Abstract
Purpose
While coronavirus disease 2019 (COVID-19) has emerged as a global pandemic, millions of patients with COVID-19 have recovered and returned to their families and work, although the key factors of sequela-free survival remain unknown. Here we determine key factors associated with sequela-free survival of patients with COVID-19.
Methods
Sequela-free survival is defined as having none of the long-term sequelae measured with the Activity of Daily Living (ADL) Scale, modified Medical Research Council (mMRC) Dyspnoea Scale, New York Heart Association (NYHA) classification, Self-rating Depression Scale (SDS), Carcinologic Handicap Index (CHI) and Modified Telephone Interview for Cognitive Status (TICS-M). Multivariable logistic regression was used to examine the key factors of sequela-free survival.
Results
The follow-up period for the 464 patients was 10 August to 30 September 2020. Of these, 424 patients completed the assessment of all scales. 30.2% (128 of 424) were categorised as sequela-free at follow-up. The most common sequelae were psychosocial problems (254 [57.7%]), respiratory function abnormality (149 [32.6%]) and cardiac function abnormality (98 [21.5%]). Risk factors associated with COVID-19 sequelae were anaemia on admission, longer duration from the onset of symptoms to admission and increasing of age, whereas anaemia treatment was a protective factor against sequelae. A haemoglobin level of < 113 g/L for female patients or < 92 g/L for male patients at admission discriminated a likely higher risk of any COVID-19 sequelae.
Conclusions
Only one third COVID-19 survivors were sequela-free. Anaemia on admission, longer duration from the onset of symptoms to admission and increasing of age were the risk factors of any long-term sequela. Anaemia treatment was associated with beneficial outcomes in COVID-19 survivors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has emerged as a global pandemic. At the time of this writing, > 100.0 million confirmed cases and 2.9 million deaths globally have been reported based on weekly data from the World Health Organization. Fortunately, many kinds of vaccine have been proven effective worldwide, which would be helpful to control the pandemic [1,2,3,4]. Although many patients with COVID-19 remain hospitalised, millions of people have recovered from COVID-19 and returned to family and work. The long-term prognosis of a large number of patients has become a public health issue. Huang et al. [5] have preliminarily revealed that 76% of patients have reported at least one symptom at 6-month follow-up, including weakness, sleep difficulties, anxiety and depression. Although Huang et al. demonstrated that female sex and higher severity scale score were risk factors of many consequences, such as lung diffusion impairment, anxiety or depression and fatigue or muscle weakness, the key associated factors of sequela-free survival after 6 months were unknown.
Identifying key factors of sequela-free survival of patients with COVID-19 is undoubtedly important so that clinicians can provide relatively early intervention, make comprehensive assessments and appropriate decisions needed in daily clinical practice to reduce or prevent long-term harm, enabling more patients to fully recover and return to normal life and work. This study followed the 6-month outcomes of 464 patients with COVID-19 discharged from Leishenshan Hospital, which was one of the designated COVID-19 hospitals in Wuhan, China and aimed to identify key factors that may account for sequela-free recovery from COIVD-19 to advance the knowledge of the disease and guide the current clinical treatment strategies.
2 Methods
2.1 Study design and participants
This research was designed as a retrospective‐prospective cohort study. The long-term outcomes were assessed for all patients admitted between 15 February and 1 April 2020 at Leishenshan Hospital, which was one of the hospitals designated to treat patients with COVID-19 in Wuhan, China. All study participants met the criteria according to the Chinese clinical guidance for the COVID-19 pneumonia diagnosis and treatment issued by the National Health Commission [6]. This study was approved by the Institutional Research Ethics Committee of Renji Hospital, School of Medicine, Shanghai Jiaotong University (ethics committee approval No. KY2020-037). Oral consent was acquired from patients or from their immediate family members in cases of communication disorder or death.
2.2 Data collection
The medical records for all patients were independently obtained by the authors, who worked for the Critical Care Medicine Department of Leishenshan Hospital at that time. Laboratory data were reviewed from electronic medical records. The medical information collected included patient age, sex, laboratory tests and comorbidities at admission, such as cardiac, cerebrovascular, pulmonary, and neurologic diseases, as well as malignancies, diabetes, and anaemia (haemoglobin < 120 g/dL for males and < 110 g/dL for females). In accordance with clinical reference values, other laboratory items were classified as normal or abnormal.
2.3 Outcomes
The long-term outcome data were collected by telephone interview and then assessed with a series of evaluations by trained operators (physicians and nurses) following a standardised instruction for practicing questionnaires and data recording in a formal case report form (CRF). CRF comprised six items, namely the Activity of Daily Living (ADL) Scale [7]; modified Medical Research Council (mMRC) Dyspnoea Scale [8]; Modified Telephone Interview for Cognitive Status (TICS-M) [9]; Self-rating Depression Scale (SDS) [10] and Carcinologic Handicap Index (CHI), which contains psychosocial, pain, swallowing, feeding, phonation, hearing, vision, olfaction and gustation domains [11], and New York Heart Association (NYHA) classification [12]. The results of the different questionnaires were compared and analysed for functional measures. The final intake for follow-up data was on 30 September 2020.
