.
Angelici, F.M., Luiselli, L., 2001. Distribution and status of the Apennine hare Lepus cor-sicanus in continental Italy and Sicily. Oryx 35, 245–249.
Angelici, F., Randi, E., Riga, F., Trocchi, V., 2008. Lepus corsicanus. The IUCN Red List of Threatened Species. Version 2015.1. http://www.iucnredlist.org.
Baamrane, M.A.A., Shehzad, W., Ouhammou, A., Abbad, A., Naimi, M., Coissac, E., et al., 2012. Assessment of the food habits of the Moroccan Dorcas gazelle in M’Sabih Talaa, West Central Morocco, using the trnL approach. PLoS One 4, e35643.
Bellemain, E., Carlsen, T., Brochmann, C., Coissac, E., Taberlet, P., Kauserud, H., 2010. ITS as an environmental DNA barcode for fungi: an in silico approach reveals potential PCR bi-ases. BMC Microbiol. 10, 189.
Bolger, A.M., Lohse, M., Usadel, B., 2014. Trimmomatic: a flexible trimmer for illumina sequence data. Bioinformatics, btu170.
Bradley, B., Stiller, M., Doran-Sheehy, D., Harris, T., Chapman, C., Vigilant, L., 2007. Plant DNA sequences from feces: potential means for assessing diets of wild primates. Am. J. Primatol. 69, 699–705.
Camacho, C., Coulouris, G., Avagyan, V., Ma, N., Papadopoulos, J., Bealer, K., et al., 2009. BLAST+: architecture and applications. BMC Bioinf. 10, 421.
Coissac, E., 2012. OligoTag: a program for designing sets of tags for next-generation se-quencing of multiplexed samples. In: Pompanon, F., Bonin, A. (Eds.), Data Production and Analysis in Population Genomics. Humana Press, New York, NY, USA, pp. 13–31.
Cristobal-Azkarate, J., Arroyo-Rodriguez, V., 2007. Diet and activity pattern of howler mon-keys (Alouatta palliata) in Los Tuxtlas, Mexico: effects of habitat fragmentation and implications for conservation. Am. J. Primatol. 69, 1013.
De Barba, M., Miquel, C., Boyer, F., Mercier, C., Rioux, D., Coissac, E., et al., 2014. DNA metabarcoding multiplexing and validation of data accuracy for diet assessment: application to omnivorous diet. Mol. Ecol. Resour. 14, 306–323.
De Battisti, R., Migliore, S., Masutti, L., Trocchi, V., 2004. The diet of the Italian hare Lepus corsicanus on Etna Mountain, Sicily. In: Paper Presented at the Second World Lagomorph Conference, Vaira¨o (Portugal), 26–31 July.
Deagle, B.E., Tollit, D.J., 2007. Quantitative analysis of prey DNA in pinniped faeces: potential to estimate diet composition? Conserv. Genet. 8, 743–747.
Deagle, B., Eveson, J., Jarman, S., 2006. Quantification of damage in DNA recovered from highly degraded samples–a case study on DNA in faeces. Front. Zool. 3, 11.
Deagle, B.E., Kirkwood, R., Jarman, S.N., 2009. Analysis of Australian fur seal diet by py-rosequencing prey DNA in faeces. Mol. Ecol. 18, 2022–2038.
Deagle, B.E., Chiaradia, A., McInnes, J., Jarman, S.N., 2010. Pyrosequencing faecal DNA to determine diet of little penguins: is what goes in what comes out? Conserv. Genet. 11, 2039–2048.
Dixon, P., 2003. Vegan, a package of R functions for community ecology. J. Veg. Sci. 14, 927–930.
Doyle, J.J., Doyle, J.L., 1987. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 19, 11–15.
Drummond, A., Ashton, B., Buxton, S., Cheung, M., Cooper, A., Duran, C., et al., 2011. Geneious v 5.5. http://www.geneious.com.
Duffy, J.E., Cardinale, B.J., France, K.E., McIntyre, P.B., Thèbault, E., Loreau, M., 2007. The func-tional role of biodiversity in ecosystems: incorporating trophic complexity. Ecol. Lett. 10, 522–538.
