Abstract
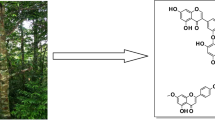
Aphelandra scabra (Vahl) Sm., Acanthaceae, is a shrub widely used by some Mayan communities as carminative, antidote, and remedy for some infections. Bio-guided isolation of the methanol extract of leaves led us to the purification of the anti-giardial metabolites cirsimaritin and sorbifolin, along with the inactive metabolites cirsimarin, sorbifolin-6-O-β-glucopyranoside, and squalene. Cirsimaritin displayed high activity in the anti-giardial bioassay with an IC50= 3.8 μM, being considered as outstanding when compared to previous reported metabolites, while sorbifolin showed a low activity with an IC50= 75.6 μM. Additionally, both compounds proved not to be cytotoxic in an in vitro bioassay against HEK-293, a normal cell line. This is the first investigation on anti-giardial properties of A. scabra and its phytochemistry as well, thus the isolated compounds are considered as new for the plant genus and for the species.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amaral, F.M.M., Ribeiro, M.N.S., Barbosa-Filho, J.M., Reis, A.S., Nascimento, F.R.F., Macedo, R.O., 2006. Plants and chemical constituents with giardicidal activity. Rev. Bras. Farmacogn. 16, 696–720.
Bai, N., He, K., Roller, M., Lai, C., Shao, X., Pan, M., Bily, A., Ho, C., 2011. Flavonoid glycosides from Microtea debilis and their cytotoxic and anti-inflammatory effects. Fitoterapia 82, 168–172.
Ben Sghaier, M., Skandrani, I., Nasr, N., Dijoux-Franca, M., Chekir-Ghedira, L., Ghedira, K., 2011. Flavonoids and sesquiterpenes from Teucrium ramosissimum promote antiproliferation of human cancer cells and enhance antioxidant activity: a structure–activity relationship study. Environ. Toxicol. Pharmacol. 32, 336–348.
Bussati, H.G.N.O., Santos, J.F.G., Gomes, M.A., 2009. The old and new therapeutic approaches to the treatment of giardiasis: where are we? Biol. Targets Ther. 3, 273–278.
Calzada, F., 2005. Additional antiprotozoal constituents from Cuphea pinetorum, a plant used in Mayan traditional medicine to treat diarrhoea. Phytother. Res. 19, 725–727.
Calzada, F., Cervantes-Martínez, J.A., Yépez-Mulia, L., 2005. In vitro antiprotozoal activity from the roots of Geranium mexicanum and its constituents on Entamoeba histolytica and Giardia lamblia. J. Ethnopharmacol. 98, 191–193.
Calzada, F., Alanís, A.D., 2007. Additional antiprotozoal flavonol glycosides of the aerial parts of Helianthemum glomeratum. Phytother. Res. 21, 78–80.
Cedillo-Rivera, R., Enciso-Moreno, J., Martínez-Palomo, A., Ortega-Pierres, G., 1991. Isolation and axenization of Giardia lamblia isolates from symptomatic and asymptomatic patients in Mexico. Arch. Med. Res. 22, 79–85.
Cedillo-Rivera, R., Muñoz, O., 1992. In-vitro susceptibility of Giardia lamblia to albendazole, mebendazole and other chemotherapeutic agents. J. Med. Microbiol. 37, 221–224.
Cedillo-Rivera, R., Darby, J.M., Enciso-Moreno, J.A., Ortega-Pierres, G., Ey, P.L., 2003. Genetic homogeneity of axenic isolates of Giardia intestinalis derived from acute and chronically infected individuals in Mexico. Parasitol. Res. 90, 119–123.
El-Hawiet, A.M., Toaima, S.M., Assad, A.M., Radwan, M.M., El-Sebakhy, N.A., 2010. Chemical constituents from Astragalus annularis Forssk. and A. trimestris L., Fabaceae. Rev. Bras. Farmacogn. 20, 860–865.
