Abstract
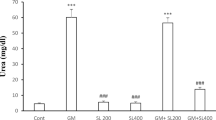
Gentamicin induced renal complications are well known in humans and animals. Medicinal properties of Withania somnifera (L.) Dunal, Solanaceae, are recognized to improve renal functions. However, the pharmacological function of W. somnifera is not completely understood. We sought to unravel medicinal therapeutic function of W. somnifera on gentamicin-induced nephrotoxicity in wistar rats. Twenty-four adult male wistar rats evenly divided into four groups to evaluate in vivo nephroprotective and nephrocurative function of W. somnifera in gentamicin induced nephrotoxic rats. Experimental design as follows: Group I, saline control for 21 days; Group II, gentamicin nephrotoxic control for eight days; Group III, alcoholic extract of W. somnifera for 13 days + simultaneous administration of gentamicin and W. somnifera, from day 14 to 21 (nephroprotective) and Group IV, gentamicin for 8 days + alcoholic extract of W. somnifera from day 9 to 21 (nephrocurative). End of experiment, respective serum and kidney tissue samples used to analyze renal function. Withania somnifera as a nephroprotective and nephrocurative molecule significantly restore the renal function on gentamicin-induced nephrotoxicity. This phenomenon is accompanied with significantly reduced blood urea nitrogen, creatine, alkaline phosphatase, gamma-glutamyl transferase, albumin, total protein, calcium, potassium and kidney malon-dialdehyde concentrations. Additionally, W. somnifera significantly increased antioxidant activities of glutathione and superoxide dismutase to protect renal tissue damage from gentamicin in wistar rats. Over all, W. somnifera treated nephroprotective animal shows improved recovery compared to nephrocuartive. The nephroprotective or nephrocurative effect of W. somnifera could be due to inherent antioxidant and free-radical-scavenging principle(s). In the near future, biologically active compounds of W. somnifera (withanolides) could appear as a novel therapeutic molecule for renal disorders.
Article PDF
Similar content being viewed by others
References
Abuelezz, SA, Hendawy, N., Abdel Gawad, S., 2016. Alleviation of renal mitochondrial dysfunction and apoptosis underlies the protective effect of sitagliptin in gentamicin-inducednephrotoxicity. J. Pharm. Pharmacol. 68, 523–532.
Ahn, J., You, S.J., Lee, Y.M., Oh, S.W., Ahn, S., Kim, S., Chin, HJ., Chae, D.W., Na, K.Y., 2012. Hypoxia-inducible factor activation protects the kidney from gentamicin-induced acute injury. PLoS One 7, e48952.
Ali, B.H., Abdel Gayoum, A.A., Bashir, A.A., 1992. Gentamicin nephrotoxicity in rat: some biochemical correlates. Pharmacol. Toxicol. 70, 419–423.
Beutler, E., Duron, O., Kelly, B.M., 1963. Improved method forthe determination of bloodglutathione. J. Lab. Clin. Med. 61, 882–888.
Bhattacharya, S.K., Satyan, K.S., Ghosal, S., 1997. Antioxidant activity of glycowith-anolides from Withania somnifera. Indian J. Exp. Biol. 35, 236–239.
Cardiff, R.D., Miller, C.H., Munn, R.J., 2014. Manual hematoxylin and eosin staining of mouse tissue sections. Cold Spring Harb. Protoc. 2014, 655–658.
Davis, L., Kuttan, G., 2000. Effect of Withania somnifera on cyclophosphamide-induced urotoxicity. Cancer Lett. 148, 9–17.
Dhuley, J.N., 2000. Adaptogenic and cardioprotective action of ashwagandhain rats andfrogs. J. Ethnopharmacol. 70, 57–63.
Hernández-López, F., Rodríguez-Landa, J.F., Puga-Olguín, A., Germán-Ponciano, L.J., Rivadeneyra-Domínguez, E., Bernal-Morales, B., 2017. Analysis of activity and motor coordination in rats undergoing stereotactic surgery and implantation of a cannula into the dorsal hippocampus. Neurologia 32, 579–586.
Jayaprakasam, B., Zhang, Y., Seeram, N.P., Nair, M.G., 2003. Growth inhibition of human tumor cell lines by withanolides from Withania somnifera leaves. Life Sci. 74, 125–132.
Jeyanthi, T., Subramanian, P., 2009. Nephroprotective effect of Withania somnifera: a dose-dependent study. Ren. Fail. 31, 814–821.
Jiang, M., Karasawa, T., Steyger, P.S., 2017. Aminoglycoside-inducedcochleotoxicity: a review. Front Cell Neurosci. 11, 308.
Karadeniz, A., Yildirim, A., Simsek, N., Kalkan, Y., Celebi, F., 2008. Spirulina platensis protects against gentamicin-induced nephrotoxicity in rats. Phytother. Res. 22, 1506–1510.
Karahan, I., Ates¸s¸ahin, A., Yilmaz, S., Ceribas¸i, A.O., Sakin, F., 2005. Protective effect of lycopene on gentamicin-induced oxidative stress and nephrotoxicity in rats. Toxicology 215, 198–204.
