Abstract
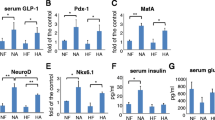
Recently, lupin seed (Lupinus albus L., Fabaceae) products have emerged as a functional food due to their nutritional and health benefits. Numerous reports have demonstrated the hypoglycemic effects of lupin’s gamma conglutin protein; nonetheless, its mechanism of action remains elusive. To understand the role of this protein on glucose metabolism, we evaluated the effect of administering L. albus’ gamma conglutin on Slc2a2, Gck, and Pdx-1 gene expression as well as GLUT2 protein tissue levels in streptozotocininduced diabetic rats. While consuming their regular diet, animals received a daily gamma conglutin dose (120 mg/kg per body weight) for seven consecutive days. Serum glucose levels were measured at the beginning and at the end of the experimental period. At the end of the trial, we quantified gene expression in pancreatic and hepatic tissues as well as GLUT2 immunopositivity in Langerhans islets. Gamma conglutin administration lowered serum glucose concentration by 17.7%, slightly increased Slc2a2 and Pdx-1 mRNA levels in pancreas, up-regulated Slc2a2 expression in the liver, but it had no effect on hepatic Gck expression. After gamma conglutin administration, GLUT2 immunopositivity in Langerhans islets of diabetic animals resembled that of healthy rats. In conclusion, our results indicate that gamma conglutin up-regulates Slc2a2 gene expression in liver and normalizes GLUT2 protein content in pancreas of streptozotocin-induced rats.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Agius, L., 2014. Lessons from glucokinase activators: the problem of declining efficacy. Expert. Opin. Ther. Pat. 24, 1155–1159.
Ahlgren, U., Jonsson, J., Jonsson, L., Simu, K., Edlund, H., 1998. Beta-cell-specific inactivation of the mouse Ipf1/Pdx1 gene results in loss of the beta-cell phenotype and maturity onset diabetes. Genes Dev. 12, 1763–1768.
Alvim, R.O., Cheuhen, M.R., Machado, S.R., Sousa, A.G.P., Santos, P.C., 2015. General aspects of muscle glucose uptake. An. Acad. Bras. Cienc. 87, 351–368.
American Diabetes Association, 2017. Classification and diagnosis of diabetes. Sec 2. In: Standards of medical care in diabetes. Diabetes Care 40 (Suppl. 1), S11–S24.
Arden, C., Petrie, J.L., Tudhope, S.J., Al-Oanzi, Z., Claydon, A.J., Beynon, R.J., Towle, H.C., Agius, L., 2011. Elevated glucose represses liver glucokinase and induces its regulatory protein to safeguard hepatic phosphate homeostasis. Diabetes 60, 3110–3120.
Arnoldi, A., Boschin, G., Zanoni, C., Lammi, C., 2015. The health benefits of sweet lupin seed flours and isolated proteins. J. Funct. Foods 18, 550–563.
Bae, J.S., Kim, T.H., Kim, M.Y., Park, J.M., Ahn, Y.H., 2010. Transcriptional regulation of glucose sensors in pancreatic beta-cells and liver: an update. Sensors (Basel) 10, 5031–5053.
Brandsch, C., Kappis, D., Weisse, K., Stangl, G.I., 2010. Effects of untreated and thermally treated lupin protein on plasma and liver lipids of rats fed a hypercholesterolemic high fat or high carbohydrate diet. Plant Foods Hum. Nutr. 65, 410–416.
Capraro, J., Magni, C., Faoro, F., Maffi, D., Scarafoni, A., Tedeschi, G., Maffioli, E., Parolari, A., Manzoni, C., Lovati, M.R., Duranti, M., 2013. Internalisation and multiple phosphorylation of gamma-Conglutin, the lupin seed glycaemia-lowering protein, in HepG2 cells. Biochem. Biophys. Res. Commun. 437, 648–652.
Chen, L., Alam, T., Johnson, J.H., Hughes, S., Newgard, C.B., Unger, R.H., 1990. Regulation of beta-cell glucose transporter gene expression. Proc. Natl. Acad. Sci. U. S. A. 87, 4088–4092.
Duranti, M., Consonni, A., Magni, C., Sessa, F., Scarafoni, A., 2008. The major proteins of lupin seed: characterisation and molecular properties for use as functional and nutraceutical ingredients. Trends Food. Sci. Technol. 19, 624–633.
Fleet, T., Zhang, B., Lin, F., Zhu, B., Dasgupta, S., Stashi, S., Tackett, B., Thevananther, S., Rajapakshe, K.I., Gonzales, N., Dean, A., Mao, J., Timchenko, N., Malovannaya, A., Qin, J., Coarfa, C., DeMayo, F., Dacso, C.C., Foulds, C.E., O’Malley, B.W., York, B., 2015. SRC-2 orchestrates polygenic inputs for fine-tuning glucose homeostasis. Proc. Natl. Acad. Sci. U. S. A. 112, E6068–E6077.
