Abstract
Keratose sponges (Keratosa Grant, 1861) of India have not been studied much in detail. The present investigation is part of exploratory sponge taxonomy studies conducted at several localities in the Andaman and Nicobar Islands during 2015–2018. Three keratose sponges distributed among three different families are reported herein for the first time from the Andaman and Nicobar Islands: Dysidea granulosa Bergquist, 1965 (Dysideidae Gray, 1867); Dictyodendrilla cavernosa (Lendenfeld, 1888) (Dictyodendrillidae Bergquist, 1980), Phyllospongia papyracea (Esper, 1806) (Thorectidae Bergquist, 1978). Among these, D. cavernosa is a new record to India. Taxonomic descriptions of all the three species are provided and their distribution within the Andaman and Nicobar Islands vis-à-vis Indo-Pacific is discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Keratose sponges (Keratosa Grant, 1861) have recently been recognized as an important contributor to reefs [1]. These sponges have constituted an informal taxonomic group of non-spicular demospongiae [2] that are characterized by skeletal structures of organic collagenous material, or spongin fibers [1, 3, 4]. However, Keratosa has been genetically and phylogenetically accepted and is currently represented by two orders; viz., Dendroceratida Minchin, 1900 and Dictyoceratida Minchin, 1900 [5, 6].
Keratose sponges of India have not been studied much in detail. Knowledge of taxonomy and diversity of keratose sponges in India mainly stems from the works of Carter [7, 8], Dendy [9, 10], Burton [11], Rao [12], Thomas [13] and Sivaleela [14] in the east coast of India and from Lendenfeld [15], Dendy [16] and Subba Rao and Sastry [17] in the west coast of India. Contributions from the island ecosystems of India include Thomas [18, 19], Prabhakaran [20] and Gopi and Kumar [21] in the Lakshadweep and from Dam Roy et al. [22], Immanuel et al. [23] and Das et al. [24] in the Andaman and Nicobar Islands. More recently, George et al. [25] provided a checklist of marine sponges including keratosa from the peninsular coasts of India. Altogether, a total of 46 keratosa sponges (37 dictyoceratids and nine dendroceratids) distributed among 12 families and 24 genera are reported to be present in India so far.
This report is a part of exploratory sponge taxonomy studies conducted at several localities in the Andaman and Nicobar Islands during 2015–2018. Three keratose sponges distributed among three different families are reported herein for the first time from the Andaman and Nicobar Islands: Dysidea granulosa Bergquist, 1965 (Dysideidae Gray, 1867); Dictyodendrilla cavernosa (Lendenfeld, 1888) (Dictyodendrillidae Bergquist, 1980), Phyllospongia papyracea (Esper, 1806) (Thorectidae Bergquist, 1978). Among these, D. cavernosa is a new record to India. Taxonomic descriptions of all the three species are provided and their distribution within the Andaman and Nicobar Islands vis-à-vis Indo-Pacific according to the marine ecoregions defined by Spalding et al. [26] is discussed.
2 Material and methods
2.1 Keratose sponges reported to be present in India in earlier studies
Scientific literature dealing with records of these sponges from India were critically reviewed from present-day taxonomic, nomenclatural and distribution aspects. All keratose sponges recorded from India were reviewed in consultation with the World Porifera Database [6] and classified into three categories, viz., verified, unverified and inaccurate (Table 1).
2.2 Study area
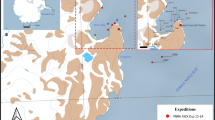
Andaman and Nicobar Islands (06° 45′–13° 41′ N; 92° 12′–93° 57′ E) (Fig. 1, map indicating the position of the Andaman and Nicobar Islands in the Indian Ocean on top left was prepared using SimpleMappr [27]; Table 2) are the emergent peaks of long ridge that extends from the Arakan-Yoma range of Myanmar to Sumatra of Indonesia in a sub-linear north–south direction [28]. The Andaman Island group and the Nicobar Island group are separated by the Ten Degree Channel, which is about 160 km wide [29]. Marine Protected Areas in these Islands comprise nine National Parks and 96 island sanctuaries [30].
Map of the Andaman and Nicobar Islands depicting survey localities (n = 15) where species occurrences of keratose sponges were recorded. See Table 2 for names and geographic coordinates
2.3 Sampling
The studied material came from 15 sub-tidal localities. The habitats were coral reef with various substrates, viz., rocky, sandy and muddy bottoms. Depths of the surveyed localities ranged from 4 to 18 m. Sponges were collected using snorkeling and SCUBA and specimens were fixed and preserved in 70% ethanol and brought to the laboratory.
