Abstract
Metal exposure through cereal-based breakfasts was studied in humans of varied ages (infants, toddlers, children, adolescents, adults, and the elderly) to assess the potential health risks metals in food pose to consumers. The total concentrations of As (0.59–0.69 mg/kg), Cd (1.27–1.41 mg/kg), Cr (4.66–9.85 mg/kg), Mn (8.26–9.73 mg/kg), Ni (5.01–5.81 mg/kg) and Pb (0.83–0.92 mg/kg) were all higher than the regulatory limits for metals in cereal-based foods. Metal concentrations assessed via in-vitro bioaccessibility extracts were below the detection limits. Toxicity indices of As, Cd, Cr, Ni, and Pb were above their respective WHO-permissible tolerable daily intake for all age groups, implying possible health risks due to over-exposure to metals. While the hazard quotients for Cr and Mn among the age groups were less than 1, those for Cd and Ni were greater than 1. There was no public health concern for cancer risk associated with oral exposure to Pb among the various age groups. However, the estimated cancer risk of Cd (185.4 × 10–3) and As (9.2 × 10–3) was greater than the de minimus (10–6), suggesting a public health concern among various age groups. The study found a significant level of metal contaminants in cereal-based foods, which can potentially pose health risks to consumers who consume them.
Similar content being viewed by others
1 Introduction
Cereal-based breakfast meals constitute a major human diet. Cereals have long been a staple of the human diet due to their widespread cultivation and variety, high nutritional value, and remarkable storage qualities [1]. Metals enter cereal grains through the soil in which they are cultivated, agrochemicals used on farms, or processing machinery. They might also contaminate with metal through water, fertilizers, and pesticides [2]. As a result, some levels of potentially toxic metals are often detected in cereal meals. Long-term consumption of trace levels of metals through food, on the other hand, causes adverse physiological effects in humans, particularly in infants, children, and the elderly, who are the primary consumers of cereal-based breakfast meals.
The ingestion pathway accounts for nearly 90% of human exposure to metals [3], making it the primary entry route for metals in the human body [4]. Once ingested, the metals accumulate in the human body’s tissues and organs and affect the central nervous system [5]. They may also be deposited in the circulatory system and disrupt the normal functioning of internal organs such as the kidney and liver [6]. Though some metals, such as Mn and Ni, play essential roles in human growth, metabolism, and immune development, the others, such as As, Cd, and Pb, have no biological importance in the human body and are toxic even at the lowest concentrations [7]. For example, chronic exposure to Pb could lead to growth and intellectual disorders in children and blood pressure increase in adults [8]. Cadmium is a probable carcinogen [9] and causes a painful bone disease [10]. Arsenic exposure can cause cancer, skin lesions, cardiovascular disease, and diabetes through long-term ingestion via food [11].
As a result, heavy metal contamination in food is among the most critical components of food quality control. However, there is a scarcity of data on the extent to which cereal-based breakfast meals expose consumers to toxic metals. Furthermore, cereals grown in contaminated fields can translocate toxic metals from the soil into their grains [12]. Consequently, given that cereals are the first staple foods in most parts of the world, they can contribute to toxic metals exposure, mainly due to their high consumption rates [13].
Metal exposure has been routinely assessed using the total metal concentrations. Nonetheless, this method overestimates exposure because not all metal contaminants are adsorbed into the bloodstream during digestion. The bioavailability of a contaminant represents the amount absorbed into systemic circulation via major exposure pathways (dermal, oral, and inhalation). In-vivo studies are the best way to assess oral bioavailability. However, due to the high cost, time commitment, and ethical issues associated with animal testing, in-vitro bioaccessibility assays that mimic the processes of the gastrointestinal system in humans have been developed for use [3, 14]. Integrating bioaccessibility data in risk estimations increases the reliability of the assessment [15, 16]. This study assessed the levels of potentially toxic metals in cereal-based breakfast meals and determined the potential health risks associated with human exposure to the metals through ingestion using in-vitro bioaccessibility assay as a substitute for oral bioavailability.
2 Materials and methods
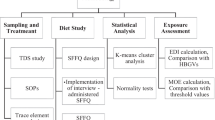
2.1 Study area
The Kumasi Metropolis of the Ashanti Region lies between latitudes 6.35–6.40° and longitude 1.30–1.37° with tropical rainy and dry climatic conditions and fairly steady temperature levels throughout the year [17]. According to the National Population and Housing Census [18] data, 443,981 people reside in the Kumasi metropolis. Kumasi is a large trade and commercial convergence hub, with two big markets (Kejetia and Adum) and many satellite markets, such as Asafo, Atonsu, Ayigya, Bantama, and Oforikrom Markets (Fig. 1), that serve traders from West African sub-region.
2.2 Sampling
A total of 54 samples of locally-produced cereals-based breakfast meals consisting of 31 breakfast cereals, 20 biscuits and 3 bread types were obtained from markets in the study area in December 2021. Samples were categorized according to the main cereal classes (wheat, rice, maize, sorghum and millet) and taken to the laboratory for further treatment prior to analyses.