2.4 Statistical analysis
Baseline characteristics were analysed with descriptive statistics. Continuous variables are described as mean and standard deviation (SD) or median and the Interquartile Range (IQR)), whichever is appropriate, and frequencies and percentages are used to describe categorical variables.
Participants were categorised into two groups, a sequela-free group and a sequelae group, according to their questionnaire results for the following scales: ADL (Barthel Index), mMRC, SDS, CHI and NYHA. In this study, we defined "sequela-free" as a patient showed no change in any of the scales. Otherwise, the patient was classified into the sequelae group. Univariate analyses were conducted to screen out the possible association between candidate predictors and the outcome, and factors identified were thereafter included in a multivariate logistic regression, with adjustments for age, sex and intensive care unit (ICU) admission. Adjusted odds ratios and their precision (i.e., 95% confidence interval (CI)) are presented.
To maintain the sample size and therefore strengthen the statistical power of our analyses, missing values of any covariate in the final multivariate logistic model were handled using multiple imputation with a random sample from observed values. No imputation was conducted on outcomes with missing values. Moreover, to evaluate the robustness of the model, we also performed several other multiple imputations using different procedures.
We also determined the cut-off haemoglobin level in each sex subgroup, which maximised the odds ratio (OR), to provide more information on future decisions with regard to maintaining the haemoglobin level. This was followed by a multivariate reanalysis using this new cut-off point as the criterion for diagnosing anaemia.
All analyses were performed using the open-source statistical software R (version 4.0.2), and hypothesis tests were two-sided at an α level of 0.05.
3 Results
3.1 Patient demographic and baseline characteristics
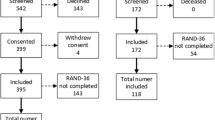
This study sought to assess the outcomes of 990 patients aged ≥ 18 years with confirmed COVID-19 who were admitted at Leishenshan Hospital between 15 February and 1 April 2020 and discharged as survivors. Of the 990 patients, 8 (1%) were excluded because of post-discharge all-cause death, 313 (31%) patients could not be reached by telephone and 205 (21%) patients refused to provide information, leaving 464 (47%) patients eligible for analysis (Fig. 1; Table S1).
The median age of eligible patients was 57 years (15–93 years); 236 (51.5%) were men. The two most common comorbidities were hypertension (25%) and diabetes (12.1%). Approximately 5% of patients had ischaemic heart disease. The incidence of other comorbidities was < 5% for the following: cancer; respiratory, cerebrovascular and non-ischaemic heart diseases; liver and renal dysfunctions and hypoproteinaemia. Besides receiving the conventional symptomatic supportive treatment, 13 patients (2.8%) received anaemia treatment (Table 1).
During hospitalisation, 19 (4.1%) patients were transferred to the ICU. The mean duration of hospital stay of all patients was 19.4 days (range, 2–50 days); the mean duration of ICU stay was 11.7 days (range, 2–32 days). The mean duration from symptom onset to admission was 24.4 days (range, 1–68 days), and the mean time from discharge to follow-up was 160.6 days (range, 79–183 days). Laboratory tests on admission are shown in Table 2. Of the patients, 44.1% had d-dimer levels of > 0.5 mg/L; 21.3% had haemoglobin levels of < 120 g/L for males or 110 g/L for females; 2.5% had procalcitonin levels of ≥ 0.5 ng/mL; 13.5% had C-reactive protein levels of > 10 mg/L; 20.8% had alanine aminotransferase levels of > 40 U/L; 8.7% had aspartate aminotransferase levels of > 40 U/L; 26.2% had albumin levels of < 35 g/L and 2.5% had creatinine levels of > 133 μmol/L. On admission, lymphocytopaenia was present in 7.4% of the patients; thrombocytopenia, in 2.1% and leukopenia, in 11.7%. The median neutrophil-to-lymphocyte ratio of patients on admission was 2.0.
3.2 Long-term sequelae
Long-term sequelae were observed in the population of COVID-19 survivors (Fig. 2; Table S2), with some patients having missing data in different sections because of refusal to provide the relative information.
