Abstract
The tubers of Tropaeolum tuberosum, locally known as Isaño and native to the Andean region of South America, have been known since ancient times for their multiple uses in the Bolivian population. They are used both as food in various preparations and in traditional medicine. In this investigation, we report the study of three Isaño cultivars currently consumed in Bolivia. We determined their proximal composition, characterized carotenoids, determined antioxidant capacity, measured total phenols and total flavonoids, and quantified the major polyphenols. The results show that, apart from being a source of important nutrients such as proteins, Bolivian Isaño is a source of antioxidants and has a higher concentration of flavonoids and anthocyanidins, particularly in the purple cultivar. Additionally, we identified the presence of three carotenoids in this food for the first time: Lutein, Neoxanthin, and β-carotene. Through these types of studies, we aim to revalue this food, which is little known both in Bolivia and outside the Andean region of South America. Considering its nutritional properties, we seek to increase its consumption.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Tropaeolum tuberosum, better known in the Bolivian highlands as “Isaño”, also has other vernacular names dependent on the region as isañu, mashua, mashwa, mishwa, añu among others; it is a species used and distributed in various high Andean communities of the Andes in South America [1].
Tropaeolum tuberosum Ruiz & Pavon (Tropaeolaceae family) is herbaceous plant with shiny dark green thin leaves. It is a tetraploid species, with chromosome number x = 13 (2n = 4x = 52) [2], characterized by the presence of tubers. The tubers present a turbine or conical shape, 5 to 15 cm long and 3 to 6 cm wide showing several colors such us gray, white, yellow, red, purple and black [3]. They are commonly confused with tubers of oca (Oxalis tuberosa) another crop from de Andean region, since the Isaño has similar colors but easily distinguishable mainly by its conical shape [1, 4].
In traditional medicine, it is used for the treatment of venereal, pulmonary and skin diseases, anemia, reduction of sexual appetite, wound healing, analgesic, anti-inflammatory for kidneys, prostate and against bladder [2, 5] and it also has diuretic effects [6]. These biological activities could be closely related to the presence of its secondary metabolites, such as hydroxybenzoic acids, phenolic compounds, tannins, flavonols, anthocyanins, glucosinolates, isothiocyanates, phytosterols, carotenoids and fatty acids among others [2, 7, 8].
The crop of Isaño grows from a range of 2400 to 4300 meters above sea level (m.a.s.l.) [4] and currently is considered an underutilized crop, there is a clear risk of loss as a crop due to factors such as crop change, other species with higher economic profitability, lack of public knowledge, climate change among others [9]. Isaño used to grow in poor soil without the support of fertilizers and pesticides [3].
The strong flavor of Isaño usually improves after exposure to solar radiation where it acquires a more pleasant taste to the palate; the popular forms of Isaño consumption are as “huatia” (cooking underground) and boiled [5], but the most accepted form by the Bolivian population is frozen and it is known by the trade Aymara name "Thayacha" which is a type of Andean ice cream, marketed in winter [5, 10].
In the present investigation we report the chemical study of three Isaño cultivars (one the most consumed by the population of the Bolivian altiplano), determining their proximal composition, the characterization and identification of carotenoids, determination of antioxidant capacity, total phenols and total flavonoids, in addition to the quantification of the most abundant polyphenols.
2 Experimental
2.1 Chemicals
The reagents used were: Folin-Ciocalteu (Sigma-Aldrich, St. Louis, USA.), gallic acid (Sigma life Science, China), sodium carbonate (Biopack, Buenos Aires, Argentina), ABTS (2,2-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid 98%) Sigma Aldrich, Canada; catechin gallate, potassium persulfate 99, 0% (Sigma-Aldrich, Japan), Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid, 97%) Sigma Aldrich, Denmark, TPTZ (2,4,6-tripyridyl-s-triazine) Sigma Aldrich, Switzerland; ferric chloride (Biopack, Argentina), acetic acid (glacial p. a.) Merck KGaA, Darmstadt, Germany; sodium acetate (Sigma Aldrich, India), hydrochloric acid (Sigma-Aldrich, St. Louis, USA), sodium acetate (Sigma Aldrich, India), hydrochloric acid (Sigma-Aldrich, St. Louis, USA), absolute methanol from J.T. Baker (Mexico DF, Mexico), ethanol from Merck (Darmstadt, Germany), acetone from Biopack, Argentina; aluminum chloride hexahydrate (Sigma-Aldrich, Germany), sodium nitrite (Scharlab S.L., Spain), Ethyl acetate suitable for HPLC, ≥ 99.7%, petroleum ether p.a., sodium hydroxide (Merck KGaA, Darmstadt, Germany) β-Carotene 97% (Sigma-Aldrich, Switzerland), BHT (2,6-Di-tert-butyl-4-methylphenol) from Acros Organics, New Jersey, USA; delphinidin, cyanidin, pelargonidin obtained from Extrasynthèse (Genay, France), finally, The mili-Q ultrapure water obtained from a Millipore simplicity 185, Brazil.