Ficetola, G.F., Coissac, E., Zundel, S., Riaz, T., Shehzad, W., Bessiere, J., et al., 2010. An in silico approach for the evaluation of DNA barcodes. BMC Genom. 11, 434.
Frediani, M., Caputo, P., 2005. Phylogenetic relationships among annual and perenni-al species of the genus Cicer as inferred from ITS sequences of nuclear ribosomal DNA. Biol. Plant. 49, 45–52.
Freitas, T.A.K., Li, P.E., Scholz, M.B., Chain, P.S., 2015. Accurate read-based metagenome characterization using a hierarchical suite of unique signatures. Nucleic Acids Res. 43, e69.
Freschi, P., Fascetti, S., Musto, M., Mallia, E., Blasi, A., Cosentino, C., et al., 2014. Diet of the Apennine hare in a southern Italy Regional Park. Eur. J. Wildl. Res. 60, 423–430.
Freschi, P., Fascetti, S., Musto, M., Cosentino, C., Paolino, R., Valentini, V., 2015a. Seasonal variation in food habits of the Italian hare in a south Apennine semi-natural landscape. Ethol. Ecol. Evol., 1–15 (ahead-of-print).
Freschi, P., Fascetti, S., Musto, M., Mallia, E., Cosentino, C., Paolino, R., 2015b. Diet of the Italian hare (Lepus corsicanus) in a semi-natural landscape of southern Italy. Mammalia 79, 51–59.
Fulgione, D., Maselli, V., Pavarese, G., Rippa, D., Rastogi, R.K., 2009. Landscape fragmenta-tion and habitat suitability in endangered Italian hare (Lepus corsicanus) and European hare (Lepus europaeus) populations. Eur. J. Wildl. Res. 55, 385–396.
Glenn, T.C., 2011. Field guide to next-generation DNA sequencers. Mol. Ecol. Resour. 11, 759–769.
Harris, J.K., Sahl, J.W., Castoe, T.A., Wagner, B.D., Pollock, D.D., Spear, J.R., 2010. Comparison of normalization methods for construction of large, multiplex amplicon pools for next-generation sequencing. Appl. Environ. Microbiol. 76, 3863–3868.
Holechek, J.L., Vavra, M., Pieper, R.D., 1982. Botanical composition determination of range herbivore diets: a review. J. Range Manag. 35, 309–315.
Homolka, M., 1982. The food of Lepus europaeus in a meadow and woodland complex. Folia Zool. 31, 243–253.
Hulbert, I.A.R., Iason, G.R., Mayes, R.W., 2001. The fexibility of an intermediate feeder: dietary selection by mountain hares measured using faecal n-alkanes. Oecologia 129, 197–205.
Ingerson-Mahar, J.M., 2014. Relating diet and morphology of the head, mandibles and proventriculus in adult carabid beetles. In: Holland, J.M. (Ed.), The Agroecolgy of Carabid Beetles. Intercept, Andover.
Karimi, I., Hayatgheybi, H., Shamspur, T., Kamalak, A., Pooyanmehr, M., Marandi, Y., 2011. Chemical composition and effect of an essential oil of Salix aegyptiaca (musk willow) in hypercholesterolemic rabbit model. Braz. J. Pharmacol. 21, 407–414.
Kowalczyk, R., Taberlet, P., Coissac, E., Valentini, A., Miquel, C., Kamińsk, T., et al., 2011. Influence of management practices on large herbivore diet case of European bison in Bialowiez a primeval forest (Poland). For. Ecol. Manag. 261, 821–828.
Kress, W.J., Wurdack, K.J., Zimmer, E.A., Weigt, L.A., Janzen, D.H., 2005. Use of DNA barcodes to identify flowering plants. Proc. Natl. Acad. Sci. U. S. A. 102, 8369–8374.
Lassmann, T., Hayashizaki, Y., Daub, C.O., 2011. SAMStat: monitoring biases in next gener-ation sequencing data. Bioinformatics 27, 130–131.