Long-term sequelae in the COVID-19 survivor population. Of 464 COVID-19 survivors who were followed up, 424 patients completed the assessment of all scales. 128 (30.2%) were categorised as sequela-free; 2 (0.4%) were assessed to have a worsened Barthel Index without other sequelae; 4 (0.9%) were assessed to have an increased NYHA score without other sequelae; 1 (0.2%) was assessed to have an increased SDS grade without other sequelae; 10 (2.2%) were assessed to have an increased mMRC grade without other sequelae and 126 (27.2%) were assessed to have any sequelae, including psychosocial disability, pain, feeding difficulties, dysphonia, hyposmia and impaired hearing, vision, swallowing or gustation. Two patients were assessed to have two to four different sequelae, while two (0.4%) had all kinds of sequelae. COVID-19, coronavirus disease 2019; mMRC, modified Medical Research Council; NYHA, New York Heart Association; SDS, Self-rating Depression Scale; ADL, activity of daily living
Most patients retained similar capacities of ADL as those immediately before the onset of COVID-19. Assessment on the ADL scale for 459 patients showed a decreased Barthel Index score for 70 (15.3%) patients. Of these 70 patients, 61 (13.3% of 459) had a ≥ 1-point reduction on the ADL scale and 5 (1.1%) had ≥ 2-point reduction.
Moderate cardiopulmonary functional abnormalities were observed. On the mMRC scale for 457 patients, 149 (32.6%) patients experienced worse dyspnoea after COVID-19, 43 (9.4%) of whom had a ≥ 2-point increase in dyspnoea grade. On NYHA classification for 456 patients, 98 (21.5%) patients had impaired cardiac function after COVID-19, 17 (3.7%) of whom had an increase in NYHA functional class ≥ 2.
Psychosocial abnormalities were manifested to varying degrees. On screening by the psychosocial part of CHI scale for 441 patients, 254 (57.7%) had commonly experienced psychosocial problems. To explore these psychosocial problems in detail, the levels of depression and anxiety were evaluated. On the SDS questionnaire for of 434 patients, 152 (35.0%) had increased SDS scores, whereas none had met depression criteria before COVID-19. Of these patients, 18 (4.2%) had an increase of ≥ 1 in depression grade and 6 (1.4%) had an increase of ≥ 2. Cognitive function was also assessed; on TICS-M screening for 383 patients, most (376 [98.2%]) were diagnosed with normal cognitive function.
Minor sensory functional abnormalities were manifested. Assessment on the CHI scale for 441 patients showed hearing impairment in 40 (9.1%), visual impairment in 37 (8.4%), hyposmia in 27 (6.1%) and gustation impairment in 18 (4.1%). Other findings were pain in 55 patients (12.5%), swallowing difficulty in 30 (6.8%), feeding difficulty in 54 (12.2%) and dysphonia in 46 (10.4%). Overall, increased CHI scores were noted for 274 (62.1%) patients after COVID-19.
3.3 Univariate analysis of the 6-month consequences of COVID-19
Of the patients, 424 patients completed the assessment of all scales. 30.2% (128/424) were categorised as sequela-free at follow-up, while 63.8% reported at least one sequela. Here, we compared a completely healthy cohort and patients with sequela. Patients in the sequela-free group were significantly younger than those in the sequela group (52.2 SD 15.6 vs. 58.4 SD 12.8, p < 0.001). The duration from symptom onset to admission in the sequela-free group was significantly shorter than that in the sequela group (22.1 SD 12.3 vs. 25.3 SD 13.9, p = 0.046). The proportion of patients with anaemia, whose haemoglobin level on admission was < 120 g/L for males or 110 g/L for females, in the sequela-free group was significantly lower than that in the sequela group (13.6% vs. 23.4%, p = 0.034). The proportion of patients who were receiving anaemia treatment was significantly higher in the sequela-free group than that in the sequela group (5.5% vs. 1.4%, p = 0.014). No significant difference was observed in sex, hospitalisation, ICU admission, comorbidities or other laboratory findings between the two cohorts (Table 3).
3.4 Multivariate analysis of the 6-month consequences of COVID-19
Increasing of age, which means 1-year increase in age; anaemia on admission and longer duration from symptom onset to admission, which means each additional day, were risk factors of at least one sequela after COVID-19, while anaemia treatment was a protective factor against sequelae. On further multivariate analysis with multiple imputation using random samples from observed values (Table 4), the increased odds of at least one sequela was found in patients with increasing of age (OR, 1.03; 95% CI, 1.01–1.05; p = 0.001), anaemia (OR, 2.30; 95% CI, 1.16–4.57; p = 0.017) and longer duration from symptom onset to admission (OR, 1.02; 95% CI 1.00–1.04; p = 0.019). Furthermore, a decreased odds of at least one sequela was observed in patients with anaemia treatment (OR, 0.05; 95% CI, 0.01–0.25; p < 0.001). Similar results were observed in the original multivariate analysis and other multiple imputations (Table S3).
3.5 Haemoglobin and anaemia treatments are strongly associated with COVID-19 sequelae
Considering that anaemia diagnosed on the basis of the haemoglobin level at admission as a strong indicator of COVID-19 sequelae and the significant association of anaemia treatment with the long-term outcomes of patients with COVID-19, we further analysed the performance of haemoglobin level on admission in determining the optimal cut-off value. A haemoglobin level of 113 g/L was identified as the optimal threshold with a maximum risk ratio (2.15) and sufficient sensitivity (0.81) to predict the risk of sequelae after COVID-19 in female patients, with an area under the receiver-operating characteristic curve (AUROC) value of 0.57. At the same time, a haemoglobin level of 92 g/L was identified as the optimal threshold with a maximum risk ratio (4.85) and sufficient sensitivity (0.99) to predict the risk of sequela after COVID-19 in male patients, with an AUROC of 0.61 (Figure S1).
