Abstract
Grape molasses is a natural sweetener widely consumed in Lebanon and Eastern Mediterranean countries. However, the lack of national standards that control its production often leads to inconsistent products and complicates international exports. Thirty grape molasses samples were collected from different Lebanese regions, and their physicochemical, microbiological, and sensory properties were investigated using a multivariate approach. Significant disparities were detected among samples obtained using different processes and from different geographical origins. The samples from the Bekaa region, manufactured with semi-industrial heat exchangers, were the most appreciated by the panelists due to their high dry matter content (80.41%), Brix degree (75°Brix), low water activity (0.55) and low titratable acidity (0.30%). Microbial contamination with osmophilic microorganisms was detected in several samples, highlighting poor hygiene practices and factory sanitation, in addition to post-contamination during the packaging and whipping steps. In summary, the key parameters that affect grape molasses quality are geographical origin, grape variety and ripening stage, soil and climatic factors, and manufacturing process, particularly juice concentration technique/time. These factors should be considered for future optimization and standardization of production processes and for monitoring and homogenizing the quality of this product.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Molasses is defined as a byproduct of sucrose production and is obtained from beets or sugarcane [1, 2]. However, molasses around the Mediterranean basin is produced by concentrating fruit juices containing high amounts of sugar, such as grapes, carob, apple, mulberry, date, apricot, watermelon, and plum [3,4,5]. Grape molasses is a dark, soft, and viscous liquid produced by grape juice at concentration up to 65–80° Brix without the addition of sugar or other food additives [6, 7]. It is a natural product widely consumed in Mediterranean basin countries, known as Pekmez in Turkey [8], Petimezi in Greece [9], Dibs El Enab in Lebanon [10] and in Middle East countries [11].
Grape molasses represents a good source of energy due to its high carbohydrate content (50–80%), mainly glucose and fructose, which can easily transfer into the bloodstream without digestion [5, 12]. The average energy value of grape molasses is 293 kcal/100 g [13]. Therefore, it is highly recommended for babies, children, athletes and situations that require immediate energy [12, 14]. This traditional product is considered a functional food due to its richness in organic acids and essential minerals such as iron, phosphorus, calcium, and potassium [13, 15]. Grape molasses is also rich in phenolic compounds such as caffeic acid, ellagic acid, gallic acid, p-coumaric acid, and rutin hydrate [13], which have health-promoting effects. Recent studies have shown that the consumption of grapes or derived products reduces the risk of chronic diseases such as certain types of cancer and cardiovascular diseases [16, 17].
In Lebanon, viticulture represents an important economic activity, and vineyards are well adapted to local agroclimatic conditions [18]. Grapes are consumed as fresh table grapes or are transformed to produce wine, Arak, verjuice, or grape molasses. The latter, locally known as Dibs El Enab, is an important component of Lebanese culinary heritage. It represents a potential alternative to white sugar, which is known for its negative impact on human health. In fact, added sugars are harmful to human health and increase the risk of cardiovascular diseases, obesity, diabetes, cognitive decline and some cancers [19].
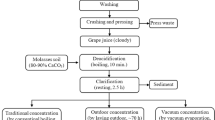
Lebanese grape molasses is often produced from local varieties characterized by a high sugar content and low acidity. These varieties include Obeidi, Mirweih, Mikseis, Maghdouchi, Saraani, and Shamouti [10, 20]. Briefly, the grapes are harvested, washed, and manually pressed to extract the juice. Marl powder (a mixture of calcium carbonate and clay), known as “houwara” in Arabic, is added to the juice (at a ratio of 1/50) to neutralize its acidity and clarify the final product. The mixture is left to stand for 6–8 h in order to complete the clarification step. The juice is then separated from the sediments, and placed in large tanks for boiling for 2–3 h. The juice is traditionally concentrated by thermal evaporation, which may affect the sensory and physicochemical quality of grape juices [6]. Other concentration techniques were reported in the literature, going from semi-industrial processes using steam to a more sophisticated technique using evaporation under a vacuum [21].
Several studies have shown that grape molasses has the potential to replace white sugar and chocolate in food formulations. It can be used as a natural sweetener, colorant, and flavoring agent to improve the nutritional and functional properties of frozen dairy desserts [11], ice cream pastries, compotes, and desserts [11, 22]. For instance, a study conducted by Mehrabi et al. [23] revealed that low-calorie cakes, prepared by replacing sugar with grape molasses, have the highest sensory scores in terms of color, texture, taste, and aroma.
In Lebanon, grape molasses is still manufactured using old and ancestral methods. Moreover, no scientific research has highlighted the diversity of molasses production or the variation in its nutritional, organoleptic or microbiological properties throughout the Lebanese regions.
