Abstract
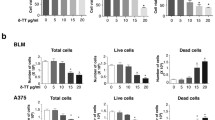
Zeaxanthin, an abundant carotenoid present in fruits, vegetables and algae was reported to exert antiproliferative activity and induce apoptosis in human uveal melanoma cells. It also inhibited uveal melanoma tumor growth and cell migration in nude mice xenograft models. Here we report that zeaxanthin purified from the rhodophyte Porphyridium purpureum (Bory) K.M.Drew & R.Ross, Porphyridiaceae, promotes apoptosis in the A2058 human melanoma cell line expressing the oncogenic BRAF V600E mutation. Zeaxanthin 40 μM (IC50) induced chromatin condensation, nuclear blebbing, hypodiploidy, accumulation of cells in sub-G1 phase, DNA internucleosomal fragmentation and activation of caspase-3. Western blot analysis revealed that zeaxanthin induced up-regulation of the pro-apoptotic factors Bim and Bid and inhibition of NF-κB transactivation. Additionally, zeaxanthin sensitized A2058 melanoma cells in vitro to the cytotoxic activity of vemurafenib, a BRAF inhibitor widely used for the clinical management of melanoma, suggesting its potential interest as dietary adjuvant increasing melanoma cells sensitivity to chemotherapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmad, I., Muneer, K.M., Tamimi, I.A., Chang, M.E., Ata, M.O., Yusuf, N., 2013. Thymoquinone suppresses metastasis of melanoma cells by inhibition of NLRP3 inflammasome. Toxicol. Appl. Pharmacol. 270, 70–76.
Alqathama, A., Prieto, J.M., 2015. Natural products with therapeutic potential in melanoma metastasis. Nat. Prod. Rep. 32, 1170–1182.
Anforth, R., Carlos, G., Clements, A., Kefford, R., Fernandez-Peñas, P., 2015. Cutaneous adverse events in patients treated with BRAF inhibitor-based therapies formetastatic melanomaforlongerthan52 weeks. Br. J. Dermatol. 172, 239–243.
Armstrong, B.K., Kricker, A., 2001. The epidemiology of UV induced skin cancer. J. Photochem. Photobiol. B63, 8–18.
Baudelet, P.H., Gagez, A.L., Bérard, J.B., Juin, C., Bridiau, N., Kaas, R., Thiéry, V., Cadoret, J.P., Picot, L., 2013. Antiproliferative activity of Cyanophora paradoxa pigments in melanoma, breast and lung cancer cells. Mar. Drugs 11, 4390–4406.
Bi, M.C., Rosen, R., Zha, R.Y., McCormick, S.A., Song, E., Hu, D.N., 2013. Zeaxanthin induces apoptosis in human uveal melanoma cells through Bcl-2 family proteins and intrinsic apoptosis pathway. Evid. Based. Complement. Alternat. Med., https://doi.org/10.1155/2013/205082.
Burri, B.J., Neidlinger, T.R., Clifford, A.J., 2001. Serum carotenoid depletion follows first-order kinetics in healthy adult women fed naturally low carotenoid diets. J. Nutr. 131, 2096–2100.
Caltagirone, S., Rossi, C., Poggi, A., Ranelletti, F.O., Natali, P.G., Brunetti, M., Aiello, F.B., Piantelli, M., 2000. Flavonoids apigenin and quercetin inhibit melanomagrowth and metastatic potential. Int. J. Cancer 87, 595–600.
Chen, Y.T., Kao, C.J., Huang, H.Y., Huang, S.Y., Chen, C.Y., Lin, Y.S., Wen, Z.H., Wang, H.M.D., 2017. Astaxanthin reduces MMP expressions, suppresses cancer cell migrations, and triggers apoptotic caspases of in vitro and in vivo models in melanoma. J. Funct. Foods 31, 20–31.
Chew, B.P., Park, J.S., 2004. Carotenoid action on the immune response. J. Nutr. 134, 257S–261S.
Chu, E.Y., Wanat, K.A., Miller, C.J., Amaravadi, R.K., Fecher, LA, Brose, M.S., McGet-tigan, S., Giles, L.R., Schuchter, L.M., Seykora, J.T., Rosenbach, M., 2012. Diverse cutaneous side effects associated with BRAF inhibitor therapy: a clinicopathologic study. J. Am. Acad. Dermatol. 67, 1265–1272.
Chung, T.W., Choi, HJ., Lee, J.Y., Jeong, H.S., Kim, CH. Joo, M., Choi, J.Y., Han, C.W., Kim, S.Y., Choi, J.S., Ha, K.T., 2013. Marine algal fucoxanthin inhibits the metastatic potential of cancer cells. Biochem. Biophys. Res. Commun. 439, 580–585.