Histological sections were prepared by serially dehydrating sponge fragments in 70% and by 100% ethanol for 1–2 h and immersing in xylene for 24 h. The fragments were later immersed in paraffin-xylene for 24 h and embedded in paraffin wax. Sections were made using a razor blade and followed by immersion in xylene for dewaxing. Permanent slides were prepared by mounting the sections and spicules using DPX [31, 32]. The sections were then observed and photographed using a stereo-zoom microscope (Make: Leica; Model: M205A) and a compound microscope (Make: Omax; Model: XM82ESC02).
The specimens were identified using the classification scheme of World Porifera Database and Systema Porifera [6, 33]. Original descriptions were used for comparison and establishing the identity of the reported species. The specimens were registered and deposited at the National Zoological Collections at Zoological Survey of India, Andaman Nicobar Regional Centre (ZSI/ANRC), Port Blair.
3 Results
3.1 Systematics
Order Dictyoceratida Minchin, 1900
Definition. Dictyoderatid sponges possess a skeleton made up of spongin fibers, which makes up a significant proportion of the body volume. In a few cases, the sponge may include foreign particles [34].
Family Dysideidae Gray, 1867
Definition. Dysideids are uniquely characterized by the presence of eurypylous choanocyte chambers. They also have concentrically laminated and pithed skeletal fibers (to varying degrees), cored fibers and some species have a sand-armoured surface [34].
Genus Dysidea Johnston, 1842
Definition. Sponges with marked conulose surface in which fibers are filled with detritus [34].
3.1.1 Dysidea granulosa Bergquist, 1965
Synonymy: Dysidea granulosa in Bergquist (1965: 144, Fig. 9).
Material examined: 4 specimens, ZSI/ANRC–23598, August 20, 2016, Point Island, Coll. Sudhanshu Dixit; 2 specimens, ZSI/ANRC–23601, December 26, 2017, Pigeon Island, Coll. M. P. Goutham Bharathi; 2 specimens, ZSI/ANRC–23607, February 24, 2018, Ross Island, Coll. M.P. Goutham Bharathi; 2 specimens, ZSI/ANRC–23612, November 18, 2015, Light House, Coll. Sudhanshu Dixit; 4 specimens, ZSI/ANRC–23616, June 23, 2016, Ross Island, Coll. Preeti Pereira; 3 specimens, ZSI/ANRC–23622, August 5, 2016, North Bay, Coll. Sudhanshu Dixit; 2 specimens, ZSI/ANRC–23625, October 5, 2016, Sir Hugh Ross Island, Coll. Sudhanshu Dixit; 4 specimens, ZSI/ANRC–23627, February 22, 2016, Natural Bridge, Coll. Sudhanshu Dixit; 3 specimens, ZSI/ANRC–23630, September 22, 2016, Rifleman Island, Coll. Tamal Mondal; 2 specimens, ZSI/ANRC–23632, August 22, 2016, Paget Island, Coll. Preeti Pereira; 1 specimen, ZSI/ANRC–23633, September 9, 2017, West Island, Coll. Preeti Pereira; 1 specimen, ZSI/ANRC–23635, September 15, 2017, Rutland Island, Coll. Seepana Rajendra; 1 specimen, ZSI/ANRC–23640, September 10, 2017, Thornhill Island, Coll. Seepana Rajendra; 1 specimen, ZSI/ANRC–23642, September 11, 2018, Roper Point, Coll. M.P. Goutham Bharathi.
Description: Slender finger-like branches arising from narrow base, in some cases even grows as a cushion. Surface covered with minute conules, distance between the conules ca. 1 mm. Oscules scattered over the branches in no apparent pattern. Colour when alive dull gray or white with tinges of purple (Fig. 2A). On preservation, colour lightens to dull white or beige (Fig. 2B); preservative is stained a faint purple. Detritus and foreign particles visible, embedded within the body of the sponge and on breaking a fragment of the sponge.
Skeleton: Made up of spongin fibers and foreign bodies (sand particles) (Fig. 2C); both primary and secondary fibers heavily cored with sand particles, indistinguishable from each other. Fibers arranged in a slightly irregular pattern, forming circular or oval meshes measuring 70–300 µm (Fig. 2D).