To further validate the performance of risk stratification using a haemoglobin level of 113 g/L for female patients and 92 g/L for male patients as the cut-off values, we performed a multivariate reanalysis of the 6-month consequences of COVID-19 by using the cut-off values as the diagnostic criteria for anaemia (Table 4). An increased odds of at least one sequela was found in patients with increasing of age (OR, 1.03; 95% CI, 1.01–1.05; p = 0.001), anaemia (OR, 3.54; 95% CI, 1.27–9.88; p = 0.016) and longer duration from symptom onset to admission (OR, 1.03; 95% CI, 1.01–1.05; p = 0.010). Furthermore, a decreased odds of at least one sequela was found in patients who received anaemia treatment (OR, 0.02; 95% CI, 0–0.14; p < 0.001). Specific anaemia treatment was shown in Table S4.
4 Discussion
This study provided data for 464 patients who recovered from COVID-19 and were followed up at 6 months after discharge from a special hospital in Wuhan, China. Of the COVID-19 survivors, 30.2% were sequela-free and 69.8% had at least one long-term sequelae in this population of COVID-19 survivors. The main long-term sequelae were cardiopulmonary dysfunction and psychosocial problems. Anaemia on admission, longer duration from symptom onset to admission and increasing of age were risk factors of any COVID-19 sequelae, whereas anaemia treatment was a protective factor against sequelae. Haemoglobin levels < 113 g/L for female patients or < 92 g/L for male patients at admission discriminated a likely higher risk of any COVID-19 sequelae.
The main long-term sequelae after 6 months were respiratory, cardiac, and psychosocial problems, which were similar with those reported in other studies. COVID-19 survivors were reported to be mainly troubled with fatigue or muscle weakness, sleep difficulties, and anxiety or depression at 6 months after infection in a cohort study with 1733 subjects [5]. A cohort study including 154,068 individuals showed that in the postacute phase of COVID-19, there was increased risk of an array of incident neurologic sequelae, estimating the hazard ratio as 1.42 at 12 months [13]. A similar cohort study including 153,760 individuals provided evidence that risk and 1-year burden of cardiovascular disease in survivors of acute COVID-19 were substantial [14]. The incidence of worse dyspnoea after COVID-19 was 32.6% in the present study. Likewise, pulmonary diffusion abnormalities were found in a considerable proportion (22%–56%) of patients assessed based on lung function 6 months after symptom onset in Jinyintan Hospital, Wuhan, China [5]. Moreover, fibrosis was found in patients with COVID-19 at > 30 days after hospital discharge [15], and pulmonary interstitial changes, such as ground-glass opacity and irregular lines, were observed 6 months after symptom onset [5], which may be the reason why patients developed respiratory sequelae in the present study.
Although the primary organ system affected by the virus was the lungs, decreased cardiac function was suggested in 21.5% of patients. Increased resting heart rate and discontinuous flushing were reported in a 3-month follow-up study [16], and the median 6-min walking distance of 22%–29% of patients was less than the lower limit of the normal range in a 6-month follow-up study [5]. Previous hospitalisation data showed that > 20% of patients presented with significant myocardial injuries, including infection-related myocarditis with reduced systolic function and arrhythmias [17]. Moreover, cardiovascular pathologists confirmed the presence of myocarditis in the autopsies of 21 patients [18]. These myocardial injuries could be secondary to severe lung damage. However, another study regarded direct infection of heart as a more plausible option [19]. Besides, numerous case reports link postural orthostatic tachycardia syndrome to post-acute sequela of COVID-19 [20, 21]. Prospective observational evaluation in patients with post-acute sequela of COVID-19 revealed suggestive of autonomic dysfunction on nearly all subjects [22]. Possible contributing of factors for patients diminished cardiovascular fitness include muscle weakness, residual lung damage and physical deconditioning.
In the present study, we found that 13.3% of patients were assessed using the Barthel Index for reduced capacities in ADL that focused on stair climbing. Likewise, fatigue and muscle weakness were the most common symptoms reported by 63% of patients in a 6-month follow-up study [5]. It is worth recognising the psychosocial impact of COVID-19, which likely creates an additional burden with social restrictions for patients. More than 50% of patients encountered psychosocial problems in the present study, even more serious than those observed in the SARS outbreak [23]. The main reason may be the panic caused by an alien pandemic, breakdown of social networks and fear of mortality. Chronic pain, symptoms of weakness and worrying about sequelae may further increase the potential for development of anxiety and depression. Therefore, early intervention incorporating physical and psychological therapy and pain management should be carried out to reach the goal of improving function and quality of life for these patients.