Eshbakova, K.A., Sagitdinova, G.V., Malikov, V.M., Melibaev, S., 1996. Flavone sorbifolin from Pulicaria uliginosa. Chem. Nat. Compd. 32, 82.
Fernandes, D.C., Regasini, L.O., Vellosa, J.C.R., Pauletti, P.M., Castro-Gamboa, I., Bolzani, V.S., Oliveira, O.M.M., Silva, D.H.S., 2008. Myeloperoxidase inhibitory and radical scavenging activities of flavones from Pterogyne nitens. Chem. Pharm. Bull. 56, 723–726.
Flora digital: Península de Yucatán, 2010. Aphelandra scabra. Herbario CICY, Unidad de Recursos Naturales, pp. 2011, http://www.cicy.mx (accessed December).
Geissman, T.A., Mukherjee, R., Sim, Y., 1967. Constituents of Helichrysum viscosum var. bracteatum DC. Phytochemistry 6, 1575–1581.
Jeong, D.M., Jung, H.A., Choi, J.S., 2008. Comparative antioxidant activity and HPLC profiles of some selected Korean thistles. Arch. Pharm. Res. 31, 28–33.
Kelm, M.A., Nair, M.G., Strasburg, G.M., DeWitt, D.L., 2000. Antioxidant and cyclooxygenase inhibitory phenolic compounds from Ocimum sanctum Linn. Phytomedicine 7, 7–13.
Lane, S., Lloyd, D., 2002. Current trends in research into the waterborne parasite Giardia. Crit. Rev. Microbiol. 28, 123–147.
Lentz, D.L., 1993. Medicinal and other economic plants of the Paya of Honduras. Econ. Bot. 47, 358–370.
Lin, C., Huang, Y., Cheng, L., Sheu, S., Chen, C., 2006. Bioactive flavonoids from Ruellia tuberosa. J. Chin. Med. 17, 103–109.
Mantilla-Morales, G., Collí-Misset, J., Pozo-Román, F., Rivas-Hernández, A., 2002. Saneamiento y salud: impacto de las enfermedades diarreicas agudas en la península de Yucatán. In: Memorias del XXVIII Congreso de Ingeniería Sanitaria y Ambiental, http://www.bvsde.paho.org/bvsaidis/mexico26/ix-018.pdf (accessed 19.02.13).
Mena-Rejon, G.J., Perez-Espadas, A.R., Moo-Puc, R.E., Cedillo-Rivera, R., Bazzocchi, I.L., Jiménez-Díaz, I.A., Quijano, L., 2007. Antigiardial activity of triterpenoids from root bark of Hippocratea excelsa. J. Nat. Prod. 70, 863–865.
Meurer-Grimes, B., McBeth, D.L., Hallihan, B., Delph, S., 1996. Antimicrobial activity in medicinal plants of the Scrophulariaceae and Acanthaceae. Pharm. Biol. 34, 243–248.
Morimoto, M., Cantrell, C.L., Libous-Bailey, L., Duke, S.O., 2009. Phytotoxicity of constituents of glandular trichomes and the leaf surface of camphorweed, Heterotheca subaxillaris. Phytochemistry 70, 69–74.
Nijveldt, R.J., van Nood, E., van Hoorn, D.E.C., Boelens, P.G., van Norren, K., van Leeuwen, P.A.M., 2001. Flavonoids: a review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 74, 418–425.
Oganesyan, G.B., 2009. Minor flavonols from Dracocephalum multicaule. Chem. Nat. Compd. 45, 242–243.
Peraza-Sanchez, S.R., Poot-Kantun, S., Torres-Tapia, L.W., May-Pat, F., Sima-Polanco, P., Cedillo-Rivera, R., 2005. Screening of native plants from Yucatan for anti-Giardia lamblia activity. Pharm. Biol. 43, 594–598.