2.2 Samples
The samples cultivated (Fig. 1) were obtained in June 2022 from Aymara population of the Apuvillque community (Municipality of Huarina, Omasuyos province), La Paz department, Bolivia with coordinates 16° 08′ 02.9 ʺ S 68° 38′ 32.8ʺ W to 3893 m.a.s.l. Every isaño were collected from several plants; was collected, a representative sample (5 kg). Each cultivar was randomly separated into five groups (approximately 1 kg each), the tubers were cleaned and stored in plastic bags in a − 20 °C freezer, until the day of analysis. Each group was analyzed independently.
Each Isaño sample has different traditional names by the local culture [11], according to its shape and color, where sample 1 is known as "Santo jonk'ori" (miraculous saint) due to the red dots on the tuber sprouts (like the bleeding eyes of a saint), sample 2 as "Achakani Isaño" similar to Achakana that means cross in aymara local language regarding the shape and sample 3 known as "Ch'iyara Isaño" which means black isaño.
2.3 Proximate analysis
All fresh samples were analyzed five times for moisture percentage using a moisture analyzer (Radwag, MAC 110/WH, Poland). For pH the method recommended by AOAC 943.02 [12] was using a pH meter (pH-mV-Temp 305225, ISOLAB, Germany). The sun exposure process of tubers and total solids measuremnet of the fresh and sunned samples were analyzed with a hand refractometer (RF.5532, type 0–32% ATC, The Netherlands) as described in [13]. For ash determination a microprocessed muffle furnace (QUIMIS Q318M21 Diadema-SP-, Brazil) was used using the AOAC method 923.03 [12]. For total proteins (N × 6.25) the AOAC method 960.52A [12], was adapted to the characteristics of the equipment Compact digestion system (RAYPA MBCM-40, R. ESPINAR, S.L. Spain) and Micro Kjeldahl (RAYPA Distillation unit DNP, R. ESPINAR, S.L. Spain) and finally for the total percentage of fats the soxhlet liquid solid extraction method was used. The total carbohydrate content was determined by 100—(moisture + protein + fat + ash) % difference.
2.4 Extraction
Approximately 100 mg of sample were weighed to which 1 mL of methanol 90% was added. The samples were sonicated at 0 ºC for 15 min. They were then centrifuged at 12,000 rpm for 2 min. The extraction was used for the determination of antioxidants, phenols and total flavonoids, the supernatant was stored at 4 ºC before analysis [14].
2.5 Determination for ABTS
Through the mixture of ABTS (2,2'-azinobis(3-ethylbenzothiazoline-6-sulfonic acid)) 7 mM (colorless) with potassium persulfate 140 mM (24 h of rest, minimally prior to the analysis), the oxidized product ABTS + (green color) is obtained, this radical cation in solution was diluted in acetic acid-sodium acetate buffer (pH = 5) until obtaining an absorbance of approximately 0.700 ± 0.02 at 734 nm. Where 100 µL of sample or standard was mixed with 1 mL of ABTS + solution with 6 min rest for stabilization, then the measurement at 734 nm was performed in a multichannel reader (Biotek uQuant, USA). The decrease in absorbance caused by the addition of sample was calculated with that of a standard curve made by use of Trolox (20–200 µmol/L) [14].
2.6 Determination for FRAP
FRAP (Ferric reducing antioxidant power) solution is formed through the ferric complex from the mixture of acetic acid-sodium acetate buffer solution (pH = 3.6) with 10 mM TPTZ and 20 mM iron chloride. For 30 µL of sample, 120 µL of distilled water and 900 µL of FRAP solution were used. The absorbance was measured at 593 nm. The final absorbance of each sample was calculated with that of a Trolox standard curve (100–1000 µmol/L) [14].
2.7 Total phenol content (TPH)
The total phenolic compounds were determined from 50 µL of sample were used with 1 mL of Folin-Ciocalteu reagent solution diluted to 10% and 500 µL of 7.5% sodium carbonate. They were vortexed and incubated for 30 min at 45 °C to stabilize the reduction reaction and the formation of the blue complex. Subsequently, they were cooled to room temperature to measure their absorbance at 765 nm. The final absorbance of each sample was calculated with that of a standard curve of gallic acid (230–1170 µmol/L) [14].
2.8 Total flavonoid content (TF)
The method described in [14] slightly modified was used where 150 µL of sample, 45 µL of 10% sodium nitrite and 45 µL of 20% aluminum chloride hexahydrate were used and left to stand for 10 min for the formation of the pink colored flavonoid-aluminum complex, then 300 µL of 1 M sodium hydroxide and 600 µL of water were added. The absorbance readings were taken at 510 nm. The final absorbance of each sample was calculated with a standard curve of catechin (69–689 µmol/L).