Distribution: The present study constitutes the first record of D. granulosa for the Andaman and Nicobar Islands (Fig. 2E). The type locality of this species is Palau, Micronesia, West Caroline Islands [35]. The species has been reported earlier from Lakshadweep [25], though without species description.
Remarks: Although species of Dysidea are morphologically difficult to distinguish [35], D. granulosa can be characterized by its growth form and regular surface conules. The species described herein bears a close similarity to the type material in the morphology (growth form and irregular surface conules) and the structure of spongin fibres with irregular meshes and indistinguishable, heavily cored primary and secondary fibres. Dysidea granulosa is found to be widely distributed in the Andaman and Nicobar Islands (Table 2) and often observed to grow over bivalves as epibionts.
Family Dictyodendrillidae Bergquist, 1980
Definition. Dictyodendrillid sponges are characterized by their reticulate skeleton, with regular to irregular meshes with strongly laminated fibers and a pronounced pith region [36].
Genus Dictyodendrilla Bergquist, 1980
Definition. Sponges with strongly conulose surface, with anastomosing fibres and thick pith [37].
3.1.2 Dictyodendrilla cavernosa (Lendenfeld, 1888)
Synonymy: Dendrilla cavernosa in Lendenfeld (1888: 29–30).
Material examined: 1 specimen, ZSI/ANRC – 23597, March 23, 2018, Sir Hugh Ross Island, Coll. Preeti Pereira.
Description: Grows upright, digitate, branching at the base (Fig. 3A). Branches cylindrical or tubular (hollow), 6–7 cm high and 3 cm wide. Walls thin, measuring 1–3 mm. Surface conulose with thick, sharp conules, 2–4 mm high; distance between the conules 2–3 mm. Oscules scattered on the surface, usually at the apices, with many pseudoscula. Colour when alive is dull yellow or olive green and turns dull brownish on preservation (Fig. 3B).
Dictyodendrilla cavernosa (Lendenfeld, 1888): A In situ, B Preserved specimen, C Skeletal fibers, D Transverse section of the fibre showing pith, E Barnacles encrusting skeletal fibers, F Distribution in the Indo-Pacific: blue closed circle—type locality, red closed circle—subsequent record, green closed circle—present study
Skeleton: Comprised of thickened spongin fibers that project from the surface giving rise to the conules. Fibers thick, 1000–2000 µm, frequently anastomosing and thickest at the base (Fig. 3C). Transverse section of a fiber shows that the pith thickness is almost half the thickness of the fiber (Fig. 3D). Barnacles were found attached to the skeletal fibers within the sponge (Fig. 3E).
Distribution: This constitutes the first record of D. cavernosa for India from the Andaman and Nicobar Islands. Further, it also constitutes a new familial record for Dictyodendrillidae from the Islands. The type species was described from Port Jackson, SE Australia, Manning-Hawkesbury in the East Central Australian Shelf (Temperate Australasia) and the only other record was from Port Philips head, South Australia [38]. This is the only second record of D. cavernosa in the Indo-Pacific since its original description from Australia, highlighting a significant extension of its distribution range to the western Indo-Pacific realm (Fig. 3F).
Remarks: Dictyodendrilla cavernosa is characterized by the distinct, highly conulose surface, erect branching growth form and the pith thickness. Material examined from the Andaman and Nicobar Islands bear close resemblances to the specimens described by Lendenfeld [38] in habit and internal structure: the upright, digitate growth form, surface covered in sharp conules and internally hollow sponge.
Our material was found to be inhabited by a large number of barnacles (Fig. 3E). Barnacles are observed to be camouflaged, probably obtaining food through the canal system of the sponge. Recently, Hosie et al. [39] studied the diversity of dictyoceratid-inhabiting barnacles and highlighted the importance of extensive sampling from different geographical locations. Preliminary observations of D. cavernosa as a host sponge for the associated barnacles from the present study might help improve our understanding of sponge-barnacle symbiosis.
Family Thorectidae Bergquist, 1978
Definition. The family encompasses sponges with a wide variety of growth forms from encrusting to massive, upright and foliose or folio-digitate, in which the spongin fibers making up the finely laminated anastomosing skeleton containing a differentiated pith. The pith is not easily distinguished from the spongin fibers. These sponges lack spicules and their surface is armoured and forms ridges and troughs [34].
