Abstract
Background and purpose
Stroke is defined as a sudden onset of a neurological deficit caused by an acute focal injury to the central nervous system due to a vascular cause. It represents the second leading cause of death, the third leading cause of disability worldwide and the primary cause of disability in the elderly. Ischemic strokes are the most common (≈85%), the rest (≈15%) are hemorrhagic strokes, that include cerebral and subarachnoid. One of the commonly procedure used in patients with large-vessel occlusion is intra-arterial (IA) recanalization. Moderate conscious sedation (CS) and general anesthesia (GA) with intubation are the most used anesthesia techniques for patients with acute ischemic stroke undergoing endovascular recanalization.
Methods
In this retrospective observational study, we analyzed data of registry before and after we have changed our sedation protocol from remifentanil/propofol (P/R group) infusion in total intravenous anesthesia (TIVA) to intravenous infusion of dexmedetomidine alone (D group). The primary endpoint was to determine the rate of conversion from CS to GA for each group. The secondary outcomes were the incidence of complications (bradycardia, hypotension, tachycardia, hypertension).
Results and conclusion
12 patients in P/R group need the conversion from CS to GA while only 5 patients in D group. There weren't significant differences between the 2 groups respect to heart rate (HR), peripheral oxygen saturation (SpO2) during the procedure, for the good neurological outcome (mRS score < 3, p > .05) and NIHSS score after 48 h (p > .05). There were several organizational and economic advantages with the conscious sedation procedure, such as reduction of occupation time in the operating room and reduction of postoperative Intensive Care Unit (ICU) stay.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Background
Stroke is defined as a sudden onset of a neurological deficit caused by an acute focal injury to the central nervous system due to a vascular cause. It represents the second leading cause of death, the third leading cause of disability worldwide and the primary cause of disability in the elderly. Ischemic strokes are the most common (≈85%), the rest (≈15%) are hemorrhagic strokes, that include cerebral and subarachnoid. One of the most frequently used procedure in patients with large-vessel occlusion is intra-arterial (IA) recanalization. Timely recanalization of the occluded vessel with either intravenous tenecteplase (IV-tPA) thrombolysis or IA therapy is essential in preventing neuronal death and improving patient outcome.
For these procedures, general anesthesia (GA) is often the preferred method due to the perceptions of improved procedural safety and efficacy. However, conscious sedation (CS) and local anesthesia allows operators to monitor neurologic status during the procedure and avoid delays in procedure initiation. Furthermore, CS may be associated with improved hemodynamic stability compared with GA.
Nevertheless, some of the drugs mainly used in CS, such as benzodiazepine, remifentanil, or propofol, if not used properly, may elicit respiratory depression and blood pressure fluctuation on one side, and on the other inadequate sedation to immobilize the patient who may require subsequent GA and intubation. Dexmedetomidine (a highly selective centrally agonist of α2-adrenergic receptor) has been used in many diagnostic and therapeutic procedures safely and effectively for dose-dependent sedation (patients sedated with dexmedetomidine can be easily aroused to cooperate with breath holds or neurologic testing), anxiolysis and lack of respiratory depression, although clinically significant side-effects include bradycardia and hypotension.
We began to use sedation with dexmedetomidine for endovascular reperfusion therapy from January 2019. In this retrospective observational study, we analyzed data of registry before and after we have changed our sedation protocol from remifentanil/propofol infusion in total intravenous anesthesia (TIVA) to intravenous infusion of dexmedetomidine alone.
2 Methods
This retrospective study was conducted at the AORN Antonio Cardarelli Hospital of Naples. We have collected and examined the medical records of 50 patients admitted to the Stroke Unit of AORN Antonio Cardarelli Hospital of Naples during the period between January 1th 2019 and January 1th 2020. All procedures were in accordance with the Declaration of Helsinki. Patients affected by ischemic stroke and scheduled were enrolled. The Ethical Committee (CE) approval was not necessary because it was common clinical practice and a revision of data in retrospective way.
We performed a retrospective review of patients who underwent endovascular thrombectomy receiving intravenous continuous infusion of dexmedetomidine from January 1th 2019 to January 1th 2020. Afterwards we compared these ones with a group of 50 patients with similar characteristics receiving sedation protocol with remifentanil/propofol infusion from January 1th 2018 to January 1th 2019. No prior sample size was performed; however, all eligible patients were considered. Patients dexmedetomidine group (D group) received dexmedetomidine at the rate of 0.2–0.5 mcg/kg/h. During the twelve months before, patients in propofol/remifentanil group receive propofol 2 mg/kg/h and remifentanil 0.05 mcg/kg/min (P/R group).