2.9 Total carotenoid content (CT)
For the extraction, the method described by [15] was slightly modified. Where from 0.5 g of sample we added 5 mL of ethanol-acetone mixture (1:1 v/v) containing 200 mg/L of BHT, the mixture was vortexed for 30 s, and centrifuged at 12,000 rpm for 5 min at 4 ºC, the procedure was repeated until the disappearance of color in the extraction solvent. The supernatants were measured at 450 nm; the concentration of total carotenoids was calculated from a standard curve of β-carotene in a range of 5 to 50 µg/mL.
2.10 Quantification by HPLC–DAD
Polyphenols were analyzed by HPLC. For hydrolysis of glycosides, extracts were subjected to reflux (90 °C) where 300 µL of sample extract were mixed with 300 µL 3.0 M HCl and 300 µL methanol for 75 min. Phenolic compounds were separated using an Agilent liquid chromatography system (1100 Series, USA), consisting of a vacuum degasser (G1322A), a solvent supply module (Quat Pump-G1311A), a column oven (ColCom-G1316A). The reverse phase column used was a 5 µm Agilent Eclipse Plus C18 (150 × 4.6 mm) equipped with a 10 mm pre-column. The injection volume was 20 µL with a flow rate of 0.8 mL/min. The mobile phase was a binary solvent system consisting of A (1% acetic acid in water) and B (methanol) using a gradient program of 0 min:40% B; 5 min:65% B; 10 min: 90% B; 15 min: 40% until 17 min. The UV–VIS absorbance of the eluate was recorded with a diode array detector (G1315B) (Agilent, Santa Clara, CA) at 280, 360 and 530 nm. Compounds were identified by comparing them with the standards of each compound identified using the retention time, the profile of the absorbance spectrum [14].
2.11 Characterization of carotenoids by HPLC–MS
Prior to the analysis, the Isaño tuber was cleaned, dried (35 ºC) and ground, it was treated with petroleum ether (ratio 1:10 sample/solvent), the ethereal extract was rotavapored at 35ºC (Büchi CH-9230, Switzerland), the dry extract was subjected to column chromatography on silica gel, eluting initially with petroleum ether and later with mixtures of different concentrations of petroleum ether. petroleum and ethyl acetate of increasing polarity gradient, the fraction eluted with petroleum ether-ethyl acetate (5:1 v/v) was the one that showed the presence of reddish colored substances.
HPLC-HPLC–MS analysis was carried out on a Purospher® RP-18 column (5 µm) in a Waters 2695 separation module equipped with a 996 PDA detector and a Waters Quattro micro API quadrupole system, liquid chromatography was performed with a mobile phase composed of 0.1% formic acid in water as solvent A and methanol as solvent B. Flow rate: 1 mL/min. The analyte was separated by isocratic elution using A/B = 35/65 (v/v) in 15 min. The mass spectrometer operation conditions were: capillary Voltage 3.0 kV, cone voltage 25 V, source temp. 120 °C, desolvation temp 350 °C, Gas flow for desolvation 500 L/h all compounds were analyzed under ESI in positive mode compound was detected with a Waters Quattro ESI mass spectrometer operated in positive mode, model 4micro (Milford Massachusetts, USA).
2.12 Statistic analysis
All data are reported as the average of five analyses for each cultivar and its standard deviation. To determine significant differences in the data, they were subjected to analysis of variance (ANOVA). Differences between means were compared using the Tukey test at 5% with IBM SPSS Statistics 22.0, USA. Principal component analysis (PCA) and Pearson's correlation were carried out using as performed using the statistical language R version 4.2.3 (R Foundation for Statistical Computing, Vienna, Austria).
3 Results
The results obtained from the different isaño cultivars, which showcase the proximal composition, antioxidant capacity, characterization of carotenoids, and polyphenols, are presented below. These findings provide valuable insights and contribute significantly to the understanding of the nutritional properties of this Andean food.
3.1 Proximate analysis
Although the three cultivars show significant differences in colour, appearance and size. according to Table 1, there were no significant differences in nutritional parameters such as moisture, soluble solids, ash, protein, fat and carbohydrates. The soluble solids in the initial material are between 8.4 and 8.9 ºBrix and when soaked for seven days, they increase a third part of their initial state to 11.6–12.2 ºBrix.
It was found that the pH values maintained a similar value among the three cultivars, in addition to the percentage of ashes, which had values from 0.77 to 0.89% in the different cultivars, with no significant differences. The protein percentage is between 1.5 and 2.3% and finally the fatty material in all samples was below 0.3%. Total carbohydrates (13.7–17.1%) were calculated by difference between 100 and the sum of water, protein, total lipid and ash.