2.3 Sample preparation
Samples were milled into a fine powder using a laboratory stainless steel blender at high speed for about 20 min. The milled samples were sieved through < 250-micron mesh into labelled polyethylene zip-lock bags and sealed for subsequent laboratory analyses.
2.4 Total metals concentration by XRF and ICP-MS
The samples were analyzed for potentially toxic metals using a Niton XL3t GOLDD + X-ray fluorescence (XRF) spectrometer. The XRF was calibrated with standard reference material (NIST 2711), and a system check was carried out each day before use. An aliquot of the sieved sample (2 g) was placed in the sample holder so it was three-quarters full, covered with a Mylar film, and cupped. The cupped sample was placed in the XRF shroud and scanned for 180 s. Replicate analysis was performed on each sample, and the average of the readings was recorded [15].
The powdered samples were analyzed for total metals by ICP-MS after acid digestion using USEPA Method 3052 [19]. Approximately 0.5 g of the sample was weighed into a microwave digestion vessel. Aliquots of 9 mL of concentrated nitric acid and 3 mL hydrochloric acid were added to the vessel and digested for 15 min in an Anton Parr Multiwave GO Plus microwave system. The digested was made to 40 mL volume with deionized water and analyzed by ICP-MS based on USEPA Method 200.8 [20].
2.5 In-vitro bioaccessibility extraction
The extraction protocol was based on USEPA standard operating protocols [21]. The sieved cereal-based breakfast sample (1.00 ± 0.05 g) was weighed into a 125 mL acid-cleaned HDPE bottle. An aliquot (100 ± 0.5 mL) of extraction fluid consisting of 30 g/L glycine adjusted to a pH of 1.5 with concentrated HCl was added to the bottle. The bottle was then sealed and placed into an extractor in batches of eight and rotated end-over-end in a 37 ± 2 °C water bath for 1 h. The extract was filtered through a 0.45-μm cellulose acetate filter and subsequently analyzed for metals by ICP-MS using USEPA Method 2008.8 [20]. The percentage bioaccessibility was calculated by dividing the metal concentration in the extract by the total metal concentration of powdered cereal-based food as determined by ICP-MS analysis. Bioaccessibility was not calculated for the samples with extract concentrations below the detection limits.
2.6 Quality assurance and quality control
To ensure the results' validity, reliability, and accuracy, various quality assurance and quality control measures were employed in this study. Polyethylene zip lock bags containing the samples were carefully sealed and coded. All equipment and glassware used were cleaned to reduce cross-contamination. During analysis, the use of procedure and reagent blanks, NIST 1575a control samples, and duplicates were carried out to ensure reproducibility and minimal contamination from filters, vessels, and sample containers.
Satisfactory recoveries were obtained for three replicate analyses of the NIST 1575a standard reference material, with an average recovery of 92 ± 11% for As, 85 ± 5% for Cd, 91 ± 16% for Cr, 90 ± 9% for Mn, 98 ± 6% for Ni, and 99 ± 6% for Pb. The method’s detection limits, computed based on the mean of the blank, the standard deviation of the blank, and the analytical sensitivity (slope) of the calibration plot employing a confidence factor of 3.2, ranged from 0.5 mg/kg (As) to 3.4 mg/kg (Mn). Reproducibility was assessed through the analyses of 5 duplicate samples, and the average relative percent difference between duplicate samples ranged from 9 to 14%, indicating satisfactory reproducibility.
2.7 Health risk assessment
The human health risk assessment was determined by the estimated dietary intake, carcinogenic and non-carcinogenic risks. To estimate both carcinogenic and non-carcinogenic health risks from cereal-based breakfast among various age groups, the estimated daily intake (EDI) of As, Cd, Cr, Ni, Mn, and Pb were computed (Eq. 1) based on Monte Carlo simulation software (Palisade, @Risk, USA).
where C is the metal concentration in the cereal-based breakfast meal (mg/kg); the MF (mean daily intake of cereal-based breakfast meals) is 265 g/day for adolescents, elderly, adults, and 106.9 g/day for children, infants, and toddlers [22]. BW, (average body weight), as per category, is as follows: 62 kg for adolescents, 74 kg for elderly individuals, 70 kg for adults, 23 kg for children, and 12 kg for infants and toddlers [22,23,24].
The metals' hazard quotient (HQ) was calculated using Eq. 2. If the HQ value is higher than 1.0, then there may be adverse health effects on human health.
where EDI is the estimated daily intake of metal (mg/day), and the oral reference dose (RfD) for the metals under study were 1.500, 0.001, 0.00357, 0.02, 0.140, and 0.0003 mg/kg/bw for Cr, Cd, Pb, Ni, Mn, and As, respectively [25]. The cancer risks (CR) of As, Cd and Pb exposure were estimated using Eq. 3.