Inclusion criteria were as follow:
-
Time of stroke onset < 6.5 h, age 40 years or older
-
National Institute of Health Stroke Scale (NIHSS) < 20
-
Occlusion in the anterior circulation identified with computed tomographic (TC) angiography or digital subtraction angiography (DSA).
Exclusion criteria were as follow:
-
Occlusion in the posterior circulation
-
Neurological recovery or recanalization before endovascular therapy
-
Modified Rankin Scale (mRS) score ≥ 4
-
Contraindications for intravenous thrombolysis.
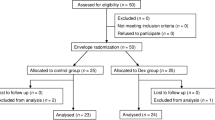
The flow chart (Fig. 1) shows in detail the procedure of patients’ selection.
68 medical records were evaluated; 12 patients did not meet the required inclusion criteria; from the remaining 56 patients, in 6 patients we were not able to collect all the data and so were excluded; 50 patients were evaluated, and all of them were included for the present study. All the data were collected at Stroke Unit of AORN Antonio Cardarelli Hospital of Naples but were pooled, analyzed, stored and available in the medical records archive of “Luigi Vanvitelli” University Hospital (Piazza Luigi Miraglia 2—Naples, Italy). The primary endpoint was to determine the rate of conversion from CS to GA for each group. The patients were put under GA if they did not reach the ideal status after the maximum dose of propofol/remifentanil. The secondary outcomes are the incidence of complications during the procedure (bradycardia, hypotension, tachycardia, hypertension).
We obtained demographics (age, sex, race), clinical (height, weight, Body Mass Index—BMI) and anesthesiological (American Society of Anesthesiologists—ASA—grade, duration of procedure and anesthesia) data. The intraoperative hemodynamic and respiratory data were collected during the procedure: heart rate (HR), mean arterial pressure (MAP), peripheral oxygen saturation (SpO2). We recorded the intraoperative data at the following time points: arrival at the operating room (T0), 5 min (T1), 10 min (T2), 20 min (T3), 30 min (T4), 60 min (T5), 90 min (T6) during the procedure. Hemodynamic adverse effects included new onset of tachycardia, bradycardia, hypertension and hypotension. Tachycardia and bradycardia were defined as a heart rate greater than 110 beats per minute (bpm) and less than 60, respectively. Hypertension and hypotension were classified as a systolic blood pressure (BP) greater than 180 mm Hg and less than 90 mm Hg, respectively. Respiratory adverse effect included onset of respiratory depression with necessity of oxygen supplementation.
2.1 Statistical analyses
Data were analyzed using a standard computer program (Excel, 2016). Results were reported as mean ± Standard Deviation (SD). The level of statistical significance was p < 0.05. This observational study is in accordance with the STROBE statement.
3 Results
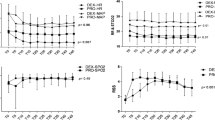
Patients enrolled for this study were similar according to age, sex, duration of surgery and ASA grade (Table 1). Mean age was 63.7 (SD ± 6.4) in D group and 62.06 years (SD ± 6.40) in P/R group. D group included 31 male and 29 female patients, while P/R group included 32 male and 28 female patients. Duration of procedure was 92.55 min (SD ± 4.07) in D group and 93.1 min (SD ± 4.01) in P/R group. 12 patients in P/R group need the conversion from CS to GA, while only 5 patients need that in D group (Fig. 2).
There weren't statistically significant differences respect to HR and SpO2 during the procedure between the 2 groups. MAP was significantly lower in the P/R group. There weren't any other hemodynamic adverse event (Fig. 3). There weren’t significant differences between the 2 groups to good neurological outcome (mRS score < 3, p > 0.05) and NIHSS score after 48 h (p > 0.05).
4 Discussion
Timely recanalization of the occluded vessel with either IV-tPA or IA therapy is essential in preventing neuronal death and improving patient outcome. Several Randomized Clinical Trials (RCTs) and an individual patient data meta-analysis have shown that thrombectomy combined with standard treatment was more effective than standard treatment alone (most often including IV thrombolysis) for severe acute ischemic stroke caused by large-vessel occlusion in the anterior circulation. Guidelines for endovascular intervention do not include posterior circulation occlusions (vertebrobasilar system) because these are the least studied. Although the posterior circulation stroke has the highest mortality, endovascular treatment is therefore warranted within an extended acceptable time window.
Despite GA remain the preferred method, CS and local anesthesia has many advantages.