3.2 Carotenoids
Although previous studies show the quantification of carotenoids, these do not report which carotenoids are present in T. tuberosum, therefore, this study will be the first to attempt to characterize carotenoids in Isaño samples by HPLC–MS. The major carotenoids are described in Table 2 and, such compounds are supported by literature [16]. For the first time, the characterization was performed by comparing the UV–VIS max value, molecular mass and fragments where: Lutein shows a molecular weight of 569 (M + H) with mass fragments of 551 (M + H-H2O), 430 (M + H-139). Neoxanthin with a molecular weight of 601 plus fragments of 583(M + H-H2O), 565(M + H-2H2O), 547 (M + H-3H2O), 509 (M + H-H2O-92) and β-carotene with a molecular weight [M + H] of 537 and mass fragments 413, 400, 269. According to their structures it can be seen that the presence of xanthophyll type carotenoids (except β-carotene).
3.3 Antioxidant capacity and polyphenols
Table 3 shows the results of the antioxidant; the results indicate that by the ABTS ranked between 2.9 to 11.6 μmol/g while FRAP method ranked 4.5 to 30.0 μmol/g of fresh matter (FM). In the case of total phenols, the values were in a range of 2.0 to 11.7 μmol/g of fresh sample expressed in gallic acid equivalents (GAE). Total flavonoids expressed in catechin equivalents (CE) shows a range of 1.8 to2.4 μmol/g of fresh sample.
It can be seen that between samples 1 and 2 there is no significant difference, compared to sample 3 which evidently has higher antioxidant capacity, phenolic compounds and flavonoids.
Finally, the analysis of total carotenoids showed a range between 57.3. to 144.0 μg ß-carotene/g, where the samples 1 and 2 have a higher concentration of total carotenoids in comparison of sample 3, which showed an inversely proportional relationship with its antioxidant capacity.
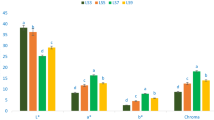
Table 4 shows the main polyphenols found in the Isaño samples, where they present 31.95–40.11 μg /g of gallic acid, with no significant difference between samples. 7.0–13.3 μg /g of epicatechin where sample 2 presented lower concentrations; 0.7–4.8 μg /g of catechin gallate where sample 2 presented higher concentrations. In addition, after the hydrolysis of the anthocyanins, it was possible to quantify the anthocyanidins delfidin, cyanidin and perlagonidin (Fig. 2), where clearly sample 3 presents higher concentrations of these polyphenols, which would be related to its high antioxidant capacity.
4 Discussions
4.1 Proximate analysis
The results of proximal analysis are shown in Table 1, where no significant difference was evident, which could indicate that despite their differences mainly in color, these tubers have the similar nutritional characteristics. The moisture content, in the samples of the present study showed values between 80.3 to 83.0%, which is in a similar range reported in previous studies [2, 17, 18]. The range of soluble solids in the samples (without sunlight) of this study were between 8.4 to 8.9 ºBrix which are comparable to literature (6.1–8.5 ºBrix) [17]; but this value can increase significantly when samples are exposed to solar radiation as can be seen in Table 1. Consumers of this food traditionally expose the tubers to the sun to improve their taste, by increasing the free sugars [5], these changes in soluble solids content were also observed in Oca tubers (Oxalis tuberosa) previously, this phenomenon is attributed to an increase in free sugars such as glucose and sucrose [13].
The pH values were around 6, which are in agreement with previous studies [17], but it was observed that there are also cultivars that have more acidic pH values (3.9–4.2) [18], this pH variation could be related to the post-harvest time showing that the cultivars of the present study were fresh. The values of ash determined in Isaño ranged from 0.77 to 0.89% and they are in accordance with the literature [2, 17], The ash content is related to the minerals present in the samples particularly phosphorus, calcium, magnesium, manganese, zinc, iron, sodium and potassium [2].
The protein values in Isaño were determined in several studies and in the present study a wide variability that could be linked to the type of study accession, sampling site, sampling method used among other parameters but the range is between 1.1 and 2.7% in FM [2, 17], while the values measured in this study is between 1.5 and 2.3%, showing higher protein content compared to other tubers such as arracacha, yacon [19] and comparable to different varieties of potato [20, 21]. The fatty material in the Isaño is scarce as it is reported values less than or equal to 1% [2], which was replicated in this study when values below 0.3% were found. The content of carbohydrates measured in fresh Isaño range from 13.78–17.1% while in previous studies, a range of 9.8 to 12.8% were reported [2, 17].
4.2 Carotenoids
Carotenoids are the most extensive group of natural pigments, characterized by being indispensable cellular components in living beings [22], responsible for the attractive colorations in various foods [23]. These compounds are classified into two groups according to their structural characteristics: carotenes consisting of carbon and hydrogen (β-carotene, α-carotene and lycopene), and xanthophylls, consisting of carbon, hydrogen and additionally oxygen (lutein, zeaxanthin, astaxanthin and fucoxanthin) [24]. The importance of carotenoid intake in food is due to the fact that no member of the animal kingdom, including humans, is able to biosynthesize these compounds and they are closely associated with good health, due to the properties attributed to them such as the effect against oxidative stress [25], as well as preventing the onset of certain types of cancer and macular degradation, protecting against sunburn reactions and improving immune function [23].