In the present study, only 30.2% of COVID-19 survivors were sequela-free, consistent with a previous study [5]. According to observations of the current pandemic, many survivors could develop long-term functional and psychosocial sequelae. Therefore, we developed a multivariate regression model to predict sequela-free survival by using demographic and clinical characteristics at admission.
We found that patients with increasing of age, which means a 1-year increase in age; anaemia on admission and longer duration from symptom onset to admission (an additional day) are more likely to have COVID-19 sequelae, while anaemia treatment was a predictor of sequela-free survival. Age has been reported to be associated with both short- and long-term sequelae [5, 16]. It is understandable that patients with increasing of age are more vulnerable to sequelae, due to increased likelihood that a person will have comorbidities in older age, the medications that some of these patients take for their comorbidities, and the ageing immune system perhaps with greater viral replication [24]. This factor, although non-modifiable, may call for action to provide early intervention both during hospitalisation and after discharge, hopefully improving the eventual outcome for elderly patients. In addition, elderly people might be given priority for vaccination if clinical safety permits.
A prolonged duration of symptom onset to hospitalisation may lead to severe cases and poor outcomes. This finding is consistent with those of previous studies [25, 26]. Early intervention benefited patients’ recovery from critical illness, reminding clinicians and patients not to miss the valuable window of treatment. At present, the delay in treatment could be inevitable considering the current pandemic situation, and COVID-19 is a rapidly evolving and emerging situation. Here, we call on public health departments and medical institutions to strengthen medical investment and provide more beds for patients with COVID-19 to reduce delays in treatment. In case of an outbreak of local epidemics, establishing a field hospital in different areas [27, 28] is recommended to receive and treat patients to decompress hospitals, which could lead to more sequela-free survivors.
Moreover, the most encouraging finding of our study was that haemoglobin and anaemia treatment were strongly associated with sequelae-free survival from COVID-19, which has not been reported in other studies yet. Accumulated evidence from previous clinical studies suggests that patients with COVID-19 had low haemoglobin levels [29, 30], while severe cases showed significantly lower [31, 32] and lower haemoglobin concentration has been shown to be a risk factor of severe illness in patients with COVD-19 [33]. In our study, 21.3% of patients were diagnosed as having anaemia at admission. This phenomenon, as reported, might be due to the lack of synthetic materials for haemoglobin, such as iron, due to the poor appetite and abnormal diet after SARS-CoV-2 infection [34]. As known to all, deoxyhaemoglobin confers a higher risk of viral attacks than oxidised haemoglobin, which may cause respiratory distress symptoms because of lesser oxygen carrier haemoglobin in the body. Though controversial, a computational proteomic analysis revealed that some SARS-CoV-2 proteins could bind to porphyrins [35], which is an essential component of a haeme group. In fact, blood transfusion has been recommended by physicians and front-line healthcare professionals in the management of patients with anaemia to improve the condition of clinically complex conditions [35]. Hence, despite that, no strict evidence has been found to prove the attack of blood cells, physicians should follow the principle of active treatment for anaemia. Moreover, our analysis has shown that the optimal cut-off haemoglobin level is 113 g/L for female patients and 92 g/L for male patients, which suggests that females with anaemia should maintain high levels of haemoglobin by transfusion of red blood cells, erythropoietin and diet as prescribed by their attending physicians. Complete blood cell test is the most commonly available assay in regions with different levels of medical resources, and anaemia treatment is a modifiable factor. Therefore, medical staff may pay more attention to patients with anaemia, implement reasonable anaemia treatment and reduce the incidence of COVID-19 sequelae and improve prognosis, acting quickly and consistently to provide early interventions, even from the beginning of admission. As plasma from donors recovered from COVID-19 has been a therapy for critical patients with COVID-19 [36, 37], we advise clinicians on considering that convalescent whole blood collected from patients who have recovered from COVID-19 that contains antibodies against SARS-CoV-2 might be a potentially effective therapeutic strategy for sequela-free survival.
This study has several limitations. First, it was a retrospective–prospective design and relied on data collected from case records, which may have missed some important information. Functional status before COVID-19 was assessed on the basis of the memory of patients and informants, whereas the functional status of patients after COVID-19 was based on chief symptoms reported and scale evaluations rather than objective assessments, which may cause a recall bias. To a certain extent, the information gathered depended on how patients understood the surveyors’ explanation of the scales due to telephone-based pattern, which may cause an information bias. Besides, although the multiple imputation was imputed based on the assumption that the data were missing at random (MAR), bias might be introduced due to this MAR assumption is technically impossible to validated. Therefore, accuracy of the findings may be compromised and the validity of the predictive factors for long-term sequelae derived remains tentative; further validation with another data set is necessary. Although the response rate in our study was relatively low, the rate was fair for a telephone-based survey, and we compared the characteristics of responders and non-responders to examine the potential of non-response bias. Strengths of this study include the large sample size and long follow-up period, which make results of the statistical analysis more reliable.
