Abstract
Medicinal herbs have an essential function in health and therapy. Bioactives of medicinal plants showed fewer side effects than manufactured drugs and their antioxidant properties are responsible for their different therapeutic properties. Essential oils (EOs) are an aromatic mixture of active components with a strong aroma obtained from aromatic plants. Natural EOs are composed of monoterpenes, sesquiterpenes, oxygenated monoterpenes, oxygenated sesquiterpenes, and phenolics. Because of their volatility, EOs are easily extracted from a variety of natural sources using the steam distillation method. Essential oils are one of the important therapeutic ingredients used in developing new therapeutic vehicles because of their anticancer, antiviral, antidiabetic, anticancer, antibacterial, antioxidant, and aromatherapy capabilities. This review includes different literature to survey the nature of essential oils, therapeutic potentials, and the key components of various essential oils.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Medicinal plants are an excellent source of different bioactive secondary metabolites, used in developing innovative therapeutic agents presenting novel health benefits. Saudi Arabia is an excellent source of various medicinal and aromatic plants. Many Saudi plants differ in their chemo-diversity and therapeutic effects from the same plants growing in other areas and climates because of the distinct geographical location of the country [1]. Different aromatic plants growing in Saudi Arabia have been reported as promising sources to produce EOs [2]. Aromatic plants producing high amounts of EOs can be utilized as a therapeutic option to treat different illnesses thanks to their known efficacy. EOs are a chemical mixture of aromatic components that have a strong aroma and are extracted from aromatic plants. EOs are complex combinations of bioactive ingredients with a variety of structures, such as mono-, sesqui-, and di-terpenes, phenol elements, sulfur-containing ingredients, and phenylpropanoid components [2]. The efficient antimicrobial properties of EOs may help in overcoming the problems of high bacterial resistance to current drugs and the excessive fees of recent generations of antibiotics [3]. The enormous variety of bioactive components of EOs obstructs the resistance mechanisms of various bacteria by interacting with cellular enzymes or cell structures of the invasive microorganisms, which increases the rate of microbial cell death [4]. Besides their excellent antimicrobial properties, EOs and their constituents are utilized in fragrances, cosmetics, pharmaceuticals, aromatherapy, sanitary goods, dentistry, agriculture, food preservers and additives, and natural cures. As a result, EOs became a far more appealing issue for both research and industry, with yearly profits in the billions of dollars [5]. Thus, EOs have become an interesting source for finding new, effective, and safe bioactive molecules with diverse therapeutic benefits especially antioxidant and antimicrobial effects. This study aims to point out the importance of EOs by looking at the nature of EOs, extraction methods of EOs, the antioxidant, antibacterial, and antifungal effects of EOs, as well as aromatic plants that produce EOs.
2 What are essential oils?
Essential oils are the scented liquids produced by aromatic plants' secondary metabolism. They are called "essential" since they are the plant's most critical component. EOs are mixtures of organic ingredients derived from various plant sources, they provide plants with their distinct perfume [6, 7]. Various organs of aromatic herbs are utilized to distill EOs, for example, seeds (Caraway, Cumin, and Coriander), leaves (Mint, Thyme, Sage, Rosemary, Oregano, Basil, Celery, and Parsley), fruits (Anise, Fennel, and Lemon), flowers (Rose and Rosemary), bark (Cinnamon), cloves or buds (Clove and Garlic), and rhizomes (Ginger) [6]. Aromatic plants can synthesize EOs as combinations of organic constituents in the cytoplasm and plastids of plant cells by different pathways including mevalonic acid, malonic acid, and methyl-D-erythrol-4-phosphate (MEP), they store it in epidermal cells, secreting fissure, glandular trichomes, or resin canals [8, 9]. The characteristic odor and color of EOs depend on the origins of plants, species, and organs. The common color of EOs is pale yellow or colorless, even though a few have a deep color, for instance, green European valerian and blue chamomile [6]. The volatile compounds present in Eos have an important environmental function as they can protect plants from invasive bacteria, fungi, insects, and viruses, and are also able to attract certain insects for plant pollination [5]. Plant essential oils have different chemical contents based on geographical location, environment, and stage of maturity are all factors that can affect plants and the percentage of essential oils in them [10, 11].