Therefore, this study aims to characterize this traditional product in terms of its physicochemical, microbiological, sensory, and antioxidant properties and to investigate the effect of the concentration technique (traditional and semi-industrial) on these characteristics. This is a prerequisite step for optimizing grape molasses production in Lebanon, obtaining a consistent and stable product, and promoting it as a natural sweetener.
2 Material and methods
2.1 Survey and sample collection
To gather information on different processing techniques and raw materials used in grape molasses production and shed the light on the similarities or discrepancies among producers, a survey has been carried out among 30 artisanal and semi-industrial producers located across various regions in Lebanon. The producers’ selection was made referring to the statistics of the Lebanese Ministry of Agriculture [24] for viticulture in Lebanon: Beqaa (46%), Baalbeck-Hermel (23%), Mount-Liban (13%), North (13%), Akkar (2%), South and Nabatiyeh (3%). Thirty (30) grape molasses samples were collected respecting this regional distribution of vineyards in Lebanon [24], between August and September 2019. The samples were stored in glass jars at 4°C until analysis.
2.2 Physicochemical analyses
The determination of the total contents of dry matter (DM), ash (A), protein (P) and fat (F) was carried out according to the AOAC standard methods [25]. All the chemicals were of analytical reagent grade and were purchased from Sigma‒Aldrich (Darmstadt, Germany). All the determinations were made in triplicate. The water activity (aw) was measured at 25 °C using a water activity meter (Novasina AG, Lacen, Switzerland). The total soluble solids (Brix) were determined at 20 °C using a digital refractometer (KERN & SOHN GmbH, Balingen, Germany). pH measurements were made at 20 °C using a digital pH meter (SI Analytics GmbH, Mainz, Germany). The titratable acidity (g tartaric acid/100 g) was calculated by the titration of 5 g of molasses diluted using distilled water, with NaOH 0.1 N, to an end point of pH 8.1 [13].
To determine the glucose (Glu), fructose (Fru), and sucrose (Sac) content of grape molasses, the Sucrose/D-Glucose/D-Fructose enzyme kit (R-Biopharm AG, Darmstadt, Germany) was used. The samples were discolored with activated charcoal (Sigma‒Aldrich, Darmstadt, Germany) before the sugar content was measured. The absorbance of the blanks and different samples was measured at a wavelength of 340 nm. The results are expressed in g/100 mL molasses solution.
The total phenolic content (TPC) was determined using Folin–Ciocalteu reagent and is expressed in mg of gallic acid equivalent (GAE)/g of dry sample. Gallic acid standards ranging from 0 to 500 mg/L were prepared. The readings were performed at 760 nm [6]. The anthocyanin content (An) was measured as a function of the discoloration by SO2. The optical density was measured after 20 min at 520 nm by referring to a tube that contained distilled water [26]. The antioxidant activity of the grape molasses mixture was determined by the DPPH method described by Tounsi et al. [27] with slight adjustments. The absorbance was measured at 517 nm. The antioxidant activity (AA) was expressed as a percentage inhibition of the DPPH radical. From the Trolox calibration curve, the Trolox equivalent antioxidant capacity (TEAC) was calculated and expressed as μmol Trolox equivalents (μmol TE/g). All absorbance readings were performed on a UV‒Vis spectrophotometer (Thermo Fisher Scientific, Waltham, USA).
2.3 Microbiological analyses
The microbiological analysis of grape molasses included the determination of total aerobic bacteria (totgerm), yeast and mold (Y & M) counts according to APHA methods [28]. The samples (10 g) were dispersed in 100 mL of peptone water solution and homogenized using a Stomacher 400 Circulator Lab blender (Thomas Scientific, Swedesboro, USA). Additionally, tenfold serial dilutions (up to 10–6) were prepared, and 0.1 mL of the different dilutions was plated onto the appropriate media. Incubation was carried out at 35 °C for 48 h using plate count agar for total aerobic bacteria. Sabouraud dextrose agar was used to determine the presence of yeasts and molds at 25 °C for 72 h. After the plates were incubated, 3 to 300 colonies were counted, and the results are expressed in CFU/g.
2.4 Sensory analysis
Forty (40) panelists, aged between 18 and 60, and familiar with grape molasses consumption and use, were selected for sensory analysis. 17 panelists were females and 23 were males. Samples were coded with three random-digit numbers and presented in balanced order. The panelists evaluated the color (Col), texture (Tex), odor (Od), sweet taste (Fla), and overall acceptability (Acc) attributes on a 9-point hedonic scale (1: extreme dislike; 5 = neither like nor dislike; 9: extremely like). Six (6) sessions were held over 3 days, and samples were evaluated based on a rate of 5 samples/session to avoid saturation issues. Coded samples were presented one-by-one in plastic cups with small plastic spoons. The panelists were provided with room temperature potable water to rinse their mouths between samples.