In conclusion, this follow-up study revealed that only one third COVID-19 survivors were sequela-free. Most survivors had a nearly normal ADL, moderate cardiopulmonary functional and psychosocial problems, and minor sensory abnormalities. Anaemia on admission; longer duration from symptom onset to admission, which means each additional day, and increasing of age, which means 1-year increase in age, were adverse risk factors of any long-term sequelae, while anaemia treatment was a protective factor against sequelae. Our study established the clinical evidence that anaemia at admission is a highly practical and cost-effective parameter for stratification of patient risk of long-term sequelae, paving the way for investigating possible therapeutic principles to achieve sequelae-free survival. Although COVID-19 poses a major challenge to the medical system and social community, understanding the post-COVID-19 effects on patients and associated factors may urge the academic, health and social communities to implement multidisciplinary interventions to promote the physical and psychosocial recovery of COVID-19 survivors.
Availability of data and materials
The data and materials analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ADL:
-
Activity of daily living
- AUROC:
-
Area under the receiver-operating characteristic curve
- CHI:
-
Carcinologic Handicap Index
- CI:
-
Confidence interval
- COVID-19:
-
Coronavirus disease 2019
- CRF:
-
Case report form
- ICU:
-
Intensive care unit
- IQR:
-
Interquartile range
- MAR:
-
Missing at random
- mMRC:
-
Modified Medical Research Council
- NYHA:
-
New York Heart Association
- OR:
-
Odds ratio
- SARSCoV-2:
-
Severe acute respiratory syndrome coronavirus 2
- SD:
-
Standard deviation
- SDS:
-
Self-rating Depression Scale
- TICS-M:
-
Modified Telephone Interview for Cognitive Status
References
Mahase E. Covid-19: Vaccine candidate may be more than 90% effective, interim results indicate. BMJ. 2020;371:m4347. https://doi.org/10.1136/bmj.m4347.
Logunov DY, Dolzhikova IV, Shcheblyakov DV, Tukhvatulin AI, Zubkova OV, Dzharullaeva AS, et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. 2021;397:671–81. https://doi.org/10.1016/S0140-6736(21)00234-8.
Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020;383(27):2603–15. https://doi.org/10.1056/NEJMoa2034577.
Zhu FC, Guan XH, Li YH, Huang, JY, Jiang T, Hou LH, et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet. 2020;396(10249):479–88. https://doi.org/10.1016/S0140-6736(20)31605-6.
Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397(10270):220–32. https://doi.org/10.1016/S0140-6736(20)32656-8.
China National Health Commission Chinese clinical guidance for COVID-19 pneumonia diagnosis and treatment. [cited 2023 April 28]. Available from: https://www.gov.cn/zhengce/zhengceku/2020-02/09/content_5476407.htm.
Shinar D, Gross CR, Bronstein KS, Licata-Gehr EE, Eden DT, Cabrera AR, et al. Reliability of the activities of daily living scale and its use in telephone interview. Arch Phys Med Rehabil. 1987;68(10):723–8.
Mahler DA, Wells CK. Evaluation of clinical methods for rating dyspnea. Chest. 1988;93(3):580–6. https://doi.org/10.1378/chest.93.3.580.
Cook SE, Marsiske M, McCoy KJ. The use of the Modified Telephone Interview for Cognitive Status (TICS-M) in the detection of amnestic mild cognitive impairment. J Geriatr Psychiatry Neurol. 2009;22(2):103–9. https://doi.org/10.1177/0891988708328214.
Zung WW. A self-rating depression scale. Arch Gen Psychiatry. 1965;12:63–70. https://doi.org/10.1001/archpsyc.1965.01720310065008.
Balaguer M, Percodani J, Woisard V. The Carcinologic Handicap Index (CHI): a disability self-assessment questionnaire for head and neck cancer patients. Eur Ann Otorhinolaryngol Head Neck Dis. 2017;134(6):399–403. https://doi.org/10.1016/j.anorl.2017.06.010.
Holland R, Rechel B, Stepien K, Harvey I, Brooksby I. Patients’ self-assessed functional status in heart failure by New York Heart Association class: a prognostic predictor of hospitalizations, quality of life and death. J Card Fail. 2010;16(2):150–6. https://doi.org/10.1016/j.cardfail.2009.08.010.
Xu E, Xie Y, Al-Aly Z. Long-term neurologic outcomes of COVID-19. Nat Med. 2022;28(11):2406–15. https://doi.org/10.1038/s41591-022-02001-z.
Xie Y, Xu E, Bowe B, Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022;28(3):583–90. https://doi.org/10.1038/s41591-022-01689-3.