3 The nature of essential oils
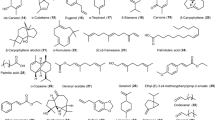
Terpenes and phenylpropanoids are two characteristic chemical groups that make up most plant EOs. Terpenes and terpenoids are major components of various EOs, however, in some species, phenylpropanoids represent the major ingredients of their EOs, which provide a particular scent and flavoring to such oils [6, 12, 13]. The key elements of EOs are derived through three chemical routes: mono- and diterpenes are synthesized through the methylerythritol route, sesquiterpenes are synthesized through the mevalonate route, and phenylpropenes are synthesized through the shikimic acid route [12]. The distinct secondary metabolites (terpenes, aldehydes, ketones alcohols, esters, and phenylpropanoids) of a plant are the main key affecting the chemical characteristics of each plant’s EOs [14]. The condensation of several isoprene units forms different terpenes, such as mono-, di-, sesqui-, tri-, tetra-, and poly-terpenes. Figure 1 presented p-cymene, limonene, sabinene, β-myrcene, and γ-terpinene as examples of monoterpenes [15]. The chemical structure of terpenes is changed by some enzymes forming varied terpenoids with varied chemical structures and different positions of hydroxyl groups (Fig. 1), such as menthol, carvacrol, α-terpineol, geraniol, and thymol [16]. Phenylpropanoids, such as cinnamaldehyde, safrole, eugenol, and isoeugenol, are found in low contents in most EOs (Fig. 1) [5, 17]. EOs chemical ingredients and concentrations are different amongst plant species, and within similar species, because of differences in plant part, harvesting time, drying, storage, distilling process, and climatic issues [18]. Chemical qualities of EOs differ in the concentration and kind of ingredient present and their stereochemical structures, which can be influenced by the extraction method used. Essential oils are natural mixes of organic compounds, containing 20–100 different volatile compounds of different chemical classes, only two or three molecules, found with high percentages (20–70%) are responsible for the distinct characterizations of EOs [19]. In this consensus, Bakkali et al. [5] demonstrated that the key components in Origanum EO were carvacrol and thymol with high percentages of 30% and 27% respectively, and the uppermost percentage (68%) of linalool was detected in Coriandrum EO. The high percentage (50%) of 1,8-cineole was a characteristic feature of Cinnamomum EO, and EO of Artemisia was characterized by high contents (57%) of α- and β-thuyone in addition to 24% of camphor. Also, EO of Mentha was distinguished by high contents (59%) of menthol along with 19% of menthone, while 36% of α-phellandrene and 31% of limonene were detected in EO of Anethum leaves and 58% of carvone and 37% of limonene were found in EO of Anethum seeds [5]. Moreover, methyl carvacrol (13.4%), carvacrol (55.7%), δ-3-carene and β-bisabolene (9.1%) are the major component of Lavandula pubescens oil, linalool (33.0%) is the major component of Pulicaria incisa, and δ-3-carene (7.3–30.3%) and α-pinene (31.3–62.5%) are the major component of Juniperus procera [2].
The GC–MS is one of the most used chromatographic systems to separate and identify EOs different ingredients. The MS uses information of the unique fragmentation pattern of each separated component in EOs within the applied ionizing mode to identify the chemical structure of the molecule [6].
4 Extraction of essential oils
There are two main steps that are applied to analyze plant EOs: extraction/distillation of the oil, which takes several hours, and chemical analysis, which takes several minutes. Various extraction/distillation procedures are utilized to refine plant EOs, Figure 2explains it following the previous report of Farhat et al. [20]. Thanks to the volatile nature of EOs, the hydro-distillation using the Clevenger system is the conventional system applied to refine EOs on the laboratory scale. While steam distillation is the conventional system applied to refine EOs in industrial productions [21]. The extraction of EOs using solvents is markedly applied in industrial practices, however, it is restricted in food industries because of the remarkable toxicity of the applied organic solvents [22]. Other different techniques have been checked for EOs extraction in the industry to improve the efficacy, sustainability, and economy of the applied system, this includes the assisted extraction with microwave and ultrasonic, along with ohmic hydro-distillation system [18, 23]. The extraction method used is the most significant factor in assuring the quality of essential oils, as improper extraction methods can change the chemical compositions of aromatic oils and alter their quality and function [18]. Also, if the steam distillation process is used for EOs extraction, the derived chemicals will always be volatile, while if solvents are used for Eos extraction, the chemical composition will differ from that of a similar essential oil obtained through distillation. Because the chemical composition of any oil is affected by the used extraction method, selecting the proper extraction method based on the characteristics of each plant material is a critical point. The annual extraction of EOs should be made under the same conditions to maintain consistency in chemical composition, quality, and quantity, such as using similar plant parts, a similar extraction process, and a similar harvesting season. Plant parts collected for extraction can be picked fresh, partially dried, or dehydrated, but flowers must be selected fresh [24].