2.5 Multivariate assessment: principal component analysis (PCA)
PCA is an unsupervised exploratory method frequently used in, among other fields, data compression, modeling, outlier detection, variable selection, classification, and visualization. Briefly, PCA produces a series of orthogonal axes (principal components (PCs)), a linear combination of the original variables, by decomposing the initial data matrix X into a matrix product T.PT (the “T” in PT means the “transposed matrix”). The T matrix is commonly referred to as the “scores” matrix. The graphical display of these scores can often reveal meaningful patterns or clustering. The P matrix, the “loadings”, shows which variables are responsible for the patterns found in the scores. In this study, the dataset consisted of 30 molasses samples, described by 22 physicochemical, microbiological and sensory variables. The data were grouped in a matrix X (30 × 22). PCA was performed on X after column standardization (mean centering of each variable and dividing the resulting values by the column’s standard deviation). Various preliminary tests, such as the Kaiser, Meyer and Olkin index (KMO) and Bartlett tests, were performed to examine the appropriateness of the data for PCA. Data analysis was performed using MATLAB version R2016a (The MathWorks, Natick, USA).
3 Results and discussion
3.1 Survey results
To the best of our knowledge, this is the first study to explore the production of Lebanese grape molasses and to evaluate the diversity of its characteristics. Therefore, it was crucial to analyze the survey responses to assess the variations in the manufacturing methods used between different Lebanese regions.
According to the results of the survey, most of the production (85.7%) is performed in small-scale family businesses or by households, using traditional and artisanal techniques to crush grapes using human foot or hand or using a traditional/electrical grape press, and to boil the must using wood or gas heaters. Production occurs between September and October, depending on the maturity index of the grapes and climatic factors. The grape variety and cultivar, growing conditions, and soil composition are important factors to consider when evaluating grape molasses [6].
Grape molasses is prepared from white grapes (51.9%), red grapes (11.1%), or a mixture of both (37.0%). The most common grape cultivars (Obeidi and Merweh) were used separately or in mixture, due to their high sugar content. When in mixtures, other local cultivars were added (Maghdouchi, Tfeifihi, Beitamouni, Chamouti and Sareaiby). Using these grape combinations accounted for 66.7%.
Moreover, Merweh, Obeidi and Maghdouchi are the main varieties used in Mount Lebanon and North, while Obeidi, Maghdouchi, Tfeifihi and Beitamouni are preferred in Baalbeck-Hermel, with a significant difference between regions (p < 0.05). Nevertheless, producers in the Bekaa valley, known as the main area of viticulture in Lebanon with many endogenous varieties [18], use Obeidi, Maghdouchi, Tfeifihi, Chamouti, Sareaiby and Kassoufi to produce grape molasses.
Marl powder with high calcium carbonate content is either added to grape bunches (68%) or to grape juice (32%) at a ratio of 1/50 to 1/25 w/w to clarify it and reduce its acidity [6] caused by natural tartaric and malic acids. Most producers (57%) allow the juice to settle overnight (8–12 h) before it is clarified and concentrated. However, 43% of producers boil unclarified grape juice (2–4 h) and allow it to settle for 4–6 h prior to its concentration. This optional step facilitates juice clarification and inactivates the enzymes responsible for undesirable color changes. Moreover, it prevents spontaneous fermentation during the settling stage [29].
The desired consistency of grape molasses is reached through the concentration step, which takes place for 4–8 h in open vessels (43%) using the traditional method (wood/gas heating). Using semi-industrial heat exchangers (57%), concentration can be achieved in 1–1.5 h, thus preventing any potential decomposition of grape juice components due to excessive and long-term heating [6]. This technique is widespread in Beqaa and South Lebanon (p < 0.05), while heating is still traditional in Mount Lebanon and North Lebanon.
After cooling, 33% of the producers stir and beat liquid molasses manually using a fig branch, wooden spoon, or an electric mixer. Whipping and incorporating air into molasses results in a creamy, spreadable texture with a lighter color [30]. Therefore, Lebanese grape molasses can be classified according to its consistency into two categories: liquid or semi-liquid molasses (called in Arabic “Dibs seyel”) and solid molasses (called “Dibs matrouf” or “Dibs makhbout”), which is yellow or light brown in color.
Based on the abovementioned information and results, a general flow diagram for Lebanese grape molasses production was established (Fig. 1).
3.2 Physicochemical and microbiological properties
A summary of the minimum, maximum, mean, and standard deviation values of the main physicochemical characteristics of the grape molasses is presented in Table 1. Total dry matter varied between 53.05 and 87.42%, with significant differences between the Lebanese regions (p < 0.05). The total sugar content varied between 51.60 and 84.67° Brix. According to Aktop et al. [12], grape molasses (pekmez) has a high sugar content between 50 and 80%. These differences can be attributed to the grape cultivar and ripening stage, differences in regions and altitudes, in addition to the processing technique and conditions [31].