Khan, M.R., Badar, I., Siddiquah, A., 2011. Prevention of hepatorenal toxicity with Sonchus asper in gentamicin treated rats. BMC Complement Altern. Med. 11.
Krause, K.M., Serio, A.W., Kane, T.R., Connolly, L.E., 2016. Aminoglycosides: an overview. Cold Spring Harb. Perspect. Med. 6.
Lee, S.I., Lee, J.H., Park, S.Y., Park, J.W., 2013. Do bupivacaine, clindamycin, and gentamicin at their clinical concentrations enhance rocuronium-induced neuromuscular block? Korean J. Anesthesiol. 64, 346–352.
Lopez-Giacoman, S., Madero, M., 2015. Biomarkers in chronic kidney disease, from kidney function to kidney damage. World J. Nephrol. 4, 57–73.
Madesh, M., Balasubramanian, K.A., 1998. Microtiter plate assay for superoxide dis- mutase using MTT reduction by superoxide. Indian J. Biochem. Biophys. 35, 184–188.
McMahon, G.M., Waikar, S.S., 2013. Biomarkers in nephrology: core curriculum 2013. Am. J. Kidney Dis. 62, 165–178.
Mingeot-Leclercq, M.P., Tulkens, P.M., 1999. Aminoglycosides: nephrotoxicity. Antimicrob. Agents Chemother. 43, 1003–1012.
Niki, E., Omata, Y., Fukuhara, A., Saito, Y., Yoshida, Y., 2008. Assessment of radical scavenging capacity and lipid peroxidation inhibiting capacity ofantioxidant. J. Agric. Food Chem. 56, 8255–8260.
Perazella, M.A., 2018. Pharmacology behind common drug nephrotoxicities. Clin. J. Am. Soc. Nephrol. 13, 1897–1908.
Raghavan, V., Weisz, O.A., 2016. Discerning the role of mechanosensors in regulating proximal tubule function. Am. J. Physiol. Renal Physiol. 310, F1–F5.
Rai, D., Bhatia, G., Sen, T., Palit, G., 2003. Anti-stress effects of Ginkgo biloba and Panax ginseng: a comparativestudy. J. Pharmacol. Sci. 93, 458–464.
Randjelovic, P., Veljkovic, S., Stojiljkovic, N., Sokolovic, D., Ilic, I., 2017. Gentamicin nephrotoxicity in animals: current knowledge and future perspectives. EXCLI J. 16, 388–399.
Rhee, W.J., Lee, S.Y., Lee, J.H., Choi, S.R., Lee, S.-C., Lee, J.H., Lee, S.I., 2015. The effect of high concentration of magnesium with ropivacaine, gentamicin, rocuronium, and their combination on neuromuscular blockade. Korean J. Anesthesiol. 68, 50–61.
Sayed-Ahmed, M.M., Nagi, M.N., 2007. Thymoquinone supplementation prevents the development of gentamicin-induced acute renal toxicity in rats. Clin. Exp. Pharmacol. Physiol. 34, 399–405.
Shafiq-Ur-Rehman, 1984. null Lead-induced regional lipid peroxidation in brain. Toxicol. Lett. 21, 333–337.
Sodimbaku, V., Pujari, L., Mullangi, R., Marri, S., 2016. Carrot (Daucus carota L.): nephroprotective against gentamicin-induced nephrotoxicity in rats. Indian J. Pharmacol. 48, 122–127.
Tavafi, M., Ahmadvand, H., Toolabi, P., 2012. Inhibitory effect of olive leaf extract on gentamicin-induced nephrotoxicity in rats. Iran J. Kidney Dis. 6, 25–32.
Teo, S.H., Endre, Z.H., 2017. Biomarkers in acute kidney injury (AKI). Best Pract. Res. Clin. Anaesthesiol. 31, 331–344.
Wiland, P., Szechcinski, J., 2003. Proximal tubule damage in patients treated with gentamicin or amikacin. Pol. J. Pharmacol. 55, 631–637.
Yaman, I., Balikci, E., 2010. Protective effects of Nigella sativa against gentamicin-induced nephrotoxicity in rats. Exp. Toxicol. Pathol. 62, 183–190.
Yanagida, C., Ito, K., Komiya, I., Horie, T., 2004. Protective effect of fosfomycin on gentamicin-induced lipid peroxidation of rat renal tissue. Chem. Biol. Interact. 148, 139–147.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author’s contribution
All authors contributed significantly to the work presented in this manuscript. YPS and VG supervised the whole experiments. PKG and VG conceptualized the project and performed the experiments. PKG, VG, SMT and RHL organized, analyzed and interpreted the results. PKG and SMT wrote the manuscript draft. RHL and SMT read the manuscript and provided critical assessment and conceptual insights.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Govindappa, P.K., Gautam, V., Tripathi, S.M. et al. Effect of Withania somnifera on gentamicin induced renal lesions in rats. Rev. Bras. Farmacogn. 29, 234–240 (2019). https://doi.org/10.1016/j.bjp.2018.12.005
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.12.005