The coloration of the different cultivars of T. tuberosum ranging from yellow to purple and black, due to natural pigments such as anthocyanins and carotenoids. The presence of carotenoids in Isaño was confirmed by several spectrophotometric studies at different concentrations; 1 to 25 µg g−1 [26], 2.6 to 96.7 µg g−1 [19], 12.8 to 85.8 µg g−1 [27] and 70 to 132 µg g−1 [28] expressed in β-carotene equivalents.
In the present study it was found similar concentrations. It should be taken into account that the concentration of carotenoids tends to vary according to the stage of maturity, variety and post-harvest storage. It was also found that the darker cultivars showed lower concentrations of carotenoids [28].
Moreover, this study reports for the first time the presence of lutein, neoxanthin and β-carotene characterized by HPLC–MS/MS through the comparison of molecular weight, mass fragments and UV–VIS spectra. As it was mentioned below, it is the first time that these molecules are identified in Tropaeolum tuberosum but not in the Tropaeolaceae family since they were identified previously in Tropaeolum majus [29].
4.3 Antioxidant capacity and polyphenols
Some of the compounds previously found in Isaño were caffeic acid, chlorogenic acid, gallic acid, hydroxycinnamic acid and hydroxybenzoic acid; the flavonoids rutin, quercetin, anthocyanins derived from delphinidin, cyanidin, pelargonidin, as well as other uncommon compounds such as glucosinolates, alkamides, phytosterols, among others [2, 30], which could contribute to the antioxidant activity in Isaño.
Previously, several studies of antioxidant capacity were reported [2, 18, 26] that could not be compared with the present study because the differences in sample preparation or because they are expressed in other units, as shown in Table 5. The results obtained in the present research are comparable with other Andean tubers such as Oca (Oxalis tuberosa), Ullucu (Ullucus tuberosus), Arracacha (Arracacia xanthorrhiza) and colored potatoes (Solanum sp.)while the purple Isaño (sample 3) has a higher antioxidant capacity than all the tubers in Table 5, except for the concentration of total phenols and flavonoids, which is lower than that of the colored potatoes. This can be explained due to the presence of anthocyanins.
From literature a previous study presented the quantification of polyphenols in Isaño showed the presence of caffeic acid, rutin, chlorogenic acid and quercetin [31]. In the present study (Table 4), it was quantified gallic acid, epicatechin and catechin gallate also present in other tubers [37, 38]. The main anthocyanidins delphinidin, cyanidin and pelargonidin were quantified for the first time in Isaño. These anthocyanidins are also present in various tubers, fruits, berries, vegetables, wines and flowers, and they are responsible for the red to blue color [32, 36, 39]. In addition, biological properties are attributed to it as antimicrobial, anti-inflammatory, antioxidant, chemoprotective, cardiovascular, anticarcinogenic, photoprotective, positive modulation of intestinal bacterial growth, inhibition of platelet aggregation and support in the regulation of diabetes [40].
The methods presented a high correlation between FRAP and ABTS with the content of phenols and total flavonoids, it was also found a positive correlation of the ABTS, FRAP, TPH and TF with malvidin, cyanidin and pelargonidin, while all of these methods showed negative correlations with the concentration of total carotenoids. These results could indicate that there is a trend related to color where darker (purple) cultivars have higher antioxidant capacity and anthocyanins but low carotenoid concentrations and conversely for lighter (yellow) cultivars. All the correlations are presented in Fig. 3.
While the PCA reveals that the three samples are classified into different clusters according to the parameters analyzed, as shown in the Fig. 4. PC1 and PC2 together explained more than 95% of the total variance. It could also be confirmed that sample 3 has the highest antioxidant capacity due to its high content of anthocyanidins, while sample 1 and 2 showed a high content of carotenoids and proteins.
5 Conclusions
This study reports for the first time the presence of the carotenoids lutein, neoxanthin and β-carotene in samples of Isaño from Bolivia, determined by HPLC–MS/MS through the comparison of molecular weight, mass fragments and UV–VIS spectra. In addition, the antioxidant capacity and quantification of polyphenols were performed, showing higher values in comparison with other tubers in particular sample 3 (purple cultivar).
Proximal analysis was carried out among the cultivars, and it was possible to find a high protein and carbohydrates content in Isaños. It was also seen that the results of the antioxidant capacity have statistically significant correlation with the anthocyanins content and an inverse correlation with the carotenoids content; these results can indicate that the intensity of purple color in the tubers will have higher antioxidant capacity but lower concentration of carotenoids.