A lot of studies compare these two types of anesthesiologic procedures and different pharmacological protocols:
Anesthetic gases are associated with an increased risk of cerebral hypoperfusion with poor self-regulation by inducing cerebral vasodilatation and increased ischemic injury [1,2,3]. Davis et al. in 2012 [4] demonstrated that patients with ischemic stroke undergoing endovascular recanalization had higher rates of intraprocedural hypotension with GA versus CS. The induction and awakening phases of general anesthesia are often associated with significant hemodynamic changes which could exacerbate the damage ischemic. More over the role of ketamine for analgo-sedation in the intensive care unit (ICU) is yet to be defined [5].
A post hoc analysis of the SIESTA trial suggests that peri-interventional drops BP where both sedation regimes, GA and CS, were performed according to strict protocols directed at avoiding BP extremes drops, were not associated with either early neurological improvement or long-term functional outcome [6].
Brinjikji and colleagues showed that patients receiving CS had higher rates of functional success and recanalization and reduced rates of respiratory mortality and complications than those receiving GA. They observed no difference in procedure times, recanalization time between study groups. Rates of intracranial hemorrhage or other vascular complications were also similar between the 2 groups [7].
The results of the DEFUSE 3 trial shows patients who underwent thrombectomy with CS in the extended time window experienced a higher likelihood of functional independence at 90 days, a lower NIHSS score at 24 h, and a shorter time from femoral puncture to reperfusion compared with those who had GA. This effect remained robust in institutions that only treated patients with a single anesthesia technique [8].
In the GOLIATH trial 128 subjects with anterior circulation large vessel occlusion stroke within 6 h were randomized to either GA or CS, and compared the quality of reperfusion, treatment delay, use of catheters and contrast and radiation dosage between the trial arms. This trial shows the safety and quality of endovascular therapy under either GA and CS are comparable [9].
In a Cochrane systematic review published in 2022 the objectives of the study was to assess the effects of different types of anaesthesia for endovascular interventions in people with acute ischaemic stroke. It included seven trials with 982 participants, which investigated the type of anaesthesia for endovascular treatment in large vessel occlusion in the intracranial circulation. The outcomes were assessed at different time periods, ranging from the onset of stroke symptoms to 90 days after the procedure. Therefore, all included studies reported data for early (up to 30 days) and long‐term (above 30 up to 90 days) time points. The Authors’ conclusions were the uncertainty whether general anaesthesia improves functional outcomes and time to revascularisation because the certainty of the evidence is very low for the early outcomes. However, regarding long‐term outcomes, general anaesthesia makes no difference to functional outcomes compared to non‐general anaesthesia with low‐certainty evidence. General anaesthesia did not change stroke‐related mortality when compared to non‐general anaesthesia with low‐certainty evidence. In view of the limited evidence of effect, more randomised controlled trials with a large number of participants and good protocol design with a low risk of bias should be performed to reduce the uncertainty and to aid decision‐making in the choice of anaesthesia [10].
The goal of the General Anesthesia vs Sedation for Stroke (GASS) protocol is to demonstrate that conscious sedation will be associated with a better clinical outcome measured with the mRs score 3 months after the procedure. The GASS study is the first multicentric RCT including a detalied anaesthesia protocol with a tight haemodynamic control, comparing GA and CS during endovascular treatment (EVT) and evaluating the functional outcome at 3 months [11]. The conclusions at the end of the trial was the functional outcomes 3 months after endovascular treatment for stroke were similar with general anesthesia and sedation. Our results, therefore, suggest that clinicians can use either approach [12].
Numerous studies have evaluated the use of Dexmedetomidine for CS in patients undergoing diagnostic and therapeutic procedures compared to GA.
Alfieri et al. used dexmedetomidine in the management of awake fiberoptic intubation. For its sedative, anxiolytic, analgesic and sympatholytic properties can be a useful drug during awake intubation, reducing patient discomfort, without depressing the respiratory function and having a negligible impact on the cardiovascular system [13]. Cristiano et al. have successfully used sedation with dexmedetomidine in patients undergoing transfemoral transcatheter aortic valve implantation procedures. Dexmedetomidine ensures patient immobility, an acceptable level of sedation while keeping patients spontaneous breathing and a higher stability of the hemodynamic parameters. Complications such as apnea, hypoventilation and hypoxemia did not occur [14].
Huncke et al. [15] reported the efficacy of Dexmedetomidine sedation during vascular procedures and Whalin et al. [16] demonstrated that Dexmedetomidine can be safely administered to patients undergoing endovascular reperfusion therapy.