Glucose (20.65%) and fructose (31.61%) were the most abundant carbohydrates found in Lebanese grape molasses. Türkben et al. [13] reported that the glucose and fructose contents of grape pekmez ranged from 27.57 to 41.11 g/100 g and from 22.34 to 34.69 g/100 g, respectively. Thus, grape molasses can be recommended as a natural sweetener for babies, children and athletes due to its natural richness in glucose and fructose, which are easily assimilated into the bloodstream without digestion [32]. Sucrose was detected in a very limited number of samples as shown by the maximum level of 5.74%. Adulteration was suspected in this case, probably to overcome some sensory defects that were attributed to these samples.
The protein content ranged from ND to 0.80%. These findings are in agreement with those obtained by Özcan et al. [3] for pekmez, in which the protein content ranged between 0.30 and 0.44%. During juice concentration, the protein content decreases due to the involvement of proteins in non-enzymatic browning reactions (Maillard reaction), which occur between amino acids and reducing sugars present in grape juice [27].
The average ash content of the grape molasses mixture was 4.44%, which was strongly related to the addition of marl powder during the clarification stage. In fact, grape molasses is rich in vital minerals such as potassium, calcium, phosphorus, magnesium, sodium, and iron [13, 33, 34]. Similar results have been reported for pekmez (3.72%) [34], where pekmez earth (80% CaCO3) is used to reduce acidity, eliminate pectin, and clarify must [29].
When the pH varied between 3.35 and 5.80, the total acidity ranged from 0.14 to 0.83 g of tartric acid/100 g. These results are consistent with those of Türkben et al. [13] and Aliyazicioglu et al. [33], where the pH of grape pekmez ranged from 3.59 to 5.23 and from 4.9 to 5.6, respectively. The addition of marl powder to the production of grape pekmez decreases the titratable acidity to less than 1% by precipitating naturally existing tartaric and malic acids as calcium tartarate and calcium malate [29, 30].
The average TPC was 55.04 mg GAE/g dry sample. Lower TPC values were found for sugarcane molasses (13.91–19.40 mg GAE/g) [35] and grape juice concentrate [36]. The disparities among samples produced in different regions and reflected by large standard deviation values can be attributed to many factors. In fact, polyphenol concentrations may vary depending on the grape variety, ripeness, culture conditions, and technology applied to obtain grape juice, among which contact time with grape skins rich in phenols [37]. Large losses of phenolic compounds are common during the concentration of grape juice, in addition to the deacidification and clarification treatments [21].
The total anthocyanin content was approximately 46.11 mg/L, with significant regional variation (p < 0.05) mainly related to a combination of factors: grape variety, cultivation conditions, ripening degree, environmental factors, and differences in processing techniques (concentrations) [36]. For instance, grape molasses samples that were exposed to long-term heat application via traditional concentration methods exhibited a low anthocyanin content. Özcan et al. [3] obtained similar anthocyanin values (54.25–64.75 mg/L) for pekmez. Anthocyanins are mostly located in the berry skin of the varieties used for grape juice processing. They are either degraded in the pressing and concentration steps or by subsequent clarification and filtration treatments [36].
The antioxidant activities of the grape molasses samples ranged from 0.38 to 9.81 μmol TE/g. These values are lower than those obtained by Türkben et al. [13] for pekmez and by [35] for sugarcane molasses. Although the grape molasses samples were rich in phenolic compounds, their low antioxidant activity may be attributed to different factors. In fact, the chemical structure of phenolic compounds is influenced by various conditions, such as enzymatic reactions, changes in pH, matrix effects, and temperature [38]. Moreover, phenolic compounds may be exposed to structural decomposition during the long concentration process of grape molasses and long-term application of heat [6], leading to a decrease in their antioxidant activity. Several studies have shown that heating accelerates initiation reactions and decreases antioxidant activity with increasing cooking/heating temperatures [39, 40]. However, other studies [41] have shown an increase in the antioxidant activity of grape extract after thermal treatment due to the formation of oligomers from free polyphenols.
Changes in the phenolic composition and antioxidant activity are highly complex and depend on the structure, food matrix and cooking/heating method used [42]. Moreover, the antioxidant activity of grapes is influenced not only by heat treatment but also by other factors, such as growing conditions, environmental factors, grape genotype, soil type and postharvest treatments, which may affect and alter phenolic compound levels and antioxidant activity in grape molasses [6].