The PF (potency factor) used for estimating cancer risk due to As Cd and Pb were adopted from USEPA regional screening levels of 1.5, 15, and 8.5 × 10–3 mg/kg (bw)-day, respectively [26].
2.8 Principal component analysis
The principal component analysis (PCA) assessed the multivariate relationships and variances among the elements after the Varimax rotated data [27]. Principal components having eigenvalues of 1.0 or higher were considered. The PCA allows a considerable reduction in the number of variables and the detection of structure in the relationships of different variables.
3 Results and discussion
3.1 Total metal concentration
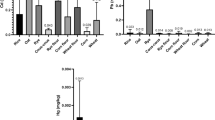
Table 1 presents the statistical analysis of the concentrations of the metals (As, Cd, Cr, Mn, Ni, and Pb) in food samples (maize, oats, millet and sorghum, rice, and wheat) analyzed. The mean As concentration ranged from 0.59 ± 0.03 mg/kg in oats to 0.69 ± 0.08 mg/kg in millet and sorghum. The As concentrations in all the five cereal classes were above the permissible limits of 0.2 mg/kg for cereal-based foods [28]. Increased exposure of human beings to arsenic from food causes cancer, diabetes and cardiovascular diseases, increased mortality in young adults and negative impacts on cognitive development in children [11, 29].
The concentration of Pb showed a minimum of 0.83 ± 0.03 mg/kg in oats samples and a maximum of 0.92 ± 0.09 mg/kg in millet and sorghum samples. The maximum permissible limit of Pb in cereal-based products is 0.2 mg/kg [11, 29], which is lower than what was reported in this study, raising concerns about the potential health risks associated with the consumption of these products. In a study from Brazil, a mean Pb concentration of 0.36 mg/kg was reported for rice-based meals [30]. A higher Pb concentration of 2.2177 mg/kg and 2.2780 mg/kg was reported in maize and millet samples, respectively, from the Tolon District of Ghana [31]. Lead is a persistent, bio-accumulative and toxic element. The presence of Pb in cereal-based breakfast is of great concern since it can bioaccumulate in small quantities and cause cardiovascular diseases in adults and neuro-developmental disorders in children [32].
Among the five cereal-based classes, the mean Cd concentration ranged from 1.27 ± 0.08 to 1.41 ± 0.13 mg/kg. The maximum permissible level of Cd in cereal foods is 0.1 mg/kg [11]. In Iran [34], a Cd concentration of 0.41 mg/kg was found in wheat cereal. Frequent ingestion of developmental toxicants, such as Cd-contaminated meals, has been linked to osteoporosis and lung cancer, which are more common in adults [35].
The mean Cr content in the current study ranged from 4.66 ± 2.08 to 9.85 ± 10.66 mg/kg. The mean Cr concentration of 5.06 mg/kg in rice-based and 5.29 mg/kg in wheat-based samples in this study were higher than Cr levels in rice (0.024 mg/kg) and wheat cereals (0.021 mg/kg) in Beijing, China [36]. The mean Cr concentration in wheat-based (5.29 mg/kg) and maize-based samples (4.66 mg/kg) in this study were higher than those reported (0.43 mg/kg for wheat and 0.52 mg/kg in maize, respectively) in Ambo City, Ethiopia [37]. The WHO has not evaluated permissible limits for Cr [11]. The Chinese authorities have set the standard residual level for cereal-based products to 1 mg/kg [33], which was lower in all five cereal-based food samples. Oral Cr exposure has been linked to chronic kidney disease, stomach ulcers, bone tissues, liver cancer, and oral cancer [38, 39].
The mean Ni in this study ranged from 5.01 ± 0.29 to 5.81 ± 1.02 mg/kg. There are currently no maximum limits for nickel as a food contaminant in the European Union’s regulations [40]. A precise nickel migration restriction of 0.02 mg/kg has been set for natural mineral water, drinking water, food, and food simulant from plastic materials and items, which are included in Commission Regulation (EU) No 10/20116 [40]. The value reported in this study for Ni in wheat and oat cereal exceeds the specific ingestion limit of 0.02 mg/kg for food and drinking water [41], although a previous study [42] found mean Ni levels of 0.27 mg/kg and 2.53 mg/kg in wheat and oat cereal, respectively.
Nickel is a transition metal that naturally occurs in the environment and can be found in various food sources. However, excessive exposure to high levels of Ni can potentially lead to adverse health effects, particularly for individuals sensitive or allergic to nickel. Regular oral exposure to high levels of Ni has been linked to heart disease, gastrointestinal disease, cancer, neurological and thyroid disorders in humans [43].