Peraza-Sánchez, S.R., Cen-Pacheco, F., Noh-Chimal, A., May-Pat, F., Simá-Polanco, P., Dumonteil, E., García-Miss, M.R., Mut-Martín, M., 2007. Leishmanicidal evaluation of extracts from native plants of the Yucatan peninsula. Fitoterapia 78, 315–318.
Quan, Z., Gu, J., Dong, P., Lu, J., Wu, X., Wu, W., Fei, X., Li, S., Wang, Y., Wang, J., Liu, Y., 2010. Reactive oxygen species-mediated endoplasmic reticulum stress and mitochondrial dysfunction contribute to cirsimaritin-induced apoptosis in human gallbladder carcinoma GBC-SD cells. Cancer Lett. 295, 252–259.
Rahman, A., Choudhary, M.I., Thomsen, W.J., 2001. Bioassay Techniques for Drug Development. Taylor and Francis Group, Netherlands, pp. 32–34.
SINAVE (Sistema Nacional de Vigilancia Epidemiológica), 2010. Boletín Epidemiológico: Semana 1, vol. 28, http://www.dgepi.salud.gob.mx (accessed 07.01.13).
Suleimenov, E.M., Raldugin, V.A., Adekenov, S.M., 2008. Cirsimaritin from Stizolophus balsamita. Chem. Nat. Compd. 44, 398.
Tasdemir, D., Kaiser, M., Brun, R., Yardley, V., Schmidt, T.J., Tosun, F., Rüedi, P., 2006. Antitrypanosomal and antileishmanial activities of flavonoids and their analogues: in vitro, in vivo, structure–activity relationship, and quantitative structure–activity relationship studies. Antimicrob. Agents Chemother. 50, 1352–1364.
Tejman-Yarden, N., Eckmann, L., 2011. New approaches to the treatment of giardiasis. Curr. Opin. Infect. Dis. 24, 451–456.
Treyvaud-Amiguet, V., Arnason, J.T., Maquin, P., Cal, V., Sánchez-Vindas, P., Poveda, L., 2005. A consensus ethnobotany of the Q’eqchi’ maya of southern Belize. Econ. Bot. 59, 29–42.
Vázquez-Tsuji, O., Campos-Rivera, T., 2009. Giardiasis. La parasitosis más frecuente a nivel mundial. Rev. del Centro de Inv. (Méx) 8, 75–90.
Vukovic, N., Sukdolak, S., Solujic, S., Mihailovic, V., Mladenovic, M., Stojanovic, J., Stankovic, M.S., 2011. Chemical composition and antimicrobial activity of Teucrium arduini essential oil and cirsimarin from Montenegro. J. Med. Plants Res. 5, 1244–1250.
Wright, C.W., Allen, D., Cai, Y., Chen, Z., Phillipson, J.D., Kirby, G.C., Warhurst, D.C., Tits, M., Angenot, L., 1994. Selective antiprotozoal activity of some Strychnos alkaloids. Phytother. Res. 8, 149–152.
Yim, S., Kim, H.J., Lee, I., 2003. A polyacetylene and flavonoids from Cirsium rhinoceros. Arch. Pharm. Res. 26, 128–131.
Acknowledgments
This research was supported by the National Council of Science and Technology of Mexico (Conacyt, project No. 105346). We are grateful to Paulino Sima-Polanco for collection and identification of plant material.
Author information
Authors and Affiliations
Contributions
GIHB contributed in running the laboratory work, analysis of the data, structure elucidation of the isolated compounds, and drafted the paper. LWTT contributed to chromatographic and spectrophotometric analysis. RMP supervised biological studies and analysis of the results. SRPS designed the study, supervised the laboratory work, and contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hernández-Bolio, G.I., Torres-Tapia, L.W., Moo-Puc, R. et al. Antigiardial activity of flavonoids from leaves of Aphelandra scabra. Rev. Bras. Farmacogn. 25, 233–237 (2015). https://doi.org/10.1016/j.bjp.2015.04.004
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.04.004