Microbiological analysis of grape molasses showed that the total aerobic germ count varied between 5 CFU/g and 1.2 × 103 CFU/g in 46% of the samples. Yeasts and molds were detected in ten samples within the following range: 5 CFU/g and 6.55 × 102 CFU/g. Despite the low aw (0.45–0.78) of grape molasses, contamination with several osmophilic microorganisms can occur during the packaging and whipping steps, especially if hygiene practices are not well respected. Similarly, in previous studies [43, 44], osmophilic and osmotolerant yeasts were isolated from grape juice concentrates. These concentrates have low water availability, normally leading to a low risk of spoilage by microorganisms [43, 44]. Nevertheless, microorganisms such as osmophilic yeasts are able to grow due to their extreme osmotolerance and vigorous fermentation of hexose sugars [45]. They are consequently considered the most common spoilage agents in sugar-rich foods [44, 46]. Contamination and spoilage may be attributed to raw material contamination, poor hygiene practices, and poor factory sanitation, which can lead to post-contamination after heat treatment [47].
3.3 Multivariate analysis of grape molasses attributes
Principal component analysis (PCA) was performed on the data matrix X (30 × 22), where 30 grape molasses samples were grouped and characterized by 22 physicochemical, microbiological, and sensory attributes. The indexing of samples was performed according to their geographical origin to highlight potential discriminations underlined by the 22 tested parameters. In fact, geographical origin is not an exclusive indicator of sampling location but underlies additional information related to grape varieties, climatic conditions and manufacturing processes. After data standardization, the Kaiser–Meyer–Olkin (KMO) and Bartlett’s tests were found to be adequate, proving the appropriateness of the data matrix for PCA.
PCA revealed that the first component (PC1) enclosed about 44% of the total variance, while the second component (PC2) accounted for 13%, explaining a total of 57% of the total variance. These two components were therefore considered significant for further interpretation. In Fig. 2, loadings (column chart) and scores (scatter plot) are presented. The loadings represent the contributions of the 22 variables to the different components, while the score plot illustrates the potential geographical discrimination within the analyzed samples, colored based on the sampling region.
According to their positions on the score plot (Fig. 2), the 30 molasses samples were found to be mainly discriminated according to the PC1 direction, where three clusters could be distinguished: the first one on the positive side of PC1, which included all the samples from the Bekaa region, two from Mount Lebanon and one from South/Nabatiyeh. A second cluster can be highlighted on the negative side of PC1, enclosing molasses sampled from the Baalbeck/Hermel region. The remaining samples, mainly from the Mount Lebanon and North/Akkar regions, form, to a certain extent, a cluster toward the center of the plot.
This difference following the PC1 axis is clearly related to the geographical distribution of the grape molasses samples, where two main regions can be highlighted: Bekaa and Baalbeck/Hermel. To further understand this clustering, the contribution of loadings on PC1 was studied. The samples from the Bekaa region were mainly affected by high positive loadings for sensory attributes (Col, Tex, Od, Fla, and Acc), which indicated high sensorial acceptability of these products. This high acceptance level can be correlated with the remaining parameters contributing to PC1. In fact, these samples had higher dry matter content (DM), Brix and pH, indicators of high viscosity, sweeter taste, low acidity, and lower water content (W and Aw), titratable acidity (TA) and fat content (F), which all account for an overall liking score for grape molasses. The balance between sweetness and acidity (°Brix/acid ratio) is a basic precept in the judgment of fruit quality and represents a good predictor of sensory attributes and consumers’ overall acceptability [48]. In fact, molasses concentration is achieved in the Bekaa region within 1–1.5 h using semi-industrial heat exchangers, which may be responsible for preserving the product’s sensory attributes and increasing consumer acceptability. In addition to the heat treatment method and duration, the properties of grape molasses can also be influenced by soil and climatic conditions, grape variety, natural sugar content and ripening stage [6, 49].
On the other hand, samples from the Baalbeck/Hermel region were less appreciated by consumers (negative scores on PC1) because of their high water content (W and Aw) and high titratable acidity (TA). These differences could be attributed mainly to the lower quantity of marl powder added to clarify and neutralize the grape juice, in addition to the short and traditional concentration leading to a more fluid product. Finally, samples from Mount Lebanon and North Lebanon have average properties and are less influenced by the parameters in question, given their position in the center of the score graph.
In addition to the contribution of PC1, some samples were scattered along the PC2 axis, particularly within the Bekaa cluster. According to the PC2 loadings, this dispersion can be attributed to the occurrence of germs (Totgerm) and yeasts and molds (Y&Ms) in the corresponding samples. The high loadings for these variables are associated with high fructose (Fru), glucose (Glu) and protein (P) levels. It is clear that using modern concentration techniques in the Bekaa region does not always guarantee the hygienic quality of grape molasses. Therefore, post-concentration steps such as filling and packaging should be carefully monitored to avoid contamination risk that can occur due to poor factory sanitation and low hygienic practices.