Wei J, Yang H, Lei P, Fan B, Qiu Y, Zeng B, et al. Analysis of thin-section CT in patients with coronavirus disease (COVID-19) after hospital discharge. J Xray Sci Technol. 2020;28(3):383–9. https://doi.org/10.3233/XST-200685.
Xiong Q, Xu M, Li J, Liu Y, Zhang J, Xu Y, et al. Clinical sequelae of COVID-19 survivors in Wuhan, China: a single-centre longitudinal study. Clin Microbiol Infect. 2021;27(1):89–95. https://doi.org/10.1016/j.cmi.2020.09.023.
Madjid M, Safavi-Naeini P, Solomon SD, Vardeny O. Potential effects of coronaviruses on the cardiovascular system: a review. JAMA Cardiol. 2020;5(7):831–40. https://doi.org/10.1001/jamacardio.2020.1286.
Basso C, Leone O, Rizzo S, De Gaspari M, van der Wal AC, Aubry MC, et al. Pathological features of COVID-19-associated myocardial injury: a multicentre cardiovascular pathology study. Eur Heart J. 2020;41(39):3827–35. https://doi.org/10.1093/eurheartj/ehaa664.
Bose RJC, McCarthy JR. Direct SARS-CoV-2 infection of the heart potentiates the cardiovascular sequelae of COVID-19. Drug Discov Today. 2020;25(9):1559–60. https://doi.org/10.1016/j.drudis.2020.06.021.
Johansson M, Stahlberg M, Runold M, Nygren-Bonnier M, Nilsson J, Olshansky B, et al. Long-haul post-COVID-19 symptoms presenting as a variant of postural orthostatic tachycardia syndrome: the Swedish experience. JACC Case Rep. 2021;3(4):573–80. https://doi.org/10.1016/j.jaccas.2021.01.009.
Blitshteyn S, Whitelaw S. Postural orthostatic tachycardia syndrome (POTS) and other autonomic disorders after COVID-19 infection: a case series of 20 patients. Immunol Res. 2021;69(2):205–11. https://doi.org/10.1007/s12026-021-09185-5.
Jamal SM, Landers DB, Hollenberg SM, Turi ZG, Glotzer TV, Tancredi J, et al. Prospective evaluation of autonomic dysfunction in post-acute sequela of COVID-19. J Am Coll Cardiol. 2022;79(23):2325–30. https://doi.org/10.1016/j.jacc.2022.03.357.
Wu KK, Chan SK, Ma TM. Posttraumatic stress, anxiety, and depression in survivors of severe acute respiratory syndrome (SARS). J Trauma Stress. 2005;18(1):39–42. https://doi.org/10.1002/jts.20004.
Mahase E. Covid-19: Why are age and obesity risk factors for serious disease? BMJ. 2020;371:m4130. https://doi.org/10.1136/bmj.m4130.
Liang WH, Guan WJ, Li CC, Li YM, Liang HR, Zhao Y, et al. Clinical characteristics and outcomes of hospitalised patients with COVID-19 treated in Hubei (epicentre) and outside Hubei (non-epicentre): a nationwide analysis of China. Eur Respir J. 2020;55(6):2000562. https://doi.org/10.1183/13993003.00562-2020.
Yuan J, Kou S, Liang Y, Lan J, Li X, Zeng L, et al. Immunological evaluation on potential treatment window for hospitalized COVID-19 Patients. J Inflamm Res. 2020;13:985–93. https://doi.org/10.2147/JIR.S280331.
Marolleau B, Rias M, Delahaye A, Aillet L, Weibel M, Romanat P. [The military intensive care field hospital, a unique experience in the fight against covid-19]. Soins. 2020;65(849):22–7. French. https://doi.org/10.1016/S0038-0814(20)30239-5.
Baughman AW, Hirschberg RE, Lucas LJ, Suarez ED, Stockmann D, Johnson SH, et al. Pandemic care through collaboration: lessons from a COVID-19 field hospital. J Am Med Dir Assoc. 2020;21(11):1563–7. https://doi.org/10.1016/j.jamda.2020.09.003.
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China Lancet. 2020;395(10223):497–506. https://doi.org/10.1016/S0140-6736(20)30183-5.
Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of Coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–20. https://doi.org/10.1056/NEJMoa2002032.
Torres JP, Izquierdo G, Acuna M, Pavez D, Reyes F, Fritis A, et al. Multisystem inflammatory syndrome in children (MIS-C): report of the clinical and epidemiological characteristics of cases in Santiago de Chile during the SARS-CoV-2 pandemic. Int J Infect Dis. 2020;100:75–81. https://doi.org/10.1016/j.ijid.2020.08.062.
Wang C, Deng R, Gou L, Fu Z, Zhang X, Shao F, et al. Preliminary study to identify severe from moderate cases of COVID-19 using combined hematology parameters. Ann Transl Med. 2020;8(9):593. https://doi.org/10.21037/atm-20-3391.