Li, H., Durbin, R., 2009. Fast and accurate short read alignment with Burrows-Wheeler Transform. Bioinformatics 25, 1754–1760.
Li, H., Handsaker, B., Wysoker, A., Fennell, T., Ruan, J., Homer, N., et al., 2009. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079.
Lopes, C.M., De Barba, M., Boyer, F., Mercier, C., da Silva Filho, P.J.S., Heidtmann, L.M., et al., 2015. DNA metabarcoding diet analysis for species with parapatric vs sympatric distribu-tion: a case study on subterranean rodents. Heredity 114, 525–536.
Luo, R., Liu, B., Xie, Y., Li, Z., Huang, W., Yuan, J., et al., 2012. SOAPdenovo2: an empirically improved memory-efficient short-read de novo assembler. GigaScience 1, 18.
Mahdi, J.G., Mahdi, A.J., Bowen, I.D., 2006. The historical analysis of aspirin discovery, its relation to the willow tree and antiproliferative and anticancer potential. Cell Prolif. 39, 147–155.
Mollot, G., Duyck, P.F., Lefeuvre, P., Lescourret, F., Martin, J.-F., Piry, S., et al., 2014. Cover cropping alters the diet of arthropods in a banana plantation: a metabarcoding approach. PLoS One 9, e93740.
Oksanen, J., Blanchet, F.G., Kindt, R., Legendre, P., Minchin, P.R., O’Hara, R.B., et al., 2011. Vegan: Community Ecology Package. R Package Version 2.0-0. http://CRAN.R-pro-ject.org/package=vegan.
Pierpaoli, M., Riga, F., Trocchi, V., Randi, E., 1999. Species distinction and evolutionary rela-tionships of the Italian hare (Lepus corsicanus) as described by mitochondrial DNA se-quencing. Mol. Ecol. 8, 1805–1817.
Pierpaoli, M., Riga, F., Trocchi, V., Randi, E., 2003. Hare populations in Europe: intra and in-terspecific analysis of mtDNA variation. C R Biol. 326, 80–84.
Piggott, M.P., 2005. Effect of sample age and season of collection on the reliability of microsatellite genotyping of faecal DNA. Wildl. Res. 31, 485–493.
Pompanon, F., Deagle, B.E., Symondson, W.O.C., Brown, D.S., Jarman, S.N., Taberlet, P., 2012. Who is eating what: diet assessment using next generation sequencing. Mol. Ecol. 21, 1931–1950.
Prokopowich, C.D., Gregory, T.R., Crease, T.J., 2003. The correlation between rDNA copy number and genome size in eukaryotes. Genome 46, 48–50.
R Development Core Team, 2010. R: A Language and Environment for Statistical Computing, 3-900051-07-0R. Foundation for Statistical Computing, Vienna, Austria http://www.R-project.org.
Rai, A., 2013. The antiinflammatory and antiarthritic properties of ethanol extract of Hedera helix. Indian J. Pharm. Sci. 75, 99–102.
Rippa, D., Maselli, V., Soppelsa, O., Fulgione, D., 2011. The impact of agro-pastoral abandonment on the Rock Partridge Alectoris graeca in the Apennines. Ibis 153, 721–734.
Rodríguez, J.P., Beard, T.D., Bennett, E.M., Cumming, G.S., Cork, S.J., Agard, J., et al., 2006. Trade-offs across space, time, and ecosystem services. Ecol. Soc. 11, 28.
Santini, A., Lucchini, V., Fabbri, E., Randi, E., 2007. Ageing and environmental factors affect PCR success in wolf (Canis lupus) excremental DNA samples. Mol. Ecol. Notes 7, 955–961.
Schoch, L.C., Seifert, K.A., Huhndorfc, S., Robertd, V., Spouge, J.L., Levesqueb, C.A., et al., 2012. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for fungi. PNAS 16, 6241–6246.
Shendure, J., Ji, H., 2008. Next genetation DNA sequencing. Nat. Biotechnol. 26, 1135–1145.