Genus Phyllospongia Ehlers, 1870
Definition. Foliose or lamellate sponges with primary and secondary fibres. Primary fibres sometimes armoured with debris, secondary fibres unarmoured [34].
3.1.3 Phyllospongia papyracea (Esper, 1806)
Synonymy: Spongionella holdsworthi in Bowerbank (1873: 25–32, pls. V–VII), Dendy (1905: 217–218 Pl. XIV, Fig. 6), Bergquist et al. 1988 (304–305, Figs. 13–17) and Bergquist (1995, 9).
Material examined: 1 specimen, ZSI/ANRC–23597, October 10, 2018, South Brother Island, Coll. M.P. Goutham Bharathi.
Description: Cup-like with thin walls, 1–2 mm, with a very slender stalk, 1–2 cm in length. Surface smooth and lacks any grooves or ridges. Colour when alive beige with purple areas and it is retained on preservation (Fig. 4A).
Skeleton: Comprised entirely of anastomosing spongin fibers (Fig. 4B). Primary fibers thick, uncored, interconnected at right angles. Secondary fibers run parallel to the surface; much thinner than the primary fibers.
Distribution: Phyllospongia papyracea is reported for the first time from the Andaman and Nicobar Islands. The species has previously been reported from Pamban [12] and is common throughout the Indo-Pacific (Fig. 4C). Originally described from the Great Pearl Bank off Arippu, Sri Lanka [40], P. papyracea is distributed in Madagascar [41], Gulf of Mannar [10] and Sri Lanka and South India [2].
Phyllospongia papyracea has a widespread distribution ranging from temperate Southern Africa to the temperate Australasia. It has been reported from Cape of Good Hope, Agulhas Bank [41], in the Agulhas Province, New Zealand [15], in Temperate Australasia and Easter Island [42] in the Eastern Indo-Pacific realm. Subsequent records from Central Indo-Pacific include Indonesia, Sulawesi Sea [43], Zamboanga, Palawan/North Borneo [44], Tetel Island in the Western Coral Triangle, Solomon Archipelago [45] in the Eastern Coral Triangle, Reef Dolman, New Caledonia [46] in the Tropical SW Pacific, John Brewer Reef, Great Barrier Reef [2] and Central and Southern Great Barrier Reef [47] in the NE Australian Shelf.
Other records of P. papyracea are from Mozambique Island, East African Coral Coast [48], Abu Latt Island, Southern Red Sea [49], Red Sea and Gulf of Aden Province, Aldabra Island, Seychelles [50] in the Western Indian Ocean, Tulear, Grand Récif, Western and Northern Madagascar [51] in the Western Indian Ocean, Hurgada, Northern and Central Red Sea [52] in the Western Indo-Pacific realm.
Remarks: The genus Phyllospongia is represented by two species in the Andaman and Nicobar Islands viz., P. folisascens and P. papyracea. Phyllospongia papyracea is morphologically similar to Phyllospongia foliascens, which is abundant in the Andaman and Nicobar Islands [53]. The differentiating morphological characters between these two similar species are the smooth surface and the uncored or lightly cored fibers in P. papyracea as opposed to the strongly verrucose surface and the strongly cored fibers of P. foliascens. The material examined from the Andaman and Nicobar Islands were assigned to P. papyracea on account of the thin and lamellate body appearing like a stalked fan. The skeleton is comprised of uncored primary and secondary fibres, typical of the genus Phyllospongia [2].
4 Discussion
Among the 46 keratose sponges reported to be present in India, distribution of eight species in India is unverified while the distribution of a further 13 species is inaccurate (Table 1). Several research works cited in Table 1 are old and require extensive taxonomic revisions and these species are in critical need of description and characterization.
In the present study, three keratose sponges are reported for the first time from the Andaman and Nicobar Islands: Dysidea granulosa, Dictyodendrilla cavernosa and Phyllospongia papyracea. Among these, D. granulosa is found to be commonly distributed in the Islands while D. cavernosa and P. papyracea are found to be rare (known from single specimens) and recorded only from Sir Hugh Ross Island and South Brother Island, respectively (Table 2). Future works on keratose sponges must expand the geographic scope of sampling for authenticating the distribution ranges of these intriguing taxa in the Indo–Pacific.
Data availability
All data generated or analysed during this study are included in this published article.