Dexmedetomidine is a rapidly metabolized drug with a plasma half-life of approximately 2 h making it very manageable [17]. Chi et al. [18] have suggested that vasoconstriction induced by Dexmedetomidine in the normal cortex it helps redistribute blood flow to the reperfused ischemic cortex, thus helping to restore blood flow to this region. Conversely, Nakano and Okamoto [19] reported that high dose Dexmedetomidine use in intracranial hypertension and ischemic brain can cause increased damage in rats.
Dexmedetomidine, depending on the dosage used, can cause decreased heart rate and blood pressure. Therefore, we recommend using Dexmedetomidine at the lowest possible dosage in order to maintain adequate sedation during the procedure. In 2018, Kouhei Nii and colleagues evaluated the usefulness of sedating consciousness with dexmedetomidine and pentazocine during endovascular treatment for acute stroke.
Sedation with Dexmedetomidine was found to be effective during endovascular treatment of patients with acute stroke. In some patients, only low-dose Dexmedetomidine failed to adequately control pain for the duration of the procedure, however, it was with the help of pentazocine that the endovascular treatment procedure could be completed without significant complications such as respiratory depression or hemodynamic instability [20].
The optimal anesthetic practice for EVT remains to be identified. Currently, CS is often an easy first-line strategy, but GA can be considered an equal and safe alternative to conscious sedation when there is a carefully administered anesthetic that maintains strict hemodynamic control [21].
5 Conclusion
In this retrospective observational study, we analyzed data of registry before and after we have changed our sedation protocol from remifentanil/propofol infusion in total intravenous anesthesia (TIVA) to intravenous infusion of dexmedetomidine alone. MAP was significantly lower in the propofol/remifentanil group. There weren't also no one hemodynamic adverse event. There were no significant differences between the 2 groups in terms of good neurological outcome and NIHSS score after 48 h.
The procedure performed under CS, taking into consideration the organizational and economic context, has several advantages compared to the procedure performed under GA: with the same number of medical and nursing personnel, and with the same duration of the procedure, the mean occupation time of the operating room is clearly less in the first group. This is due to the increased anesthetic time required for the induction and recovery phases during general anesthesia. In addition, a reduction in the mean days of postoperative ICU stay after the procedure was observed, although this data is not considered statistically significant.
The limitation of this study is that it is a retrospective observational and not a controlled prospective trial. We need several studies and RCT in order to have strong evidence on the better anesthesia management during procedure for endovascular recanalization.
Data Availability
All data are avaible near our dept.
References
Messick JM Jr, Newberg LA, Nugent M, et al. Principles of neuroanesthesia for the nonneurosurgical patient with CNS pathophysiology. Anesth Analg. 1985;64:143–74.
Steen PA. Inhalational versus intravenous anesthesia: cerebral effects. Acta Anaesthesiol Scand Suppl. 1982;75:32–5.
Petersen KD, Landsfeldt U, Cold GE, et al. Intracranial pressure and cerebral hemodynamic in patients with cerebral tumors: a randomized prospective study of patients subjected to craniotomy in propofol-fentanyl, isoflurane-fentanyl, or sevoflurane-fentanyl anesthesia. Anesthesiology. 2003;98:329–36.
Davis MJ, Menon BK, Baghirzada LB, et al. Anesthetic management and outcome in patients during endovascular therapy for acute stroke. Anesthesiology. 2012;116:396–405.
Russo E, Santonastaso DP, Gamberini E, Circelli A, Martino C, Agnoletti V. Ketamine in Neurocritical Care. J Intensive Care Med. 2020. https://doi.org/10.1177/0885066620912719.
Schönenberger S, Uhlmann L, Ungerer M, Pfaff J, Nagel S, Klose C, Bendszus M, Wick W, Ringleb PA, Kieser M, Möhlenbruch MA, Bösel J. Association of blood pressure with short- and long-term functional outcome after stroke thrombectomy: post hoc analysis of the SIESTA Trial. Stroke. 2018;49(6):1451–6. https://doi.org/10.1161/STROKEAHA.117.019709. (Epub 2018 May 2).
Brinjikji W, Murad MH, Rabinstein AA, Cloft HJ, Lanzino G, Kallmes DF. Conscious sedation versus general anesthesia during endovascular acute ischemic stroke treatment: a systematic review and meta-analysis. AJNR Am J Neuroradiol. 2015;36(3):525–9. https://doi.org/10.3174/ajnr.A4159. (Epub 2014 Nov 13).