Fujimoto, K., Polonsky, K.S., 2009. Pdx1 and other factors that regulate pancreatic beta-cell survival. Diabetes Obes. Metab. 11 (Suppl. 4), 30–37.
Gannon, N.P., Conn, C.A., Vaughan, R.A., 2015. Dietary stimulators of GLUT4 expression and translocation in skeletal muscle: a mini review. Mol. Nutr. Food Res. 59, 48–64.
Ganzera, M., Kruger, A., Wink, M., 2010. Determination of quinolizidine alkaloids in different Lupinus species by NACE using UV and MS detection. J. Pharm. Biomed. Anal. 53, 1231–1235.
Gao, T., McKenna, B., Li, C., Reichert, M., Nguyen, J., Singh, T., Yang, C., Pannikar, A., Doliba, N., Zhang, T., Stoffers, D.A., Edlund, H., Matschinsky, F., Stein, R., Stanger, B.Z., 2014. Pdx1 maintains β-cell identity and function by repressing an α-cell program. Cell. Metab. 19, 259–271.
Gonzalez-Santiago, A.E., Vargas-Guerrero, B., Garcia-Lopez, P.M., Martinez-Ayala, A.L., Dominguez-Rosales, J.A., Gurrola-Diaz, C.M., 2017. Lupinus albus conglutin gamma modifies the gene expressions of enzymes involved in glucose hepatic production in vivo. Plant Foods Hum. Nutr. 72, 134–140.
Gremlich, S., Roduit, R., Thorens, B., 1997. Dexamethasone induces posttranslational degradation of GLUT2 and inhibition of insulin secretion in isolated pancreatic beta cells. Comparison with the effects of fatty acids. J. Biol. Chem. 272, 3216–3222.
Guillam, M.T., Hummler, E., Schaerer, E., Yeh, J.I., Birnbaum, M.J., Beermann, F., Schmidt, A., Dériaz, N., Thorens, B., 1997. Early diabetes and abnormal postnatal pancreatic islet development in mice lacking Glut-2. Nat. Genet. 17, 327–330.
International Diabetes Federation, 2017. IDF Diabetes Atlas, 8th ed. International Diabetes Federation, Brussels, Belgium.
Iynedjian, P.B., Gjinovci, A., Renold, A.E., 1988. Stimulation by insulin of glucokinase gene transcription in liver of diabetic rats. J. Biol. Chem. 263, 740–744.
Lampart-Szczapa, E., Siger, A., Trojanowska, K., Nogala-Kalucka, M., Malecka, M., Pacholek, B., 2003. Chemical composition and antibacterial activities of lupin seeds extracts. Nahrung 47, 286–290.
Lin, H.V., Accili, D., 2011. Hormonal regulation of hepatic glucose production in health and disease. Cell Metab. 14, 9–19.
Lovati, M.R., Manzoni, C., Castiglioni, S., Parolari, A., Magni, C., Duranti, M., 2012. Lupin seed gamma-conglutin lowers blood glucose in hyperglycaemic rats and increases glucose consumption of HepG2 cells. Br. J. Nutr. 107, 67–73.
Lucas, M.M., Stoddard, F.L., Annicchiarico, P., Frías, J., Martinez-Villaluenga, C., Sussmann, D., Duranti, M., Seger, A., Zander, P.M., Pueyo, J.J., 2015. The future of lupin as a protein crop in Europe. Front Plant Sci. 8, https://doi.org/10.3389/fpls.2015.00705.
Magni, C., Sessa, F., Accardo, E., Vanoni, M., Morazzoni, P., Scarafoni, A., Duranti, M., 2004. Conglutin gamma, a lupin seed protein, binds insulin in vitro and reduces plasma glucose levels of hyperglycemic rats. J. Nutr. Biochem. 15, 646–650.
McKinnon, C.M., Docherty, K., 2001. Pancreatic duodenal homeobox-1, PDX-1, a major regulator of beta cell identity and function. Diabetologia 44, 1203–1214.
Ohlsson, H., Karlsson, K., Edlund, T., 1993. IPF1, a homeodomain-containing trans-activator of the insulin gene. EMBOJ. 12, 4251–4259.
Ozougwu, J.C., Obimba, K.C., Belonwu, C.D., Unakalamba, C.B., 2013. The pathogenesis and pathophysiology of type 1 and type 2 diabetes mellitus. J. Physiol. Pathophysiol. 4, 46–57.
Pedica, F., Beccari, S., Pedron, S., Montagna, L., Piccoli, P., Doglioni, C., Chilosi, M., 2014. PDX-1 (pancreatic/duodenal homeobox-1 protein 1). Pathologica 106, 315–321.