References
Luo C, Reitner J. First report of fossil “keratose” demosponges in Phanerozoic carbonates: preservation and 3-D reconstruction. Naturwissenschaften. 2014;101:467–77. https://doi.org/10.1007/s00114-014-1176-0.
Bergquist PR, Ayling AM, Wilkinson CR. Foliose Dictyoceratida of the Australian Great Barrier Reef. I. Taxonomy and phylogenetic relationships. Mar Ecol. 1988;9:291–319. https://doi.org/10.1111/j.1439-0485.1988.tb00209.x.
Erpenbeck D, Sutcliffe P, Cook SDC, Dietzel A, Maldonado M, van Soest RWM, et al. Horny sponges and their affairs: on the phylogenetic relationships of keratose sponges. Mol Phylogenet Evol. 2012;63:809–16. https://doi.org/10.1016/j.ympev.2012.02.024.
Pham D, Hong J, Lee JH. Keratose sponge–microbial consortia in stromatolite-like columns and thrombolite-like mounds of the Lower Ordovician (Tremadocian) Mungok Formation, Yeongwol, Korea. Palaeogeogr Palaeoclimatol Palaeoecol. 2021;572: 110409. https://doi.org/10.1016/j.palaeo.2021.110409.
Morrow C, Cárdenas P. Proposal for a revised classification of the Demospongiae (Porifera). Front Zool. 2015;12:1–27. https://doi.org/10.1186/s12983-015-0099-8.
de Voogd NJ, Alvarez B, Boury-Esnault N, Carballo JL, Cárdenas P, Díaz M-C, Dohrmann M, Downey R, Hajdu, E, Hooper JNA, Kelly M, Klautau M, Manconi R, Morrow CC, Pisera AB, Ríos P, Rützler K, Schönberg C, Vacelet J, van Soest RWM. 2023. World Porifera Database. https://www.marinespecies.org/aphia.php?p=taxdetails&id=366651. Accessed 19 Apr 2023.
Carter HJ. Report on specimens dredged up from the Gulf of Manaar and presented to the Liverpool Free Museum by Cap. W.H. Cawne Warren. J Nat Hist. 1880;6:35–61. https://doi.org/10.1080/00222938009458893.
Carter HJ. Supplementary report on Specimens dredged up from the Gulf of Manaar, together with others from the Sea in the Vicinity of the Basse Rocks and from Bass’s Straits respectively, presented to the Liverpool Free Museum by Capt. H. Cawne Warren. J Nat Hist. 1881;7:361–85. https://doi.org/10.1080/00222938109459534.
Dendy A. The Sponge-fauna of Madras. A report on a collection of sponges obtained in the neighbourhood of Madras by Edgar Thurston, Esq. Ann Mag Nat Hist. 1887;20:153–65. https://doi.org/10.1080/00222938709460032.
Dendy A. Report on the sponges collected by Professor Herdman, at Ceylon, in 1902, In: Herdman WA, editor. Report to the Government of Ceylon on the Pearl Oyster Fisheries of the Gulf of Manaar. 3 (Supplement 18), Royal Society, London; 1905. pp. 57–246, pls I–XVI.
Burton M. Supplement to the littoral fauna of Krusadai Island in the Gulf of Manaar: Porifera (Vol. 1, No. 2). Bulletin of the Madras Government Museum, Superintendent, Government Press; 1937.
Rao HS. Indian and Ceylon sponges of the Naturhistoriska Riksmuseet, Stockholm, collected by K. Fristedt. Rec Zool Surv India. 1941;43:417–69.
Thomas PA. Demospongiae of the Gulf of Mannar and Palk Bay. In: James PSBR, editor. Recent advance in marine biology. New Delhi: Today Tomorrow’s printers and publishers; 1985. p. 205–365.
Sivaleela G. Marine sponges of Gulf of Mannar and Palk Bay. Rec Zool Surv India. 2014;114:607–22.
von Lendenfeld R. A monograph of the horny sponges. London: Trübner and Co.; 1889. p. 1–936.
Dendy A. Report on the non-Calcareous Sponges collected by Mr. James Hornell at Okhamandal in Kattiawar in 1905–6. Report to the Government of Baroda on the Marine Zoology of Okhamandal in Kattiawar. 1916; 2: 93–146, pls I–IV.
Subba Rao NV, Sastry DRK. Fauna of Marine National Park, Gulf of Kutch (Gujarat): An overview. Conservation Area Series 23, 1–79, pls I–VIII. (Published by the Director, Zoological Survey of India, Kolkata; 2005.