Powers CJ, Dornbos D, Mlynash M, Gulati D, Torbey M, Nimjee SM, Lansberg MG, Albers GW, Marks MP. Thrombectomy with Conscious Sedation Compared with General Anesthesia: A DEFUSE 3 Analysis. Am J Neuroradiol. 2019;40(6):1001–5. https://doi.org/10.3174/ajnr.A6059.
Sørensen LH, Speiser L, Karabegovic S, et al. Safety and quality of endovascular therapy under general anesthesia and conscious sedation are comparable: results from the GOLIATH trial. J Neuro Intervent Surg. 2019;11:1070–2.
Tosello R, Riera R, Tosello G, Clezar CN, Amorim JE, Vasconcelos V, Joao BB, Flumignan RL. Type of anaesthesia for acute ischaemic stroke endovascular treatment. Cochrane Database Syst Rev. 2022;7(7):013690. https://doi.org/10.1002/14651858.CD013690.pub2.
Maurice A, Ferré JC, Ronzière T, Devys JM, Subileau A, Laffon M, Laviolle B, Beloeil H. GASS Trial study protocol: a multicentre, single-blind, randomised clinical trial comparing general anaesthesia and sedation during intra-arterial treatment for stroke. BMJ Open. 2019;9(5):e024249. https://doi.org/10.1136/bmjopen-2018-024249.
Maurice A, Eugène F, Ronzière T, Devys JM, Taylor G, Subileau A, Huet O, Gherbi H, Laffon M, Esvan M, Laviolle B, Beloeil H. General Anesthesia versus Sedation, Both with Hemodynamic Control, during Intraarterial Treatment for Stroke: The GASS Randomized Trial. Anesthesiology. 2022;136(4):567–76. https://doi.org/10.1097/ALN.0000000000004142.
Alfieri A, Passavanti MB, Di Franco S, et al. Dexmedetomidine in the management of awake fiberoptic intubation. Open Anesth J. 2019;13:1. https://doi.org/10.2174/2589645801913010001.
Cristiano L, et al. Use of Dexmedetomidine in Transfemoral Transcatheter Aortic Valve Implantation (tf-TAVI) Procedures. Adv Ther. 2020;37(5):2337–43. https://doi.org/10.1007/s12325-020-01342-w. (Epub 2020 Apr 15).
Huncke TK, Adelman M, Jacobowitz G, Maldonado T, Bekker A. A prospective, randomized, placebo controlled study evaluating the efficacy of dexmedetomidine for sedation during vascular procedures. Vasc Endovascular Surg. 2010;44:257–61.
Whalin MK, Lopian S, Wyatt K, et al. Dexmedetomidine: a safe alternative to general anesthesia for endovascular stroke treatment. J Neurointerv Surg. 2014;6:270–5.
Movafegh A, Shoeibi G, Ansari M, Sadeghi M, Azimaraghi O, Aghajani Y. Naloxone infusion and post-hysterectomy morphine consumption: a doubleblind, placebo-controlled study. Acta Anaesthesiol Scand. 2012;56:1241–9.
Chi OZ, Grayson J, Barsoum S, Liu X, Dinani A, Weiss HR. Effects of dexmedetomidine on microregional O2 balance during reperfusion after focal cerebral ischemia. J Stroke Cerebrovasc Dis. 2015;24:163–70.
Nakano T, Okamoto H. Dexmedetomidine-induced cerebral hypoperfusion exacerbates ischemic brain injury in rats. J Anesth. 2009;23:378–84.
Nii K, Hanada H, Hiraoka F, Eto A, Mitsutake T, Tsutsumi M. Usefulness of consciousness sedation with dexmedetomidine and pentazocine during endovascular treatment for acute stroke. Neurol Med Chir. 2018;58(2):79–84. https://doi.org/10.2176/nmc.oa.2017-0188. (Epub 2017 Dec 19).
Rasmussen LK, Simonsen CZ, Rasmussen M. Anesthesia practice for endovascular therapy of acute ischemic stroke in Europe. Curr Opin Anaesthesiol. 2019;32(4):523–30. https://doi.org/10.1097/ACO.0000000000000746.
Author information
Authors and Affiliations
Contributions
CF, TA, SC—performed the research. PV, CF, PMC, VR—designed the research study. DPM, IV, SP, CP, PMB—analyzed the data. CF, PV—wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
All the authors declare that they don’t have any conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vincenzo, P., Francesco, C., Crescenzo, S. et al. Dexmedetomidine infusion in the management of acute ischemic stroke undergoing endovascular recanalization: a retrospective observational study. Discov Health Systems 2, 10 (2023). https://doi.org/10.1007/s44250-023-00025-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44250-023-00025-x