The locally consumed isaños in Bolivian Altiplano are an important source of active compounds for the Andean population. These findings contribute, from both a chemical and nutritional perspective, to the valorisation of this food. Through these kinds of studies, it is intended to revalue this food, little known outside the South American region, and considering its nutritional properties, increase its consumption.
Data availability
All data generated or analysed during this study are included in this published article.
References
Lim TK. Tropaeolum tuberosum. In: Lim TK, editor. Edible medicinal and non-medicinal plants. Springer Cham: Switzerland; 2016. p. 94–102. https://doi.org/10.1007/978-3-319-26065-5_3.
Apaza Ticona LN, Tena Pérez V, Bermejo BP. Local/traditional uses, secondary metabolites and biological activities of Mashua (Tropaeolum tuberosum Ruíz & Pavón). J Ethnopharmacol. 2020;247: 112152. https://doi.org/10.1016/j.jep.2019.112152.
Siqueira MVBM, do Nascimento WF, Pereira DA, Cruz JG, de Castro Vendrame LP, Veasey EA. Origin, domestication, and evolution of underground starchy crops of South America. In: Cereda MP, Vilpoux OF, editors. Starchy crops morphology, extraction, properties and applications. Academic Press: Cambridge; 2023. p. 17–42. https://doi.org/10.1016/B978-0-323-90058-4.00011-6.
Luziatelli G, Alandia G, Rodríguez JP, Manrique I, Jacobsen SE, Sørensen M. Ethnobotany of Andean minor tuber crops: tradition and innovation—oca (Oxalis tuberosa Molina—Oxalidaceae), mashua (Tropaeolum tuberosum Ruíz & Pav—Tropaeoleaceae) and ulluco (Ullucus tuberosus Caldas—Basellaceae). In: Pascoli M, Vilpoux OF, editors. Varieties and landraces: cultural practices and traditional uses. Elsevier: Amsterdam; 2023. p. 79–100. https://doi.org/10.1016/B978-0-323-90057-7.00009-7.
Aruquipa R, Trigo R, Bosque H, Mercado G, Condori J. El Isaño (Tropaeolum tuberosum) un cultivo de consumo y medicina tradicional en Huatacana para el beneficio de la población boliviana. Rev Investig e Innovación Agropecu y Recur Nat. 2016;3(2):146–51.
Castro Rua M, Gutiérrez Patzi E, Pacheco Felipes G, Tola Guarachi E, Callisaya Mamani A, Flores Quispe M, Gonzales Beltrán H, Villca Condori A, Apaza Chambi L, Terán VE. Determinación del efecto diurético del Isaño (Tropaeolum tuberosum) y cola de caballo (Equisetum arvense) en ratones. Rev Científica Ciencias la Salud. 2022;14(2):80–90. https://doi.org/10.17162/rccs.v14i2.1658.
Guevara-Freire D, Valle-Velástegui L, Barros-Rodríguez M, Vásquez C, Zurita-Vásquez H, Dobronski-Arcos J, Pomboza-Tamaquiza P. Nutritional composition and bioactive components of mashua (Tropaeolum tuberosum Ruiz and Pavón). Trop Subtrop Agroecosyst. 2018;21(1):53–68. https://doi.org/10.56369/tsaes.2561.
Pacheco MT, Escribano-Bailón MT, Moreno FJ, Villamiel M, Dueñas M. Determination by HPLC-DAD-ESI/MSn of phenolic compounds in Andean tubers grown in Ecuador. J Food Compos Anal. 2019;84: 103258. https://doi.org/10.1016/j.jfca.2019.103258.
Padulosi S, Hoeschle-Zeledon I. Underutilized plant species: what are they? Leisa-Leusden. 2004;20:5–6.
Vidaurre PJ, Paniagua N, Moraes RM. Etnobotánica en los Andes de Bolivia. In: Moraes M, Øllgaard RB, Kvist LP, Borchsenius F, Balslev H, editors. Botánica Económica De Los Andes Centrales. La Paz: UMSA; 2006. p. 224–38.
Manrique I, Arbizu C, Vivanco F, Gonzales R, Ramírez C, Chávez O, Tay D, Ellis D. Tropaeolum tuberosum Ruiz & Pav. coleccion de germoplasma de mashua conservada en el Centro Internacional de la Papa (CIP). Centro Internacional de la papa. Lima, Perú; 2014. https://doi.org/10.4160/9789290604310.
Official Methods of Analyisis of AOAC International. 21 ed. Vol. I-III, Rockville, Maryland, USA; 2019.
Castañeta G, Castañeta R, Peñarrieta JM. Cambios fisicoquímicos por exposición a la radiación solar en tubérculos de Oxalis tuberosa, “oca” cultivados en Bolivia. Rev Boliv Quím. 2022;39(2):44–55. https://doi.org/10.34098/2078-3949.39.2.3.