4 Conclusion
This study explored, for the first time, the production of Lebanese grape molasses and evaluated the diversity of its characteristics and manufacturing processes. The results revealed that grape molasses quality is attributed not only to geographical origin but also to grape variety and ripeness, soil and climatic factors, and manufacturing processes, particularly concentration technique/time. Grape molasses produced using a heat exchanger had the highest sensory acceptability. Nevertheless, process optimization and good factory sanitation are vital for improving the food safety and quality of this nutritious product at both the traditional and industrial levels.
Lebanese grape molasses may be considered a potential healthy substitute for white sugar due to its naturally high carbohydrate and ash contents. However, the lack of a legislative framework and national standards may compromise product quality and lead to inconsistent products, which constitute a constraint to international exporting. This work can help setting the benchmark for grape molasses characterization and valorization, enabling further technological and legislative endeavors.
Data availability
The data will be available upon request to the corresponding author.
References
El Asri O, Farag MA. The potential of molasses from different dietary sources in industrial applications: a source of functional compounds and health attributes, a comprehensive review. Food Biosci. 2023;56: 103263. https://doi.org/10.1016/j.fbio.2023.103263.
Palmonari A, Cavallini D, Sniffen CJ, Fernandes L, Holder P, Fagioli L, et al. Short communication: characterization of molasses chemical composition. J Dairy Sci. 2020;103:6244–9. https://doi.org/10.3168/jds.2019-17644.
Özcan MM, Alpar Ş, AL Juhaimi F. The effect of boiling on qualitative properties of grape juice produced by the traditional method. J Food Sci Technol. 2015;52:5546–56. https://doi.org/10.1007/s13197-014-1628-8.
Tounsi L, Ghazala I, Kechaou N. Physicochemical and phytochemical properties of Tunisian carob molasses. Food Meas. 2020;14:20–30. https://doi.org/10.1007/s11694-019-00263-9.
Yücel P, Güçlü H, Mert Y, Yalçın F, Ocak SB. Detection of adulteration using statistical methods over carbon isotope ratios in carob, grape, fig and mulberry pekmez. J Food Compos Anal. 2023;115: 104979. https://doi.org/10.1016/j.jfca.2022.104979.
Helvacıoğlu S, Charehsaz M, Güzelmeriç E, Acar ET, Yeşilada E, Aydın A. Comparatively investigation of grape molasses produced by conventional and industrial techniques. Marmara Pharm J. 2018. https://doi.org/10.12991/mpj.2018.39.
Ghasemi-Varnamkhasti M, Mishra P, Ahmadpour-Samani M, Naderi-Boldaji M, Ghanbarian D, Tohidi M, et al. Rapid detection of grape syrup adulteration with an array of metal oxide sensors and chemometrics. Engin Agric Environ Food. 2019;12:351–9. https://doi.org/10.1016/j.eaef.2019.04.002.
Akan LS. Production and characteristics of a traditional food: molasses (pekmez). Food Sci Nutr Stud. 2018;2:25. https://doi.org/10.22158/fsns.v2n2p25.
Varzakas T, Labropoulos A. Other sweeteners. sweeteners: nutritional aspects, applications and production technology. T. Varzakas, A. Labropoulos, S. Anestis (Eds.) CRC Press: Boca Raton. 2012: 175–208.
Zurayk R, Rahman SA. Slow food Beirut, slow food foundation for biodiversity (florence i from `akkar to `amel: Lebanon’s slow food trail. Beirut: Bslow food foundation for biodiversity; 2008.
Soukoulis C, Tzia C. Grape, raisin and sugarcane molasses as potential partial sucrose substitutes in chocolate ice cream: a feasibility study. Int Dairy J. 2018;76:18–29. https://doi.org/10.1016/j.idairyj.2017.08.004.
Aktop S, Şanlıbaba P, Güçer Y. Grape-based traditional foods produced in Turkey. Ital J Food Sci. 2023;35:55–74. https://doi.org/10.15586/ijfs.v35i3.2339.
Türkben C, Suna S, İzli G, Uylaşer V, Demir C. Physical and chemical properties of pekmez (molasses) produced with different grape cultivars. Tarım Bilimleri Dergisi. 2016;22:339–48.
Heshmati A, Ghadimi S, Ranjbar A, Khaneghah AM. Changes in aflatoxins content during processing of pekmez as a traditional product of grape. LWT. 2019;103:178–85. https://doi.org/10.1016/j.lwt.2019.01.001.
Ozmen D, Yildirim RM, Bursa K, Kian-pour N, Toker OS, Palabiyik I, et al. Improvement of spreadability of grape molasses with induced crystallization for production of creamed molasses as a novel product. Int J Gastron Food Sci. 2023;31: 100628. https://doi.org/10.1016/j.ijgfs.2022.100628.