The values for Mn in the breakfast meals analyzed in this study were 8.26 ± 0.82 mg/kg (in oats-based cereals) and 9.73 ± 1.51 mg/kg (in millet and sorghum). Currently, no guidelines exist on maximum residual limits for Mn in foods. Breakfast cereal products in Poland [44] had Mn concentration of 21.30 mg/kg, higher than what was reported in this study. Manganese is an essential metal needed in trace amounts in biological systems for blood sugar regulation, immune function, digestion, and blood coagulation [6, 45].
The variation in metal concentrations between the five processed cereal-based foods analyzed in this study and other studies could be due to the differences in environmental factors, including contamination and pollution of soil with metals, sources of raw materials and processing technologies such as the addition of additives and food dyes. The release of metal from machinery food processing may contribute significantly to metal contamination. A study conducted in Nigeria [46] revealed that milling cereal-based foods using milling plates led to the release of significant amounts of Cr, Pb, and Ni into the food. Findings like this highlight a potential risk to human health associated with consuming contaminated cereal-based foods and emphasize the need for proper quality control measures during the milling process and adherence to recommended guidelines to ensure the production of safe and non-toxic food products.
Cereals are a common dietary source of Cd and Ni exposure, particularly for vegetarians whose diets include a high intake of cereals, nuts, oilseeds, and pulses. To protect consumers from unnecessary exposure, the European Union has established stringent regulatory thresholds for cadmium and nickel levels in food products [47]. Individuals should limit their intake of cereals known to contain high levels of cadmium and nickel to reduce potential health risks associated with heavy metal exposure.
3.2 Multivariate relationships and variances among the elements
The principal component analysis performed identified two principal components that account for 89.1% of the total variability in the analysed potentially toxic elements (Supplementary Table S1). The two principal components denote two sources of heavy metals in the analysed cereals. The first principal component, PC1, with an eigenvalue of 7.96 accounting for 79.7% of the total variance, is almost equally positively loaded with As, Cd, Ni, Mn and Pb but negatively loaded with Cr. The contribution of Cr to the variance is not significant. The PC1 source could be the contribution of natural soils on which the cereals were grown [48].
The second principal component, PC2, which has an eigenvalue of 0.94 ≈ 1, accounts for 9.4% of the total variability. It is negatively loaded heavily with Cr and Mn. The PC2 loaded positively with As, Cd, and Ni almost equally. This PC2 may be an addition to the manufacturing, processing and packaging of the cereals [49].
3.3 Bioaccessibility
Metal concentrations in most of the in vitro extracts were below the limits of detection as follows; As (< 1.7 µg/L), Cd (< 2.2 µg/L), (Cu < 1.0 µg/L), Ni (< 2 µg/L), Pb (< 1.4 µg/L). The low concentrations in the extracts could be attributed to the relatively low total metal concentrations in the cereals. Bioaccessibility was therefore not calculated, and relative bioavailability was assumed to be 1 in the risk calculations.
3.4 Estimated dietary intake (EDI)
Table 2 compares the exposure profiles for the various age groups to the toxic metals under consideration with the oral reference dose. The mean EDI indicates high exposure in the order; toddlers (1–3 years) > infants (6–12 months) > adults (18–65 years) > adolescents (10–17 years) > elderly (65–75 years) > children (3–9 years). This raises concerns about the potential health consequences of such exposure, as children and infants are more vulnerable to the adverse effects of toxic substances. These findings highlight the importance of targeted efforts and interventions to reduce exposure and mitigate health risks, particularly among the most vulnerable age groups.
All age groups are exposed to Cd levels above the PTMDI of 10–3 mg/kg (bw)/day. The exposure of the various age groups to Cd in wheat is in the order toddler > infant > adolescent > adult > elderly > children. The EDI of Cd in wheat cereal-based samples varies by age group, ranging from 3.72 × 10–3 mg/kg (bw) in children to 13.59 × 10–3 mg/kg(bw) in toddlers. Toddlers had an average Cd EDI of 12.54 × 10–3 mg/kg (bw)/day in rice, 12.14 × 10–3 mg/kg (bw)/day in oats, and 13.43 × 10–3 mg/kg (bw)/day in millet and sorghum samples. The lowest EDI of Cr was recorded in children, and the highest was recorded in toddlers, indicating that toddlers are at high risk of Cr exposure.
The highest mean EDI of As [6.55 × 10–3 mg/kg (bw)/day] was recorded in toddlers, suggesting that toddlers are at high risk of As exposure. A study [50] found the mean EDI for As in rice, wheat, and maize were 0.21 ± 0.17, 0.015 ± 0.001, and 0.017 ± 0.012 mg/kg (bw)/day, respectively. These findings indicate varying levels of arsenic exposure through the consumption of these cereal crops, with rice showing a potentially higher risk of arsenic intake. The results underscore the need for further analysis and evaluation to assess the safety implications of these arsenic levels and their potential impact on human health.