Peñarrieta JM, Alvarado JA, Åkesson B, Bergenståhl B. Total antioxidant capacity and content of flavonoids and other phenolic compounds in canihua (Chenopodium pallidicaule): an Andean pseudocereal. Mol Nutr Food Res. 2008;52(6):708–17. https://doi.org/10.1002/mnfr.200700189.
Al-Farsi M, Alasalvar C, Morris A, Baron M, Shahidi F. Comparison of antioxidant activity, anthocyanins, carotenoids, and phenolics of three native fresh and sun-dried date (Phoenix dactylifera L.) varieties grown in Oman. J Agric Food Chem. 2005;53(19):7592–9. https://doi.org/10.1021/jf050579q.
Rodriguez-Amaya DB. A guide to carotenoid analysis in foods. Washington: International Life Sciences Institute, ILSI Press; 2001.
Malpartida Yapias JR, Astete Adama J, CajachaguaUscuchagua Y, Rosales Sánchez MC. Características fisicoquímicas, composición nutricional y compuestos bioactivos en tres variedades de Mashua (Tropaeolum tuberosum Ruiz y Pavón): una revisión. Rev Tecnológica Espol. 2022;34(2):41–51. https://doi.org/10.37815/rte.v34n2.891.
Heinert Jiménez ME, Gutiérrez Gaitén YI, Yamilet Guaranda I, Miranda Martínez M. Chemical study and determination of the antioxidant activity of three varieties Tropaeolum tuberosum (Mashua). Am J Plant Sci. 2019;10(12):2279–97. https://doi.org/10.4236/ajps.2019.1012159.
Choquechambi LA, Callisaya IR, Ramos A, Bosque H, Mújica A, Jacobsen SE, Sørensen M, Leidi EO. Assessing the nutritional value of root and tuber crops from Bolivia and Peru. Foods. 2019;8(11):526. https://doi.org/10.3390/foods8110526.
De Haan S, Burgos G, Liria R, Rodriguez F, Creed-Kanashiro HM, Bonierbale M. The nutritional contribution of potato varietal diversity in Andean food systems: a case study. Am J Potato Res. 2019;96:151–63. https://doi.org/10.1007/s12230-018-09707-2.
Jimenez ME, Rossi AM, Samman NC. Phenotypic, agronomic and nutritional characteristics of seven varieties of Andean potatoes. J Food Compos Anal. 2009;22(6):613–6. https://doi.org/10.1016/j.jfca.2008.08.004.
Karnaukhov VN. Carotenoids: recent progress, problems and prospects. Comp Biochem Physiol. 1990;95(1):1–20. https://doi.org/10.1016/0305-0491(90)90241-K.
Xavier AA, Pérez-Gálvez A. Carotenoids as a source of antioxidant in the diet. In: Stange C, editor. Carotenoids in nature: biosynthesis, regulation, and function. Springer: Cham; 2016. p. 359–75. https://doi.org/10.1007/978-3-319-39126-7_14.
Jaswir I, Noviendri D, Hasrini RF, Octavianti F. Carotenoids: sources, medicinal properties and their application in food and nutraceutical industry. J Med Plants Res. 2011;5(33):7119–31. https://doi.org/10.5897/JMPRx11.011.
Allen Olson J. Vitamin A and carotenoids as antioxidants in a physiological contex. J Nutr Sci Vitaminol. 1993;39:57–65. https://doi.org/10.3177/jnsv.39.Supplement_S57.
Campos D, Noratto G, Chirinos R, Arbizu C, Roca W, Cisneros-Zeballos L. Antioxidant capacity and secondary metabolites in four species of Andean tuber crops: native potato (Solanum sp.), mashua (Tropaeolum tuberosum Ruiz & Pavón), oca (Oxalis tuberosa Molina) and ulluco (Ullucus tuberosus Caldas). J Sci Food Agric. 2006;86(10):1481–8.
Jacobo-Velázquez DA, Peña-Rojas G, Paredes-Avila LE, Andía-Ayme V, Torres-Contreras AM, Herrera-Calderon O. Phytochemical characterization of twenty-seven peruvian Mashua (Tropaeolum tuberosum Ruíz & Pavón) morphotypes and the effect of postharvest methyl jasmonate application on the accumulation of antioxidants. Horticulturae. 2022;8(6):471. https://doi.org/10.3390/horticulturae8060471.
Chirinos R, Campos D, Arbizu C, Larondelle Y, Noratto G, Cisneros-Zevallos L. Effect of genotype, maturity stage and post-harvest storage on phenolic compounds, carotenoid content and antioxidant capacity, of Andean mashua tubers (Tropaeolum tuberosum Ruiz & Pav on ). J Sci Food Agric. 2007;87(3):437–46. https://doi.org/10.1002/jsfa.2719.