Almatroodi SA, Almatroudi A, Alsahli MA, Rahmani AH. Grapes and their bioactive compounds: role in health management through modulating various biological activities. Pharmacogn J. 2020;12:1455–62. https://doi.org/10.5530/PJ.2020.12.200.
Karami S, Rahimi M, Babaei A. An overview on the antioxidant, anti-inflammatory, antimicrobial and anti-cancer activity of grape extract. Biomed Res Clin Prac. 2018. https://doi.org/10.1576/BRCP.1000162.
Chalak L, Touma S, Rahme S, Azzi R, Guiberteau F, Touma J-A. Assessment of the Lebanese grapevine germplasm reveals a substantial diversity and a high potential for selection. BIO Web of Conferences. 2016;7:01020. https://doi.org/10.1051/bioconf/20160701020.
Rippe JM, Angelopoulos TJ. Relationship between added sugars consumption and chronic disease risk factors: current understanding. Nutrients. 2016. https://doi.org/10.3390/nu8110697.
Massaad M. Mouneh: preserving foods for the Lebanese pantry. Barbara massaad; 2010. Publisher: Interlink Books; pages: 400-402
Korkmaz A. The effects of different concentration methods on the chemical composition, functional and sensory attributes of molasses produced from grape (vitis vinifera L.) juice. Food Meas. 2023;17:2016–28. https://doi.org/10.1007/s11694-023-01808-9.
Gökşen G, Ekiz HI. Pasting and gel texture properties of starch-molasses combinations. Food Sci Technol. 2019;39:93–102. https://doi.org/10.1590/fst.27817.
Mehrabi S, Koushki M, Azizi MH. Effect of grape syrup as a replacement for sugar on the chemical and sensory properties of sponge cake. Curr Res Nutr Food Sci J. 2017;5:126–36.
Lebanese Ministry of Agriculture, FAO. Resultats Globaux du Module de Base du Recensement de l’Agriculture 2010 2012. p. 119–121
AOAC. Association of official analytical chemists. official methods of analysis of AOAC international. 16th ed. Cunniff: Washington, DC; 1995.
El Darra N, Rajha HN, Saleh F, Al-Oweini R, Maroun RG, Louka N. Food fraud detection in commercial pomegranate molasses syrups by UV–VIS spectroscopy, ATR-FTIR spectroscopy and HPLC methods. Food Control. 2017;78:132–7. https://doi.org/10.1016/j.foodcont.2017.02.043.
Tounsi L, Karra S, Kechaou H, Kechaou N. Processing, physico-chemical and functional properties of carob molasses and powders. Journal of Food Measurement and Characterization. 2017;11:1440–8. https://doi.org/10.1007/s11694-017-9523-4.
Salfinger Y, Tortorello ML. Compendium of methods for the microbiological examination of foods. Am Publ Health Assoc. 2013. https://doi.org/10.2105/MBEF.0222.
Karababa E, Develi IN. Pekmez: a traditional concentrated fruit product. Food Rev Intl. 2005;21:357–66. https://doi.org/10.1080/87559120500222714.
Batu A. Production of liquid and white solid pekmez in Turkey. J Food Qual. 2005;28:417–27. https://doi.org/10.1111/j.1745-4557.2005.00045.x.
Unal MS. Effects of altitude on phenological periods with some quality features in raisins and molasses. Erwerbs Obstbau. 2023;65:2135–42. https://doi.org/10.1007/s10341-023-00980-y.
Toker OS, Dogan M, Ersöz NB, Yilmaz MT. Optimization of the content of 5-hydroxymethylfurfural (HMF) formed in some molasses types: HPLC-DAD analysis to determine effect of different storage time and temperature levels. Ind Crops Prod. 2013;50:137–44. https://doi.org/10.1016/j.indcrop.2013.05.030.
Aliyazicioglu R, Kolayli S, Kara M, Yildiz O, Sarikaya AO, Cengiz S, et al. Determination of chemical, physical and biological characteristics of some pekmez (molasses) from Turkey. Asian J Chem. 2009;21:9.
Si̇msek A, Artik N. Değişik meyvelerden üretilen pekmezlerin bileşim unsurlari üzerine araştirma. Gıda. 2002;27(6):459–467.
Molina-Cortés A, Sánchez-Motta T, Tobar-Tosse F, Quimbaya M. Spectrophotometric estimation of total phenolic content and antioxidant capacity of molasses and vinasses generated from the sugarcane industry. Waste Biomass Valor. 2020;11:3453–63. https://doi.org/10.1007/s12649-019-00690-1.
Capanoglu E, de Vos RCH, Hall RD, Boyacioglu D, Beekwilder J. Changes in polyphenol content during production of grape juice concentrate. Food Chem. 2013;139:521–6. https://doi.org/10.1016/j.foodchem.2013.01.023.