5 Biological activities of essential oils
Since the earliest times, various aromatic herbs have been utilized as preservatives and curative agents and as the main source of aroma and flavor in the food industry. EOs are the main cause of the therapeutic effects in various aromatic herbs. The varied biological effects of EOs are not only attributed to their key compounds (the two or three compounds found at high percentages) but also may be to the strong effect of synergisms of other active ingredients [25]. Essential oils are utilized in pharmaceutics mostly for aromatherapy and to improve the sensory properties of pharmacological medications. Different traditional systems globally use EOs for curing numerous health complications. For example, EO of Eucalyptus to cure bronchitis and coughing, EOs of Sage and Clove to inhibit the growth of different bacterial, Peppermint EO to cure any respiratory congestion, and EOs of Anise and Peppermint are well known as carminatives [26, 27]. Different experimental studies reported different therapeutic properties of EOs and their key components (Fig. 3), as reported in Bakkali et al. [5].
5.1 Essential oils as antioxidants
The powerful antioxidant effect of different EOs has been reported in many studies because of their nontoxic effect on health. Recently, different reports encouraged the use of natural antioxidants including EOs, because of the toxic effect of the use of unnatural antioxidants on human health, including butylated hydroxy anisole (BHA) and butyl hydroxytoluene (BHT) [28]. The excellent antioxidant compound can transfer an electron to an active free radical to deactivate it, thus making it more stable and less dangerous. Antioxidants protect cellular organs from oxidative deterioration by hunting any free radicals [29]. Various studies have used several in vitro chemical tests to assess the antioxidant power of EOs. The diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS +) are the commonly used assays, in which stable radicals (DPPH and ABTS+) were utilized as probes to measure the scavenging ability of free radicals. In the DPPH assay (Fig. 4), Electron/hydrogen-donating antioxidant compounds in EOs donate an electron to the DPPH radical, this changes the DPPH color from purple to colorless depending on the antioxidant strength of the compound. The degree of color change is calculated as absorbance readings at 519 nm wavelength using UV spectra, these absorbance values represent the antioxidant activity, and a high absorbance value means a high antioxidant activity [30].
The extra reactive oxygen species and excess free radicals produce a case of oxidative stress, in which different macromolecules are damaged in the cell, causing numerous health ailments such as aging, cancer, diabetes, arteriosclerosis, and Alzheimer’s disease [31]. Too many free radicals in the body cause cell damage, which has been linked to chronic disorders like heart disease and cancer. Different natural antioxidants including EOs bioactives can control the production of excess free radicals in the body by their potent free radical scavenging abilities [32]. The antioxidative potential of volatile oils from different aromatic plants against DPPH and ABTS was investigated. Such as, the Eos of Origanum, Tagetes, and Bacopa showed prominent antioxidant powers [33]. The EOs of Thyme and Savory presented excellent DPPH and ABTS free radical scavenging abilities, the ABTS scavenging activity of EOs was significantly higher than that of DPPH [34]. The Eos of different species of Mentha, Salvia, Curcuma, Achillea, Melaleuca, and Ocimum exhibited powerful free radical scavenging/ antioxidant capabilities [35, 36]. In another study, Romeilah et al. [37] demonstrated the effective free radical scavenging abilities of Petroselinum, Coriandrum, Cuminum, and different species of Allium. Furthermore, Tomaino et al. [38] evaluated the radical-scavenging and antioxidant efficacy of several EOs, and they found that the EOs of Clove was the highest followed by Cinnamon, Nutmeg, Basil, Oregano, then Thyme. Likewise, many investigations assessed the antioxidant capabilities of the key ingredients of different EOs, for instance, Miguel [32] stated that thymol, the key component in Thymus EOs, and carvacrol, the key component in Origanum EOs exhibited outstanding antioxidant abilities.