Soininen, E.M., Valentini, A., Coissac, E., Miquel, C., Gielly, L., Brochmann, C., et al., 2009. Analysing diet of small herbivores: the efficiency of DNA barcoding coupled with high-throughput pyrosequencing for deciphering the composition of complex plant mixtures. Front. Zool. 6, 16.
Soininen, E.M., Gauthier, G., Bilodeau, F., Berteaux, D., Gielly, L., Taberlet, P., et al., 2015. Highly overlapping winter diet in two sympatric lemming species revealed by DNA metabarcoding. PLoS One 10, e0115335.
Sokal, R.R., Rohlf, F.J., 1995. Biometry: the principles and practice of statistics. In: Biological Research, Series. Freeman W. H. and Company, New York.
Soulé, M.E., Estes, J.A., Berger, J., Del Rio, C.M., 2003. Ecological effectiveness: conservation goals for interactive species. Conserv. Biol. 17, 1238–1250.
Strumia, S., Catalanotti, A.E., Santangelo, A., de Filippo, G., 2007. Caratterizzazione floristico-vegetazionale dei siti alimentari di Lepus corsicanus nel Parco Nazionale del Cilento e Vallo di Diano. In: de Filippo, G., De Riso, L., Riga, F. (Eds.), Conservazione di Lepus corsicanus De Winton, 1898 e stato delle conoscenze. IGF Publishing, pp. 149–157.
Taberlet, P., Coissac, E., Hajibabaei, M., Rieseberg, L.H., 2012. Environmental DNA. Mol. Ecol. 21, 1789–1793.
Trocchi, V., Riga, F., 2005. I Lagomorfi in Italia. Linee guida per la conservazione e la gestione. Min. Politiche Agricole e Forestali. Ist. Naz. Fauna Selvatica, Documenti tecnici, 25, 1–28.
Troedsson, C., Simonelli, P., Nagele, V., Nejstgaard, J.C., Frischer, M.E., 2009. Quantification of copepod gut content by differential length amplification quantitative PCR (dla-qPCR). Mar. Biol. 156, 253–259.
Valentini, A., Pompanon, F., Taberlet, P., 2009a. DNA barcoding for ecologists. Trends Ecol. Evol. 24, 110–117.
Valentini, A., Miquel, C., Nawaz, M., Bellemain, E., Coissac, E., Pompanon, F., et al., 2009b. New perspectives in diet analysis based on DNA barcoding and parallel pyrosequencing: the trnL approach. Mol. Ecol. Resour. 9, 51–60.
Vynne, C., Baker, M., Breuer, Z., Wasser, S., 2012. Factors influencing degradation of DNA and hormones in maned wolf scat. Anim. Conserv. 15, 184–194.
Wang, X.C., Liu, C., Huang, L., Bengtsson-Palme, J., Chen, H., Zhang, J.H., et al., 2015. ITS1: a DNA barcode better than ITS2 in eukaryotes? Mol. Ecol. Resour. 15, 573–586.
Wasser, S., Houston, C., Koehler, G.M., Cadd, G., Fain, S., 1997. Techniques for application of faecal DNA methods to field studies of Ursids. Mol. Ecol. 6, 1091–1097.
Yokota, Y., Kawata, T., Iida, Y., Kato, A., Tanifuji, S., 1989. Nucleotide sequences of the 5.8S rRNA gene and internal transcribed spacer regions in carrot and broad bean ribosomal DNA. J. Mol. Evol. 29, 294–301.
Yu, H.G., Huang, J.A., Yang, Y.N., Huang, H., Luo, H.S., Yu, J.P., et al., 2002. The effects of ace-tylsalicylic acid on proliferation, apoptosis, and invasion ofcyclooxygenase-2 negative co-lon cancer cells. Eur. J. Clin. Invest. 32, 838–846.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buglione, M., Maselli, V., Rippa, D. et al. A pilot study on the application of DNA metabarcoding for non-invasive diet analysis in the Italian hare. Mamm Biol 88, 31–42 (2018). https://doi.org/10.1016/j.mambio.2017.10.010
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.mambio.2017.10.010