Cragg, G.M., Newman, D.J., Snader, K.M., 1997. Natural products in drug discovery and development. J. Nat. Prod. 60, 52–60.
Dankort, D., Curley, D.P., Cartlidge, RA, Nelson, B., Karnezis, A.N., Damsky, W.E., You, M.J., De Pinho, RA., McMahon, M., Bosenberg, M., 2009. Braf(V600E) cooperates with Pten loss to induce metastatic melanoma. Nat. Genet. 41, 544–552.
Davies, H., Bignell, G.R., Cox, C., Stephens, P., Edkins, S., Clegg, S., Teague, J., Woffendin, H., Garnett, M.J., Bottomley, W., Davis, N., Dicks, E., Ewing, R., Floyd, Y., Gray, K., Hall, S., Hawes, R., Hughes, J., Kosmidou, V., Menzies, A., Mould, C., Parker, A., Stevens, C., Watt, S., Hooper, S., Wilson, R., Jayatilake, H., Gusterson, BA, Cooper, C., Shipley, J., Hargrave, D., Pritchard-Jones, K., Maitland, N., Chenevix-Trench, G., Riggins, G.J., Bigner, D.D., Palmieri, G., Cossu, A., Flanagan, A., Nicholson, A., Ho, J.W.C., Leung, S.Y., Yuen, S.T., Weber, B.L., Seigler, H.F., Darrow, T.L., Paterson, H., Marais, R., Marshall, C.J., Wooster, R., Stratton, M.R., Futreal, P.A., 2002. Mutations of the BRAF gene in human cancer. Nature 417, 949–954.
Dutton-Regester, K., Irwin, D., Hunt, P., Aoude, L.G., Tembe, V., Pupo, G.M., Lanagan, C., Carter, C.D., O’Connor, L., O’Rourke, M., Scolyer, R.A., Mann, G.J., Schmidt, C.W., Herington, A., Hayward, N.K., 2012. A high-throughput panel for identifying clinically relevant mutation profiles in melanoma. Mol. Cancer Ther. 11, 888–897.
Firdous, A.P., Sindhu, E.R., Ramnath, V., Kuttan, R., 2010. Anti-mutagenic and anti-carcinogenic potential of the carotenoid meso-zeaxanthin. Asian Pac. J. Cancer Prev. 11, 1795–1800.
Gagez, A.L., Thiery, V., Pasquet, V., Cadoret, J.P., Picot, L., 2012. Epoxycarotenoidsand cancer. Curr. Bioact. Compd. 8, 109–141.
Ghodratizadeh, S., Kanbak, G., Beyramzadeh, M., Dikmen, Z.G., Memarzadeh, S., Habibian, R., 2014. Effect of carotenoid ß-cryptoxanthinoncellularand humoral immune response in rabbit. Vet. Res. Commun. 38, 59–62.
Grange, F., 2005. Epidemiology of cutaneous melanoma: descriptive data in France and Europe. Ann. Dermatol. Venereol. 132, 975–982.
Hall, W.A., Djalilian, H.R., Nussbaum, E.S., Cho, K.H., 2000. Long-term survival with metastatic cancer to the brain. Med. Oncol. 17, 279–286.
Haluska, F.G., Tsao, H., Wu, H., Haluska, F.S., Lazar, A., Goel, V., 2006. Genetic alterations in signaling pathways in melanoma. Clin. Cancer Res. 12, 2301s–2307s.
Hashimoto, T., Ozaki, Y., Mizuno, M., Yoshida, M., Nishitani, Y., Azuma, T., Komoto, A., Maoka, T., Tanino, Y., Kanazawa, K., 2011. Pharmacokinetics of fucoxanthinol in human plasma afterthe oral administration of kombu extract. Br.J. Nutr. 107, 1–4.
Hedidi, M., Erb, W., Bentabed-Ababsa, G., Chevallier, F., Picot, L., Thiéry, V., Bach, S., Ruchaud, S., Roisnel, T., Dorcet, V., Mongin, F., 2016. Synthesis of N-pyridyl azoles using a deprotometalation-iodolysis-N-arylation sequence and evaluation of their antiproliferative activity in melanoma cells. Tetrahedron 72, 6467–6476.
Jang, S., Atkins, M.B., 2014. Treatment of BRAF-mutant melanoma: the role ofvemu-rafenib and other therapies. Clin. Pharmacol. Ther. 95, 24–31.
Juin, C., Bonnet, A., Nicolau, E., Bérard, J.B., Devillers, R., Thiéry, V., Cadoret, J.P., Picot, L., 2015. UPLC-MSE profiling of phytoplankton metabolites: application to the identification of pigments and structural analysis of metabolites in Porphyridium purpureum. Mar. Drugs 13, 2541–2558.