Thomas PA. Demospongiae of Minicoy Island (Indian Ocean) Part 1 – Orders Keratosa and Haplosclerida. J Mar Biol Assoc India. 1979;21:10–6.
Thomas PA. Sponge fauna of Lakshadweep. Bull Cent Mar Fish Res Inst. 1989;43:150–61.
Prabhakaran MP. Ecological studies on the seagrass ecosystem of Minicoy Lagoon Lakshadweep. Ph.D. Thesis, Cochin University of Science and Technology; 2008.
Gopi M, Kumar TTA. Sponge Fauna of Lakshadweep Archipelago. Seshaiyana. 2012;20:3–5.
Dam Roy S, Krishnan P, Raghunathan C, Vinith Kumar NV. Cataloguing and Conservation of Marine Sponges of Andaman through DNA Barcoding. Project Completion Report, Port Blair; 2013.
Immanuel T, Krishnan P, Raghunathan C. An updated report on the diversity of marine sponges of the Andaman and Nicobar Islands. In: Sivaperuman C, Venkataraman K, editors. Marine faunal diversity in India: taxonomy, ecology and conservation. Amsterdam, Cambridge: Elsevier, Academic press; 2015. p. 3–13.
Das RR, Immanuel T, Lakra RK, Baath K, Ganesh T. First report of marine sponge Chelonaplysilla delicata (Demospongiae: Darwinellidae) from the Andaman Sea/Indian Ocean with baseline information of epifauna on a mesophotic shipwreck. J Threat Taxa. 2022;14:21961–7. https://doi.org/10.11609/jott.7495.14.10.21961-21967.
George AM, Van Soest RW, Sluka RD, Lazarus S. A checklist of marine sponges (Porifera) of peninsula India. Zootaxa. 2020;4885:277–300. https://doi.org/10.11646/zootaxa.4885.2.10.
Spalding MD, Fox HE, Allen GR, Davidson N, Ferdaña ZA, Finlayson MAX, Halpern BS, Jorge MA, Lombana AL, Lourie SA, Martin KD. Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. Bioscience. 2007;57:573–83. https://doi.org/10.1641/B570707.
Shorthouse DP. SimpleMappr, an online tool to produce publication-quality point maps. 2010; https://www.simplemappr.net. Accessed 19 Aug 2023.
Bandopadhyay PC, Carter A. Introduction to the geography and geomorphology of the Andaman-Nicobar Islands. Geol Soc London. 2017;47:9–18.
Tikader BK, Das AK. Glimpses of animal life of Andaman and Nicobar Islands. Calcutta: Zoological Survey of India; 1985.
Laxmilatha P, Sruthy TS, Varsha MS. Marine protected areas. In: Joshi KK, editor. Summer school on recent advances in marine biodiversity conservation and management. Kochi: Central Marine Fisheries Research Institute; 2015. p. 199–206.
Ackers RG, Moss D, Picton BE, Stone SM, Morrow CC. Sponges of the British Isles (“Sponge V”)—a colour guide and working document. Belfast: Marine Conservation Society/Ulster Museum; 2007.
Slaoui M, Fiette L. Histopathology procedures: from tissue sampling to histopathological evaluation. In: Gautier JC, editor. Drug safety evaluation. Methods in molecular biology (methods and protocols), vol. 691. Humana Press: New York; 2011. p. 69–82. https://doi.org/10.1007/978-1-60761-849-2_4.
Hooper JNA, Van Soest RW, editors. Systema Porifera. A guide to the classification of sponges. New York: Kluwer Academic/ Plenum Publishers (Springer); 2002. https://doi.org/10.1007/978-1-4615-0747-5.
de Cook SC, Bergquist PR. Family Thorectidae Bergquist, 1978. In: Hooper JNA, Van Soest RW, editors. Systema Porifera. Boston: Kluwer Academic/Plenum Publishers, Springer; 2002. p. 1028–50.
Bergquist PR. The sponges of micronesia, Part I. The Palau Archipelago Pac Sci. 1965;19:123–204.
Bergquist PR, de Cook SC. Family Dictyodendrillidae Bergquist, 1980. In: Hooper JNA, Van Soest RW, editors. Systema Porifera. Boston: Kluwer Academic/Plenum Publishers, Springer; 2002. p. 1072–6.