The mean EDI of Mn in all the age groups ranged from 26.55 × 10–3 to 84.8 × 10–3 mg/kg (bw)/day. All age groups are exposed to Mn levels higher than the PTMDI of 0.36 mg/kg (bw)/day at this mean exposure. The EDI of Pb to adults in maize was 3.29 × 10–3 mg/kg (bw)/day. All EDI values reported in this work are higher than those earlier reported [31] but bear only a passing resemblance to those reported in Bangladesh [51].
3.5 Hazard quotient (HQ)
Table 3 presents the mean HQ values for the cereal-based food samples for the different age groups. Metals (including Cr, Mn and Pb) in all the five cereal-based food samples had their HQ values below 1, suggesting low non-carcinogenic health risks among the various age groups [53]. However, infants, toddlers, children, adolescents, adults, and older people may have an adverse health risk of Ni and Cd exposure. The HQ of Cd and Ni in all samples was greater than one, indicating a potential high adverse health effect, particularly in toddlers and infants with the highest mean HQ values.
Continuous intake of cereals with high amounts of Cd and Ni may provide possible health hazards such as neurological, liver, kidney, respiratory, and cancer-related issues, especially in vulnerable groups such as toddlers and infants. Cadmium and nickel are toxic metals that can accumulate in the body over time and cause serious health problems. Cadmium is primarily toxic to the kidneys and can cause renal failure. Long-term exposure to cadmium has also been linked to an increased risk of cancer, osteoporosis, and cardiovascular disease. Nickel, on the other hand, is a known carcinogen and can cause lung cancer, nasal cancer, and other respiratory problems when inhaled. The severity of the non-cancer adverse health risk to the various age groups was as follows: toddler > infant > adolescent > adult > elderly > children.
3.6 Cancer risk (CR)
Carcinogenic health risks—the incremental probability of a person developing cancer at some point in a lifetime [54]—were assessed for As, Cd, and Pb for the various age groups (Table 4). The cancer risk value of 10–5 due to Pb exposure is below the USEPA threshold, implying that cancer incidence is unlikely across all age groups. The mean cancer risk values of 10–3 due to As, Cd and Pb exposure, as found in Table 4, were above the USEPA threshold level, the de minimus (10–6) across all age groups, with toddlers having the highest cancer risk.
4 Conclusion
The concentrations of the metals (As, Cd, Cr, Ni, Mn and Pb) analyzed in cereal-based foods (wheat, rice, oat, maize, and millet and sorghum) exceeded FAO/WHO limits. The estimated HQ values for Cr and Mn in all 5 cereal-based foods were below 1, suggesting a low possible adverse health risk among the various age groups. However, the HQ values for Cd and Ni were above 1, indicating a possible adverse health effect among the various age groups, especially in toddlers. Results from the study show that Pb in cereal-based food analyzed was within the acceptable limit of cancer risk to the consumer. However, the mean cancer risk values for AS, Cd, and Pb were above the acceptable range of cancer risk, the de minimus (10–6) suggesting a possible cancer health effect which raises public health concerns. As the actual health risk situations could be higher than the modelled values, this study highlights the necessity for ongoing monitoring and management of cereal-based breakfast consumption, especially for the presence of Cd and Ni, to ensure the well-being of consumers.
Because soil is the primary source of metals in cereal-based foods, the study's findings raise concerns about metal contamination and soil pollution caused by human activities in the country. This highlights the importance of continuous environmental monitoring and management, particularly in the presence of Cd and Ni, to ensure consumer well-being. We recommend a more extensive study that will consider a broader range of breakfast cereals and metals because other metals or contaminants may be present in cereal-based breakfasts that were not considered in this study. A more comprehensive understanding of the potential health risks would result from assessing a broader range of contaminants. While the study identifies potential health risks associated with metal exposure in cereal-based breakfasts, it does not establish a direct link between metal intake and adverse health effects. Other factors such as individual susceptibility, overall diet, and lifestyle must be considered to determine the actual health risks posed by these metals.
Data availability
All data obtained during this study are available to the corresponding author upon request.
References
Bangar SP, Kaushik N. Functional cereals: functional components and benefits. In: Punia Bangar S, Kumar Siroha A, editors. Functional cereals and cereal foods. Cham: Springer International Publishing; 2022. p. 3–25.
Yalçın Tepe A. Toxic metals: trace metals – chromium, nickel, copper, and aluminum. In: Motarjemi Y, editor. Encyclopedia of food safety. Amsterdam: Elsevier; 2014. p. 356–62.
Dodd M, Richardson GM, Wilson R, Rencz A, Friske P. Elemental concentrations and in vitro bioaccessibility in Canadian background soils. Environ Geochem Health. 2017;39:759–77. https://doi.org/10.1007/s10653-016-9846-9.
Arciszewska Ż, Gama S, Leśniewska B, Malejko J, Nalewajko-Sieliwoniuk E, Zambrzycka-Szelewa E, Godlewska-Żyłkiewicz B. The translocation pathways of rare earth elements from the environment to the food chain and their impact on human health. Process Saf Environ Prot. 2022;168:205–23. https://doi.org/10.1016/j.psep.2022.09.056.