Kim, K.N., Ahn, G., Heo, S.J., Kang, S.M., Kang, M.C., Yang, H.M., Kim, D., Roh, S.W., Kim, S.K., Jeon, B.T., Park, P.J., Jung, W.K., Jeon, Y.J., 2013. Inhibition of tumor growth in vitro and in vivo by fucoxanthin against melanoma B16F10 cells. Environ. Toxicol. Pharmacol. 35, 39–46.
Kumar, S.R., Hosokawa, M., Miyashita, K., 2013. Fucoxanthin: a marine carotenoid exerting anti-cancer effects by affecting multiple mechanisms. Mar. Drugs 11, 5130–5147.
Leiter, U., Garbe, C., 2008. Epidemiology of melanoma and nonmelanoma skin cancer — the role of sunlight. In: Sunlight, Vitam. DSki. Cancer. Springer, New York, NY, pp. 89–103.
Locatelli, C., Filippin-Monteiro, F.B., Creczynski-Pasa, T.B., 2013. Recent advances in the biology, therapy and management of melanoma. InTech.
Lu, M., Zhang, Y., Zhao, C., Zhou, P., Yu, L., 2015. Analysis and identification of astaxanthin and its carotenoid precursors from Xanthophyllomyces dendrorhous by high-performance liquid chromatography. Z. Naturforsch. C. 65, 489–494.
MacKie, R.M., Hauschild, A., Eggermont, A.M.M., 2009. Epidemiology of invasive cutaneous melanoma. Ann. Oncol. 20 (Suppl. 6 vi), 1–7.
Marneros, A.G., 2009. Tumor angiogenesis in melanoma. Hematol. Oncol. Clin. North Am. 23, 431–446.
Mathieu, V., de Lassalle, E.M., Toelen, J., Mohr, T., Bellahcène, A., Van Goietsenoven, G., Verschuere, T., Bouzin, C., Debyser, Z., De Vleeschouwer, S., Van Gool, S., Poirier, F., Castronovo, V., Kiss, R., Feron, O., 2012. Galectin-1 in melanoma biology and related neo-angiogenesis processes. J. Invest. Dermatol. 132, 2245–2254.
Mesquita, M.L., Paula, J.E., Pessoa, C., Moraes, M.O., Costa-Lotufo, L.V., Grougnet, R., Michel, S., Tillequin, F., Espindola, L.S., 2009. Cytotoxic activity of Brazilian Cerrado plants used in traditional medicine against cancer cell lines. J. Ethnopharmacol. 123, 439–445.
Mimouni, V., Ulmann, L., Pasquet, V., Mathieu, M., Picot, L., Bougaran, G., Cadoret, J.P., Morant-Manceau, A., Schoefs, B., 2012. The potential of microalgae for the production of bioactive molecules of pharmaceutical interest. Curr. Pharm. Biotechnol. 13, 2733–2750.
Mirzaei, H., Naseri, G., Rezaee, R., Mohammadi, M., Banikazemi, Z., Mirzaei, H.R., Salehi, H., Peyvandi, M., Pawelek, J.M., Sahebkar, A., 2016. Curcumin: a new candidate for melanoma therapy? Int. J. Cancer 139, 1683–1695.
Niles, R.M., McFarland, M., Weimer, M.B., Redkar, A., Fu, Y.M., Meadows, G.G., 2003. Resveratrol is a potent inducer of apoptosis in human melanoma cells. Cancer Lett. 190, 157–163.
Oliveira-Junior, R.G., Thiéry, V., Sergent, O., Chevanne, M., Picot, L., 2016. Could fucoxanthin interaction with lipid rafts mediate its cytotoxicity in cancer cells? J. Oceanogr. Mar. Res. 4, 144.
Pasquet, V., Morisset, P., Ihammouine, S., Chepied, A., Aumailley, L., Berard, J.B., Serive, B., Kaas, R., Lanneluc, I., Thiery, V., Lafferriere, M., Piot, J.M., Patrice, T., Cadoret, J.P., Picot, L., 2011. Antiproliferative activity of violaxanthin isolated from bioguided fractionation of Dunaliella tertiolecta extracts. Mar. Drugs 9, 819–831.
Pechinskii, S.V., Kuregyan, A.G., 2014. The impact of carotenoids on immunity (Review). Pharm. Chem. J. 47, 509–513.
Putey, A., Joucla, L., Picot, L., Besson, T., Joseph, B., 2007. Synthesis of latonduine derivatives via intramolecular Heck reaction. Tetrahedron 63, 867–879.
Reboul, E., 2013. Absorption of vitamin A and carotenoids by the enterocyte: focus on transport proteins. Nutrients 5, 3563–3581.
Roy, S., Llewellyn, C., Egeland, E., Johnsen, G. (Eds.), 2011. Phytoplankton Pigments: Characterization, Chemotaxonomy and Applications in Oceanography (Cambridge Environmental Chemistry Series). Cambridge University Press, Cambridge, pp. 728–822, https://doi.org/10.1017/CBO9780511732263.032.