5.2 Essential oils as antibacterial agents
The rising occurrence of antibiotic resistance, which leads to antimicrobial treatment inadequacy, is a major challenge in antimicrobial chemotherapy. The most important factor contributing to the development of diverse types of resistant bacteria is assumed to be the overuse of antibiotics and the resulting antibiotic selection pressure. Plant bioactives including EOs exhibited powerful antibacterial capacities against a variety of Gram-negative combined with Gram-positive bacteria [31, 39]. Because of their potent antibacterial activities, different EOs and their key molecules attained high awareness, which can be used as diverse types of useful additives to increase the storage period of food ingredients to secure consumer microbiological safety [40]. The powerful antimicrobial capabilities of abundant EOs have been established, these capabilities are commonly affected by the presence and concentration of some ingredients in EOs including phenylpropanoids, terpenoids, alcohols, terpenes, aldehydes, ketones, and esters [14, 25, 41]. The EOs showed various biochemical mechanisms against different microorganisms, including the ability of hydrophobic elements in EOs to interact with the lipids of microorganisms’ cell membranes, which resulted in damage to membrane permeability and solidity, which lead to high fluctuations in the chain of the electrons transport, in nutrients uptakes, and in the synthesis of nucleic acids and proteins, consequently, induces clotting of cellular contents and inhibiting different metabolite enzymes, these cause cells death [42]. After the disruption of bacterial membranes, EOs bioactives as well can deactivate bacterial cells by entering the cell and obstructing protein, RNA, DNA, or polysaccharides [27]. Various EOs including Thyme, Cinnamon, Oregano, Clove, Lemongrass, and Rosewood showed strong antibacterial capabilities [27]. The antibacterial efficacy of different EOs against E. coli remarkably depends on the concentration, the highest activity of Bay EOs was at 0.02%, Clove at 0.04%, oregano at 0.05%, lemon grass at 0.06%, and thyme at 0.05%. While EOs of Peppermint, Rosemary, Clove, Bay, Lemon grass, and Thyme showed remarkable antibacterial capability against S. aureus at ≤ 0.05% concentration. Similarly, the concentration of 1% of Eucalyptus and Basil EOs presented mild antibacterial effects [43]. Moreover, the powerful antibacterial powers of tea tree, lemon myrtle, and garlic EOs versus methicillin-resistant S. aureus (MRSA) have been stated [44]. The antibacterial effects of clove, cinnamon, tamarind, black cumin, nutmeg, garlic, onion, and pomegranate seeds EOs versus Salmonella enteritidis as well as Listeria monocytogenes have been stated [45]. Different wild Fennel fruits EOS, collected from Portugal, exhibited low to mild antibacterial effects versus S. enteritidis, E. coli, P. aeruginosa, and P. mirabilis [46]. Different key components of EOs showed antimicrobial capacity more than their crude oils, key molecules such as eugenol and carvacrol in Clove oil as well as terpinen-4-olin in tea-tree oil, demonstrated superior antimicrobial ability compared to their crude EOs [47]. The excellent antimicrobial capabilities of various phenylpropanoids including eugenol, cinnamaldehyde, isoeugenol, and safrole have been demonstrated [17]. Different investigations discussed various modes of action of various EOs against various bacteria, such as Behbahani et al. [48] stated that the application of Cumin EOs against Listeria innocua and E. coli negatively affected the permeability of bacterial cell membranes which led to the discharge of cell components and cell death. Similarly, the EO of Trachyspermum copticum showed an effective antimicrobial activity versus E. coli by damaging the cell membrane integrity by forming many pores and then cell lysis [49]. As well, the treatment of S. aureus with Artemisia argyi EO caused an increase in bacterial cytoplasmic membrane permeability which caused the leakage of proteins and K+ ions and cell death [50]. Alternatively, treating L. monocytogenes with Citrus chagshan – huyou EOs as well as carvacrol revealed different morphological modifications in the bacterial cells including fissures, crumpled, and collapsing sides, and fragmented cells [51, 52].