Niizu PY, Rodriguez-Amaya DB. Flowers and leaves of Tropaeolum majus L. as rich sources of lutein. J Food Sci. 2005;70(9):S605–9. https://doi.org/10.1111/j.1365-2621.2005.tb08336.x.
Chirinos R, Campos D, Betalleluz I, Giusti MM, Schwartz SJ, Tian Q, Pedreschi R, Larondelle Y. High-performance liquid chromatography with photodiode array detection (HPLC-DAD)/HPLC-Mass Spectrometry (MS) profiling of anthocyanins from Andean mashua tubers (Tropaeolum tuberosum Ruíz and Pavón) and their contribution to the overall antioxidant activity. J Agric Food Chem. 2006;54(19):7089–97. https://doi.org/10.1021/jf0614140.
Behar H, Reategui O, Liviac D, Arcos J, Best I. Phenolic compounds and in vitro antioxidant activity of six accessions of mashua (Tropaeolum tuberosum r. & p.) from Puno region, Peru. Rev Fac Nac Agron Medellin. 2021;74(3):9707–14. https://doi.org/10.15446/rfnam.v74n3.93020.
Mollinedo P, Peñarrieta JM. Anthocyanins, antioxidant capacity and content of flavonoids and other phenolic compounds oca (Oxalis Tuberosum) an Andean tuber. Austin J Nutr Metab. 2014;1(1):4.
Peñarrieta JM, Alvarado JA, Ǻkesson B, Bergenståhl B. Total antioxidant capacity in Andean food species from Bolivia. Rev Bol Quim. 2005;22(1):89–93.
Peñarrieta JM, Alvarado JA, Bergenståhl B, Åkesson B. Spectrophotometric methods for the measurement of total phenolic compounds and total flavonoids in foods. Rev Bol Quim. 2007;24(1):5–9.
Tejeda L, Mollinedo P, Aliaga-Rossel E, Peñarrieta JM. Antioxidants and nutritional composition of 52 cultivars of native andean potatoes. Potato Res. 2020;63:579–88. https://doi.org/10.1007/s11540-020-09458-w.
Tejeda L, Alvarado JA, Dębiec M, Peñarrieta JM, Cárdenas O, Alvarez MT, Chawade A, Nilsson L, Bergenståhl B. Relating genes in the biosynthesis of the polyphenol composition of andean colored potato collection. Food Sci Nutr. 2014;2(1):46–57. https://doi.org/10.1002/fsn3.69.
Peñarrieta JM, Mollinedo P, Aliaga-Rossel E, Vila JL, Bravo JA. Gallic acid and its derivatives: occurrence and identification in high altitude edible and medicinal plants. In: Thompson MA, Collins PB, editors. Handbook on gallic acid: natural occurrences, antioxidant properties and health implications. Nova Science Publishers: Hauppauge; 2013. p. 313–28.
Yilmaz Y. Novel uses of catechins in foods. Trends Food Sci Technol. 2006;17(2):64–71. https://doi.org/10.1016/j.tifs.2005.10.005.
Khoo HE, Azlan A, Tang ST, Lim SM. Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr Res. 2017;61(1):1361779. https://doi.org/10.1080/16546628.2017.1361779.
Jeyaraj EJ, Vidana Gamage GC, Cintrat JC, Cho WS. Acylated and non-acylated anthocyanins as antibacterial and antibiofilm agents. Discov Food. 2023;3:21. https://doi.org/10.1007/s44187-023-00062-8.
Acknowledgements
The present study was supported by the Swedish Development Cooperation Agency (SIDA). To Boris M. Mamani-Ichuta for his help with the statistical graphics.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
GC conceptualized, designed the research, formal analysis and writing the original draft. DMF, method validation, formal analysis and data interpretation. MB carotenoids analysed, and data interpretation. JMP sourced the funding, conceptualized, designed the research, supervised the study, and reviewed and edited the final manuscript draft.
Corresponding author
Ethics declarations
Ethics and consent to participate
This study adheres to all institutional and national regulations from Bolivia, including the collection of materials. It has received authorization and oversight from the Instituto de Investigaciones Químicas (IIQ) at the Universidad Mayor de San Andrés UMSA, a government-accredited scientific institution. It's important to note that only samples of the studied species are collected, and no individuals are sacrificed. This species has a wide distribution and is not categorized by either IUCN or CITES regulations.
Consent for publication
Not applicable.
Competing interests
No potential conflicts of interest were reported by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Castañeta, G., Miranda-Flores, D., Bascopé, M. et al. Characterization of carotenoids, proximal analysis, phenolic compounds, anthocyanidins and antioxidant capacity of an underutilized tuber (Tropaeolum tuberosum) from Bolivia. Discov Food 4, 9 (2024). https://doi.org/10.1007/s44187-024-00078-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44187-024-00078-8