Serive, B., Kaas, R., Bérard, J.B., Pasquet, V., Picot, L., Cadoret, J.P., 2012. Selection and optimisation of a method for efficient metabolites extraction from microalgae. Bioresour. Technol. 124, 311–320.
Serive, B., Nicolau, E., Bérard, J.B., Kaas, R., Pasquet, V., Picot, L., Cadoret, J.P., 2017. Community analysis of pigment patterns from 37 microalgae strains reveals new carotenoids and porphyrins characteristic of distinct strains and taxonomic groups. PLOS ONE 12, e0171872.
Spagnolo, F., Ghiorzo, P., Queirolo, P., 2014. Overcoming resistance to BRAF inhibition in BRAF-mutated metastatic melanoma. Oncotarget 5, 10206–10221.
Sugawara, T., Matsubara, K., Akagi, R., Mori, M., Hirata, T., 2006. Antiangiogenic activity of brown algae fucoxanthin and its deacetylated product, fucoxanthinol. J. Agric. Food Chem. 54, 9805–9810.
Tanaka, T., Shnimizu, M., Moriwaki, H., 2012. Cancer chemoprevention by carotenoids. Molecules 17, 3202–3242.
Thurnham, D.I., Howard, A.N., 2013. Studies on meso-zeaxanthin for potential toxicity and mutagenicity. Food Chem. Toxicol. 59, 455–463.
Van Goietsenoven, G., Hutton, J., Becker, J.P., Lallemand, B., Robert, F., Lefranc, F., Pirker, C., Vandenbussche, G., Van Antwerpen, P., Evidente, A., Berger, W., Prévost, M., Pelletier, J., Kiss, R., Kinzy, T.G., Kornienko, A., Mathieu, V., 2010. Targeting of eEF1A with Amaryllidaceae isocarbostyrils as a strategy to combat melanomas. FASEB J. 24, 4575–4584.
Van Heukelem, L., Thomas, C., 2001. Computer-assisted high-performance liquid chromatography method development with applications to the isolation and analysis of phytoplankton pigments. J. Chromatogr. A 910, 31–49.
Walne, P., 1966. Experiments in the large-scale culture of the larvae of Ostrea edulis (L). In: Fish. Investig. Ser. II. London Her Majesty’s Station. Off., pp. iii. 53.
Welsh, S.J., Corrie, P.G., 2015. Management of BRAF and MEK inhibitor toxicities in patients with metastatic melanoma. Ther. Adv. Med. Oncol. 7, 122–136.
Xu, X., Zhang, L., Shao, B., Sun, X., Ho, CT., Li, S., 2013. Safety evaluation of mesozeaxanthin. Food Control 32, 678–686.
Xu, X.L., Hu, D.N., Iacob, C., Jordan, A., Gandhi, S., Gierhart, D.L., Rosen, R., 2015. Effects of zeaxanthin on growth and invasion of human uveal melanoma in nude mouse model. J. Ophthalmol., https://doi.org/10.1155/2015/392305.
Zapata, M., Rodriguez, F., Garrido, J., 2000. Separation of chlorophylls and carotenoids from marine phytoplankton: a new HPLC method using a reversed phase C8 column and pyridine-containing mobile phases. Mar. Ecol. Prog. Ser. 195, 29–45.
Zhang, L., Wei, Y., Zhang, J., 2014. Novel mechanisms of anticancer activities of green tea component epigallocatechin- 3-gallate. Anticancer Agents Med. Chem. 14, 779–786.
Zimmer, L., Livingstone, E.U., Hillen, S., Domkes, A., Becker, D., Schadendorf, D., 2012. Panniculitis with arthralgia in patients with melanoma treated with selective BRAF inhibitors and its management. Arch. Dermatol. 148, 357–361.
Author information
Authors and Affiliations
Corresponding author
Additional information
JBB and EN produced P. purpureum biomass. CJ, RGOJ, AF, CO, LPy,IL, GP and LP performed the pigment extraction, purification, cell culture, western-blot and apoptosis experiments. CJ performed the HRMS analysis. LB and CJ performed the flux cytometry analysis. LP designed the experiments, interpreted the data, directed the study and wrote the manuscript in collaboration with VT and JRGDSA. LP takes responsibility for the integrity of the work, from inception to finished article.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Juin, C., de Oliveira Junior, R.G., Fleury, A. et al. Zeaxanthin from Porphyridium purpureum induces apoptosis in human melanoma cells expressing the oncogenic BRAF V600E mutation and sensitizes them to the BRAF inhibitor vemurafenib. Rev. Bras. Farmacogn. 28, 457–467 (2018). https://doi.org/10.1016/j.bjp.2018.05.009
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.05.009