Fu Z, Xi S. The effects of heavy metals on human metabolism. Toxicol Mech Methods. 2020;30:167–76. https://doi.org/10.1080/15376516.2019.1701594.
Jomova K, Makova M, Alomar SY, Alwasel SH, Nepovimova E, Kuca K, Rhodes CJ, Valko M. Essential metals in health and disease. Chem Biol Interact. 2022;367:110173.
Elder A, Nordberg GF, Kleinman M. Exposure, dose, and toxicokinetics of metals. In: Nordberg GF, Costa M, editors. Handbook on the toxicology of metals. Amsterdam: Elsevier; 2022. p. 55–86.
Naranjo VI, Hendricks M, Jones KS. Lead toxicity in children: an unremitting public health problem. Pediatr Neurol. 2020;113:51–5. https://doi.org/10.1016/j.pediatrneurol.2020.08.005.
Deubler EL, Gapstur SM, Diver WR, Gaudet MM, Hodge JM, Stevens VL, McCullough ML, Haines LG, Levine KE, Teras LR. Erythrocyte levels of cadmium and lead and risk of B-cell non-Hodgkin lymphoma and multiple myeloma. Int J Cancer. 2020;147:3110–8. https://doi.org/10.1002/ijc.33136.
Ma Y, Ran D, Shi X, Zhao H, Liu Z. Cadmium toxicity: a role in bone cell function and teeth development. Sci Total Environ. 2021;769:144646.
Muzaffar S, Khan J, Srivastava R, Gorbatyuk MS, Athar M. Mechanistic understanding of the toxic effects of arsenic and warfare arsenicals on human health and environment. Cell Biol Toxicol. 2023;39:85–110. https://doi.org/10.1007/s10565-022-09710-8.
Du F, Yang Z, Liu P, Wang L. Accumulation, translocation, and assessment of heavy metals in the soil-rice systems near a mine-impacted region. Environ Sci Pollut Res. 2018;25:32221–30. https://doi.org/10.1007/s11356-018-3184-7.
Mousavi Khaneghah A, Fakhri Y, Nematollahi A, Pirhadi M. Potentially toxic elements (PTEs) in cereal-based foods: a systematic review and meta-analysis. Trends Food Sci Technol. 2020;96:30–44. https://doi.org/10.1016/j.tifs.2019.12.007.
Intawongse M, Dean JR. In-vitro testing for assessing oral bioaccessibility of trace metals in soil and food samples. TrAC Trends Anal Chem. 2006;25:876–86. https://doi.org/10.1016/j.trac.2006.03.010.
Akanchise T, Boakye S, Borquaye LS, Dodd M, Darko G. Distribution of heavy metals in soils from abandoned dump sites in Kumasi. Ghana Sci African. 2020;10:e00614. https://doi.org/10.1016/j.sciaf.2020.e00614.
Darko G, Boakye KO, Nkansah MA, Gyamfi O, Ansah E, Yevugah LL, Acheampong A, Dodd M. Human health risk and bioaccessibility of toxic metals in topsoils from Gbani mining community in Ghana. J Heal Pollut. 2019;9:190602. https://doi.org/10.5696/2156-9614-9.22.190602.
Darko G, Dodd M, Nkansah MA, Aduse-Poku Y, Ansah E, Wemegah DD, Borquaye LS. Distribution and ecological risks of toxic metals in the topsoils in the Kumasi metropolis. Ghana Cogent Environ Sci. 2017;3:1354965. https://doi.org/10.1080/23311843.2017.1354965.
Ghana Statistical Service (GSS). Ghana 2021 population and housing census. Ghana Stat Serv. 2021;3A:112.
da Silva YJAB, do Nascimento CWA, Biondi CM. Comparison of USEPA digestion methods to heavy metals in soil samples. Environ Monit Assess. 2014;186:47–53. https://doi.org/10.1007/s10661-013-3354-5.
US EPA. Method 200.8 Determination of trace elements in waters and wastes by inductively coupled plasma -mass spectrometry revision 5.4 EMMC version determination of trace elements in waters and wastes by inductively coupled plasma -mass spectrometry. Ohio. 1990.
US EPA Standard operating procedure for an in vitro bioaccessibility assay for lead in soil. United States Environmental Protection Agency. 2012.
USEPA. Exposure Factors Handbook 2011 Edition (Final Report) U.S. Environmental Protection Agency, Washington, DC, EPA/600/R-09/052F, 2011.
Afrin S, Alam MK, Ahmed MW, Parven A, Jubayer MF, Megharaj M, Meftaul IM, Khan MSI. Determination and probabilistic health risk assessment of heavy metals in widely consumed market basket fruits from Dhaka city Bangladesh. Int J Environ Anal Chem. 2021;00:1–16. https://doi.org/10.1080/03067319.2021.2018578.