Bergquist PR. A revision of the supraspecific classification of the orders Dictyoceratida, Dendroceratida and Verongida (class Demospongiae). N Z J Zool. 1980;7:443–503. https://doi.org/10.1080/03014223.1980.11760680.
von Lendenfeld R. Descriptive catalogue of the sponges in the Australian Museum, Sidney. London: Taylor and Francis; 1888. p. 1–260.
Hosie AM, Fromont J, Munyard K, Wilson NG, Jones DS. Surveying keratose sponges (Porifera, demospongiae, Dictyoceratida) reveals hidden diversity of host specialist barnacles (Crustacea, Cirripedia, Balanidae) Mol. Phylogenet Evol. 2021;161: 107179. https://doi.org/10.1016/j.ympev.2021.107179.
Bowerbank JS. Report on a Collection of Sponges found at Ceylon by E.W.H. Holdsworth, Esq. Proc Zool Soc Lond.1873; 25–32, pls V–VII.
Hyatt A. Revision of the North American Poriferae; with Remarks upon Foreign Species. Part II. Memoirs of the Boston Society of Natural History. 1877; 2:481–554, pls XV–XVII.
Desqueyroux-Faúndez R. Sponges (Demospongiae) from Easter Island (Isla de Pascua) (South Pacific Ocean). Rev Suisse Zool. 1990;97:373–410.
Thiele J. Studien über pazifische Spongien. II. Ueber einige Spongien von Celebes. Zoologica. Original-Abhandlungen aus dem Gesamtgebiete der Zoologie. Stuttgart. 1899; 24: 1–33, pls I–V
Lévi C. Spongiaires des Iles Philippines, principalement récoltées au voisinage de Zamboanga. Philipp J Sci. 1961;88:509–33.
Bergquist PR, Morton JE, Tizard CA. Some Demospongiae from the Solomon Islands with descriptive notes on the major sponge habitats. Micronesica. 1971;7:1–2.
Bergquist PR. Dictyoceratida, Dendroceratida and Verongida from the New Caledonia Lagoon (Porifera: Demospongiae). Mem Queensl Mus. 1995;38:1–51.
Hooper JNA, Wiedenmayer F. Porifera. In: Wells A, editor. Zoological catalogue of Australia, vol. 12. Melbourne: CSIRO; 1994.
Ridley SO. Spongiida. Report on the Zoological Collections made in the Indo-Pacific Ocean during the Voyage of H.M.S. ‘Alert', 1881–2. (British Museum (Natural History): London). 1884; 366–482, pls 39–43; 582–630, pls 53–54.
Lévi C. Résultats scientifiques des Campagnes de la ‘Calypso'.Campagne 1951–1952 en Mer Rouge (suite). 11. Spongiaires de Mer Rouge recueillis par la ‘Calypso' (1951–1952). Annales de l'Institut océanographique. 1958; 34: 3–46.
Lévi C. Résultats scientifiques des Campagnes de la ‘Calypso'. Campagne 1954 dans l'Océan Indien (suite). 2. Les spongiaires de l'Ile Aldabra. Annales de l'Institut océanographique. 1961; 1–32, pls 1–2.
Vacelet J, Vasseur P. Spongiaires des grottes et surplombs des récifs de Tuléar (Madagascar). Recueil des Travaux de la Station marine d’Endoume. 1965;2–4:71–123.
Helmy T, El Serehy H, Mohamed SZ, Van Soest RWM. Description and classification of dictyoceratid sponges from the northern Red Sea. Beaufortia. 2004;54:81–91.
Pereira P, Raghunathan C. Diversity of sponges in marine protected areas of North Andaman, India. Thalassas. 2018;34:361–72. https://doi.org/10.1007/s41208-018-0073-2.
Acknowledgements
The authors would like to thank the Director, Zoological Survey of India, Kolkata and the Officer-in-Charge, Andaman and Nicobar Regional Centre of Zoological Survey of India, Port Blair for the facilities.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Methodology, investigation, writing—original draft preparation: PAP; Conceptualization, investigation, supervision, writing—review and editing: CR.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors agree with the publication of this article.
Competing interests
The authors report there are no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pereira, P.A., Raghunathan, C. Three new records of Keratosa sponges (Demospongiae: Porifera) from the Andaman and Nicobar Islands, India. Discov Oceans 1, 2 (2024). https://doi.org/10.1007/s44289-024-00002-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44289-024-00002-z