Radtke, J., Schutkowski, A., Brandsch, C., Hirche, F., Hasenkopf, K., Stangl, G.I., 2015. Isolated conglutin gamma from Lupin, but not phytate, lowers serum cholesterol without influencing vascular lesion development in the ApoE-deficient mouse model. Plant Foods Hum. Nutr. 70, 113–118.
Rao, P.V., Pugazhenthi, S., Khandelwal, R.L., 1995. The effects of streptozotocininduced diabetes and insulin supplementation on expression of the glycogen phosphorylase gene in rat liver. J. Biol. Chem. 270, 24955–24960.
Rorsman, P., Renstrom, E., 2003. Insulin granule dynamics in pancreatic beta cells. Diabetologia 46, 1029–1045.
Schagger, H., von Jagow, G., 1987. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 166, 368–379.
Sharabi, K., Tavares, C.D., Rines, A.K., Puigserver, P., 2015. Molecular pathophysiology of hepatic glucose production. Mol. Aspects Med. 46, 21–33.
Terruzzi, I., Senesi, P., Magni, C., Montesano, A., Scarafoni, A., Luzi, L., Duranti, M., 2011. Insulin-mimetic action of conglutin-gamma, a lupin seed protein, in mouse myoblasts. Nutr. Metab. Cardiovasc. Dis. 21, 197–205.
Thorens, B., Wu, Y.J., Leahy, J.L., Weir, G.C., 1992. The loss of GLUT2 expression by glucose-unresponsive beta cells of db/db mice is reversible and is induced by the diabetic environment. J. Clin. Invest. 90, 77–85.
Unger, R.H., 1991. Diabetic hyperglycemia: link to impaired glucose transport in pancreatic beta cells. Science 251, 1200–1205.
Ugochukwu, N.H., Babady, N.E., 2003. Antihyperglycemic effect of aqueous and ethanolic extracts of Gongronema latifolium leaves on glucose and glycogen metabolism in livers of normal and streptozotocin-induced diabetic rats. Life Sci. 73, 1925–1938.
Vargas-Guerrero, B., Garcia-Lopez, P.M., Martinez-Ayala, A.L., Dominguez-Rosales, J.A., Gurrola-Diaz, C.M., 2014. Administration of Lupinus albus gamma conglutin (Cgamma) to n5 STZ rats augmented Ins-1 gene expression and pancreatic insulin content. Plant Foods Hum. Nutr. 69, 241–247.
Waeber, G., Thompson, N., Nicod, P., Bonny, C., 1996. Transcriptional activation of the GLUT2 gene by the IPF-1/STF-1/IDX-1 homeobox factor. Mol. Endocrinol. 10, 1327–1334.
Watada, H., Kajimoto, Y., Umayahara, Y., Matsuoka, T., Kaneto, H., Fujitani, Y., Kamada, T., Kawamori, R., Yamasaki, Y., 1996. The humanglucokinase gene beta-cell-type promoter: an essential role of insulin promoter factor 1/PDX-1 in its activation in HIT-T15 cells. Diabetes 45, 1478–1488.
Weir, G.C., Bonner-Weir, S., 2004. Five stages of evolving beta-cell dysfunction during progression to diabetes. Diabetes 53 (Suppl. 3), S16–S21.
Yonamine, C.Y., Pinheiro-Machado, E., Michalani, M.L., Freitas, H.S., Okamoto, M.M., Corrêa-Giannella, M.L., Machado, U.F., 2016. Resveratrol improves glycemic control in insulin-treated diabetic rats: participation of the hepatic territory. Nutr. Metab. (Lond.) 13, https://doi.org/10.1186/s12986-016-0103-0.
Zhao, L., Li, Z., Kullin, M., Borg, L.A., Karlsson, F.A., 2005. Alterations in net glucose uptake and in the pancreatic B-cell GLUT2 transporter induced by diazoxide and by secretory stimuli. J. Endocrinol. 185, 291–299.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contribution
All authors contributed substantially to the present work. Rde-JSM, BVG, and TJG performed the experimental work and the statistical analysis. PMGL and ALMA contributed in the protein isolation process and critical analysis of the paper. JADR carried out the histological evaluation of the tissues. CMGD contributed in the experimental conception, designing, data interpretation and critical analysis of the paper. CMGD and TJG drafted the manuscript. All authors read and approved the final version of the manuscriptbefore submission.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Sandoval-Muñíz, R.J., Vargas-Guerrero, B., Guzmán, T.J. et al. Lupin gamma conglutin protein: effect on Slc2a2, Gck and Pdx-1 gene expression and GLUT2 levels in diabetic rats. Rev. Bras. Farmacogn. 28, 716–723 (2018). https://doi.org/10.1016/j.bjp.2018.08.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.08.002