Laboni FA, Ahmed MW, Kaium A, Alam MK, Parven A, Jubayer MF, Rahman MA, Meftaul IM, Khan MSI. Heavy metals in widely consumed vegetables grown in industrial areas of Bangladesh: a potential human health hazard. Biol Trace Elem Res. 2023;201:995–1005. https://doi.org/10.1007/s12011-022-03179-6.
Morakinyo OM, Mukhola MS, Mokgobu MI. Health risk analysis of elemental components of an industrially emitted respirable particulate matter in an urban area. Int J Environ Res Public Health. 2021;18:3653. https://doi.org/10.3390/ijerph18073653.
Fairbrother A, Wenstel R, Sappington K, Wood W. Framework for metals risk assessment. Ecotoxicol Environ Saf. 2007;68:145–227. https://doi.org/10.1016/j.ecoenv.2007.03.015.
Bux RK, Batool M, Shah SM, Solangi AR, Shaikh AA, Haider SI, Shah ZH. Mapping the spatial distribution of soil heavy metals pollution by principal component analysis and cluster analyses. Water Air Soil Pollut. 2023;234:1–13. https://doi.org/10.1007/S11270-023-06361-1/METRICS.
FAO/WHO. Joint FAO/WHO food standards programme codex committee on contaminants in foods. Rome: Codex Alimentarius Commission; 2011. p. 8–13.
Kuo CC, Moon KA, Wang SL, Silbergeld E, Navas-Acien A. The association of arsenic metabolism with cancer, cardiovascular disease, and diabetes: a systematic review of the epidemiological evidence. Environ Health Perspect. 2017. https://doi.org/10.1289/EHP577.
Pedron T, Segura FR, da Silva FF, de Souza AL, Maltez HF, Batista BL. Essential and non-essential elements in Brazilian infant food and other rice-based products frequently consumed by children and celiac population. J Food Compos Anal. 2016;49:78–86. https://doi.org/10.1016/j.jfca.2016.04.005.
Larsen K, Cobbina SJ, Ofori SA, Addo D. Quantification and health risk assessment of heavy metals in milled maize and millet in the Tolon District, Northern Ghana. Food Sci Nutr. 2020;8:4205–13. https://doi.org/10.1002/fsn3.1714.
Check L, Marteel-Parrish A. The fate and behavior of persistent, bioaccumulative, and toxic (PBT) chemicals: examining lead (Pb) as a PBT metal. Rev Environ Health. 2013;28:85–96. https://doi.org/10.1515/reveh-2013-0005.
Woolsey M, Bugang W. National Food Safety Standard-Maximum Levels of Contaminants. Global Agriculture Information Network. USDA Foreign Agricultural Service. GAIN Report Number: CH10000. Beijing. 2010.
Pirsaheb M, Fattahi N, Sharafi K, Khamotian R, Atafar Z. Essential and toxic heavy metals in cereals and agricultural products marketed in Kermanshah, Iran, and human health risk assessment. Food Addit Contam Part B. 2016;9:15–20. https://doi.org/10.1080/19393210.2015.1099570.
Chandravanshi L, Shiv K, Kumar S. Developmental toxicity of cadmium in infants and children: a review. Environ Anal Heal Toxicol. 2021;36:e2021003. https://doi.org/10.5620/eaht.2021003.
Wei J, Cen K. Contamination and health risk assessment of heavy metals in cereals, legumes, and their products: a case study based on the dietary structure of the residents of Beijing. China J Clean Prod. 2020;260:121001. https://doi.org/10.1016/j.jclepro.2020.121001.
Wodaje AT. Assessment of some heavy metals concentration in selected cereals collected from local markets of Ambo City, Ethiopia. J Cereal Oilseeds. 2015;6:8–13. https://doi.org/10.5897/JCO15.0138.
Kulathunga MRDL, Ayanka Wijayawardena MA, Naidu R, Wijeratne AW. Chronic kidney disease of unknown aetiology in Sri Lanka and the exposure to environmental chemicals: a review of literature. Environ Geochem Health. 2019;41:2329–38. https://doi.org/10.1007/s10653-019-00264-z.
Tsai TL, Kuo CC, Pan WH, Chung YT, Chen CY, Wu TN, Wang SL. The decline in kidney function with chromium exposure is exacerbated with co-exposure to lead and cadmium. Kidney Int. 2017;92:710–20. https://doi.org/10.1016/j.kint.2017.03.013.
EFSA. European food safety authority (EFSA) monitoring of nickel in food and feed. EFSA J. 2015;13:4002.
Schrenk D, Bignami M, Bodin L, Chipman JK, del Mazo J, Grasl-Kraupp B, Hogstrand C, Hoogenboom LR, Leblanc J, Nebbia CS, Ntzani E, Petersen A, Sand S, Schwerdtle T, Vleminckx C, Wallace H, Guérin T, Massanyi P, Van Loveren H, Baert K, Gergelova P, Nielsen E. Update of the risk assessment of nickel in food and drinking water. EFSA J. 2020;18:6268. https://doi.org/10.2903/j.efsa.2020.6268.