Moreno-Montoro M, Olalla-Herrera M, Gimenez-Martinez R, Navarro-Alarcon M, Rufián-Henares JA. Phenolic compounds and antioxidant activity of Spanish commercial grape juices. J Food Compos Anal. 2015;38:19–26. https://doi.org/10.1016/j.jfca.2014.10.001.
Celep E, Charehsaz M, Akyüz S, Acar ET, Yesilada E. Effect of in vitro gastrointestinal digestion on the bioavailability of phenolic components and the antioxidant potentials of some Turkish fruit wines. Food Res Int. 2015;78:209–15. https://doi.org/10.1016/j.foodres.2015.10.009.
Chen YT, Lin KW. Effects of heating temperature on the total phenolic compound, antioxidative ability and the stability of dioscorin of various yam cultivars. Food Chem. 2007. https://doi.org/10.1016/j.foodchem.2006.02.045.
Réblová Z. Effect of temperature on the antioxidant activity of phenolic acids. Czech J Food Sci. 2012;30:171–5. https://doi.org/10.17221/57/2011-CJFS.
Pinelo M, Rubilar M, Sineiro J, Nuñez MJ. A thermal treatment to increase the antioxidant capacity of natural phenols: catechin, resveratrol and grape extract cases. Eur Food Res Technol. 2005;221:284–90.
Minatel IO, Borges CV, Ferreira MI, Gomez HAG, Lima C-YOC and GPP. Phenolic compounds: functional properties, impact of processing and bioavailability. In: Marcos Soto-Hernndez, Mariana Palma-Tenango, Maria del Rosario Garcia-Mateos (Eds.) Phenolic compounds—biological activity. 2017. p. 1–24; https://doi.org/10.5772/66368
Combina M, Daguerre C, Massera A, Mercado L, Sturm ME, Ganga A, et al. Yeast identification in grape juice concentrates from Argentina. Lett Appl Microbiol. 2008;46:192–7. https://doi.org/10.1111/j.1472-765X.2007.02291.x.
Rojo MC, Torres Palazzolo C, Cuello R, González M, Guevara F, Ponsone ML, et al. Incidence of osmophilic yeasts and Zygosaccharomyces rouxii during the production of concentrate grape juices. Food Microbiol. 2017;64:7–14. https://doi.org/10.1016/j.fm.2016.11.017.
Siavoshi F, Sahraee M, Heydari S, Sarrafnejad A, Saniee P, Tavakolian A, et al. Sugar-rich foods carry osmotolerant yeasts with intracellular Helicobacter pylori and staphylococcus spp. Middle East J Dig Dis. 2020;12:182–93. https://doi.org/10.34172/mejdd.2020.181.
Helvacioglu S, Charehsaz M, Ozan G, Guzelmeric E, Turkoz-Acar E, Sipahi H, et al. Comparative study of molasses produced by traditional and industrial techniques from the viewpoint of furan derivatives, mutagenicity/antimutagenicity, and microbiological safety. Int Food Res J. 2021;28:1067–77.
International Commission on Microbiological Specifications for Foods (ICMSF). Microorganisms in foods 6: microbial ecology of food commodities. 2nd ed. Springer: New York; 2005.
Jayasena V, Cameron I. Brix/acid ratio as a predictor of consumer acceptability of crimson seedless table grapes. J Food Qual. 2008;31:736–50. https://doi.org/10.1111/j.1745-4557.2008.00231.x.
Al-Rouz MA. The effect of chemical treatments and condensation technique in biologically active substances in grape molasses. Int J Chem Tech Res. 2015;8(4):1638–44.
Funding
The authors would like to acknowledge the Holy Spirit University of Kaslik (USEK) for funding the experimental work of this project.
Author information
Authors and Affiliations
Contributions
CS conceived the idea and designed the experiments. AAK carried out all the experiments and collected all the data. AK performed the chemometric analysis. CS and AK contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The protocol was approved by the Ethical committee of the Faculty of Agricultural Sciences at the Holy Spirit University of Kaslik, represented by Dr. Lara Hanna, Dr. Nabil Nemer and Dr. Marc Bou Zeidan, in accordance with the guidelines for ethical and professional practices for the sensory analysis of foods published by the Institute of Food Science and Technology. All the panelists were regular consumers of grape molasses and they gave their informed consent to participate in the sensory evaluation of grape molasses samples.
Consent for publication
All the authors have read and approved the content of this manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salameh, C., Abi Khalil, A. & Kassouf, A. Multivariate investigation of physicochemical, nutritional and sensory attributes of traditional Lebanese grape molasses. Discov Food 4, 48 (2024). https://doi.org/10.1007/s44187-024-00111-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44187-024-00111-w