Li Y, Weng Q, Huang X, Xie Y, Chen C, Qian S. Coronavirus disease 2019 (COVID-19) related cytopenia: a protocol for systematic review and meta-analysis. Medicine (Baltimore). 2020;99(36):e22033. https://doi.org/10.1097/MD.0000000000022033.
Wu Y, Huang X, Sun J, Xie T, Lei Y, Muhammad J, et al. Clinical characteristics and immune injury mechanisms in 71 patients with COVID-19. mSphere. 2020;5(4):e00362–20. https://doi.org/10.1128/mSphere.00362-20.
Chowdhury SF, Anwar S. Management of hemoglobin disorders during the COVID-19 pandemic. Front Med (Lausanne). 2020;7:306. https://doi.org/10.3389/fmed.2020.00306.
Perotti C, Del Fante C, Baldanti F, Franchini M, Percivalle E, Vecchio Nepita E, et al. Plasma from donors recovered from the new Coronavirus 2019 as therapy for critical patients with COVID-19 (COVID-19 plasma study): a multicentre study protocol. Intern Emerg Med. 2020;15(5):819–24. https://doi.org/10.1007/s11739-020-02384-2.
Janssen M, Schakel U, Djuka Fokou C, Krisam J, Stermann J, Kriegsmann K, et al. A randomized open label phase-II clinical trial with or without infusion of plasma from subjects after convalescence of SARS-CoV-2 Infection in high-risk patients with confirmed severe SARS-CoV-2 disease (RECOVER): a structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21(1):828. https://doi.org/10.1186/s13063-020-04735-y.
Acknowledgements
The authors thank physicians and health-care staff working tirelessly on the frontlines of the COVID-19 pandemic. We would like to thank Dr. Zhiyong Peng from Department of Clinical Care Medicine, Zhongnan Hospital of Wuhan University, Wuhan, China for his help in collecting patients' duration of hospital stay data.
Funding
This study was funded by grants to DSS from the National Natural Science Foundation of China (Nos.81771133, 81970995) (DSD grants), Shanghai Shenkang Hospital Development Center Founding (SHDC12017X11), Renji Hospital Clinical Innovation Foundation (PYMDT-007), Shanghai municipal Education Commission-Gaofeng Clinical Medicine Support (20191903), State Key Laboratory of Neuroscience(SKLN-201803), the National Natural Science Foundation of China (No.81701358) (DH grants), the Shanghai Municipal Health Commission Health Young Talents (2022YQ036) (CYC grants) and Renji Hospital founding (RJZZ18-019) (LLH grants).
Author information
Authors and Affiliations
Contributions
Study conception: DH, CYC, WX, WFY, DSS. Study design: WFY, DSS. Data analysis: STP, ZWF, JC, WHL, XJ, HPZ, XZ, LYZ, SHC, ZYF, XLJ, LLH, XZ, HZ, SZ, Data interpretation: DH, CYC, WX, STP, WFY, DSS. Drafting of the manuscript: DH, CYC, WX, STP, Critical revision of the manuscript for important intellectual content: ZWF, JC, WHL, XJ, HPZ, XZ, LYZ, SHC, ZYF, XLJ, LLH, XZ, HZ, SZ, WFY, DSS.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Institutional Research Ethics Committee of Renji Hospital, School of Medicine, Shanghai Jiaotong University (ethics committee approval No. KY2020-037). Oral consent was acquired from patients or from their immediate family members in cases of communication disorder or death.
Consent for publication
All authors gave their consent for publication.
Competing interests
Weifeng Yu and Diansan Su are members of the Editorial Board in Anesthesiology and Perioperative Science and recuse themselves from every editorial procedure of this submission including peer-review and academic decisions. The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
Demographic and clinical characteristics of follow-up and lost follow-up COVID-19 survivors. Table S2. Long-term outcomes. Table S3. Multivariate regression analysis in COVID-19 survivors. Table S4. Specific anemia treatment in COVID-19 survivors.
Additional file 2: Figure S1.
Optimal cut-off baseline haemoglobin level by sex subgroup. a. Density plot of baseline haemoglobin levels by sex, where vertical lines indicate the optimal cut-off points. b. Receiver-operating characteristic curves of baseline haemoglobin level by sex, where the two black dots represent the diagnostic ability corresponding to the optimal cut-off points. c. Distribution plot of the optimal cut-off points generated by bootstrapping. d. Density plot of the out-of-bag (OOB) estimates (or error) of the bootstrap procedure. OOB error is the mean prediction error in each training sample xi, using only the trees that did not have xi in their bootstrap sample.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, D., Chen, C., Xuan, W. et al. Long-term outcomes of COVID-19 survivors and an exploratory analysis of factors associated with sequela-free survival of patients treated at Leishenshan Hospital in Wuhan, China. APS 1, 35 (2023). https://doi.org/10.1007/s44254-023-00038-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44254-023-00038-y