Mania M, Rebeniak M, Orshulyak O, Postupolski J. Assessment of exposure to nickel intake with selected cereal grains and cereal-based products. Rocz Państwowego Zakładu Hig. 2020;71:371–6. https://doi.org/10.32394/rpzh.2020.0142.
EFSA. Scientific opinion on the risks to public health related to the presence of chromium in food and drinking water. EFSA J. 2014;12:3595. https://doi.org/10.2903/j.efsa.2014.3595.
Winiarska-Mieczan A, Kowalczuk-Vasilev E, Kwiatkowska K, Kwiecień M, Baranowska-Wójcik E, Kiczorowska B, Klebaniuk R, Samolińska W. Dietary intake and content of Cu, Mn, Fe, and Zn in selected cereal products marketed in Poland. Biol Trace Elem Res. 2019;187:568–78. https://doi.org/10.1007/s12011-018-1384-0.
Horning KJ, Caito SW, Tipps KG, Bowman AB, Aschner M. Manganese is essential for neuronal health. Annu Rev Nutr. 2015;35:71–108. https://doi.org/10.1146/annurev-nutr-071714-034419.
Oniya EO, Olubi OE, Ibitoye A, Agbi JI, Agbeni SK, Faweya EB. Effect of milling equipment on the level of heavy metal content of foodstuff. Phys Sci Int J. 2018;20:1–8. https://doi.org/10.9734/psij/2018/42572.
European Commission. Setting maximum levels for certain contaminants in foodstuffs. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting. Off J Eur Union L. 2006;364:5–24.
Dodd M, Amponsah LO, Grundy S, Darko G. Human health risk associated with metal exposure at Agbogbloshie e-waste site and the surrounding neighbourhood in Accra, Ghana. Environ Geochem Health. 2023. https://doi.org/10.1007/s10653-023-01503-0.
Konadu FN, Gyamfi O, Ansah E, Borquaye LS, Agyei V, Dartey E, Dodd M, Obiri-Yeboah S, Darko G. Human health risk assessment of potentially toxic elements in soil and air particulate matter of automobile hub environments in Kumasi, Ghana. Toxicol Rep. 2023;11:261–9. https://doi.org/10.1016/j.toxrep.2023.09.010.
Islam MS, Ahmed MK, Habibullah-Al-Mamun M. Heavy metals in cereals and pulses: health implications in Bangladesh. J Agric Food Chem. 2014;62:10828–35. https://doi.org/10.1021/jf502486q.
Islam MS, Ahmed MK, Habibullah-Al-Mamun M, Masunaga S. Assessment of trace metals in foodstuffs grown around the vicinity of industries in Bangladesh. J Food Compos Anal. 2015;42:8–15. https://doi.org/10.1016/j.jfca.2014.12.031.
FAO/WHO. Summary Report of the 72nd Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). 2010.
Fan Y, Zhu T, Li M, He J, Huang R. Heavy Metal contamination in soil and brown rice and human health risk assessment near three mining areas in central China. J Healthc Eng. 2017;2017:1–9. https://doi.org/10.1155/2017/4124302.
Jafari A, Asadyari S, Moutab Sahihazar Z, Hajaghazadeh M. Monte Carlo-based probabilistic risk assessment for cement workers exposed to heavy metals in cement dust. Environ Geochem Health. 2023. https://doi.org/10.1007/s10653-023-01611-x.
Acknowledgements
We are grateful to the Royal Roads University, Victoria, Canada, for funding Matt Dodd’s trips to Ghana and the Sheathe Project (www.sheathe.org) for lab space and instrumentation.
Funding
This study did not receive direct funding.
Author information
Authors and Affiliations
Contributions
PAO was involved in the study’s conceptualization and design, data acquisition, and manuscript drafting. GA-B contributed to the study’s conception, design, toxicity modelling, and manuscript drafting. MD contributed to the study’s conception and design, sample analysis, and manuscript drafting. EA contributed to the sample acquisition, sample analysis, and manuscript drafting. CD contributed to the sample acquisition, sample analysis, and manuscript drafting. LSB contributed to the study’s conception and design, sample analysis, and manuscript drafting. GD contributed to the study’s conception and design, sample analysis, and manuscript drafting.
Corresponding author
Ethics declarations
Competing interests
All the authors declare that they have no known financial and personal relationships with other people or organizations that could inappropriately influence their work. All authors have read, understood, and have complied as applicable with the statement on “Ethical responsibilities of Authors”.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oduro, P.A., Ankar-Brewoo, G., Dodd, M. et al. Health risks of potentially toxic metals in cereal-based breakfast meals in the Kumasi Metropolis, Ghana. Discov Food 3, 25 (2023). https://doi.org/10.1007/s44187-023-00067-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44187-023-00067-3