5.3 Essential oils as antifungal agents
Different aromatic plants and essential oils showed potent antifungal effects against different pathogenic fungi, including yeasts. The antifungal efficiency of EOs depends on the target pathogen and the applied oil. The volatile oils of Fennel, Coriander, and Anise presented good antifungal effects against Candida albicans at different concentrations of 1%, 0.5%, and 0.25% respectively [43]. The volatile oils of Geranium, Japanese mint, Cinnamon, Clove, Ginger grass, and Lemongrass at varying concentrations (0.01–0.15%) revealed strong antifungal activities versus C. albicans [53]. Lavandula multifida volatile oil also exhibited a potent antifungal ability versus C. albicans [13]. The Cymbopogon sp. EO revealed excellent antifungal ability versus pathogenic yeasts [54]. Eugenol, a phenylpropanoid molecule, and β-bisabolol, a monocyclic sesquiterpene alcohol, exhibited excellent antifungal abilities against various dermatophytes and their spores [55]. The Lemon grass volatile oil showed the greatest antifungal activity against Aspergillus flavus at concentrations of (0.006–0.03%) by inhibiting the fungal growth and aflatoxin production [39]. The antifungal abilities of Lemon, Mandarin, Grapefruit, and orange oils at a concentration of < 1% were reported against different species of Aspergillus and Penicillium molds [56]. Different EOs and their ingredients showed the most antifungal effects versus C. albicans drug-resistant biofilms by suppressing membrane ergosterol and varied signaling paths that prevent hyphal morphogenesis from the yeast [57]. Moreover, citral (key molecule of eucalyptus oil), citronellol (key molecule of tea tree oil), and geraniol and geranyl acetate (key molecules of geranium oil) considerably remarkably suppress the cell cycle of C. albicans at the S phase [58]. Similarly, carvacrol, eugenol, and thymol suppress S. cerevisiae growth by disrupting ions homeostasis of Ca2+ and H+ [59].
6 Different important medicinal plants producing essential oils
Medicinal herbs have been employed in traditional prescriptions by African and Asian civilizations for thousands of years. In both developing and developed countries, there has been a remarkable increase in public interest and acceptance of natural therapies during the last few decades. Herbal medicinal items are used by up to four billion people (representing 85 percent of the world's population) as an alternative to traditional medicines. Furthermore, medicinal plants constitute around 25% of all modern medicines, indirectly or directly [60].
Therapeutic characteristics of EOs isolated from medicinal and aromatic plants have diverse applications, according to research on manufactured pharmaceutical compounds. As a result, farmers and researchers have been driven to develop these compounds [61]. EOs produced by medicinal plants are typically found in warm climates [11]. Out of 3000 identified EOs, which are made by plants of various genera, only 300 EOs are of promising commercial value. Numerous plants of various families can synthesize high percentages of EOs and produce them in commercial amounts, such families are Alliaceae, Lamiaceae, Myrtaceae, Apiaceae, Asteraceae, Rutaceae, and Poaceae. These commercial amounts of EOs are utilized in many industries, mainly in cosmetics, aromatherapy, foods, pharmaceuticals, agronomics, and polishing [5, 25, 62]. Orange, basil, maize mint, lemon, camphor, citronella, eucalyptus, clove, and eucalyptus oils are among the most widely used essential oils of medicinal significance [36, 63]. Some important medicinal plants producing essential oils and the key components of their essential oils were presented in Table 1.
6.1 Fennel (Foeniculum vulgare Mill)
Fennel (Fig. 5), or Foeniculum vulgare (Family: Apiaceae), is a well-known important medicinal and fragrant herb that is widely used as a carminative, digestive, lactogogue, and diuretic, as well as in the therapy of respiratory and gastric disorders. Baked items, meat and seafood meals, ice cream, alcoholic beverages, and herb mixes all use their seeds as flavorings [64]. However, F. vulgare essential oil has potent effects against different microbes and insects, and it can be applied in the food industry as a natural preservative for various food sources and products against microbes and pests. In addition to their antifungal properties against human pathogenic fungi, Fennel has a high ability to inhibit the growth of phytopathogenic fungi [65]. Fennel is one of the popular aromatic herbs used in Iranian folk medicine as an antioxidant, antimicrobial, anti-inflammatory, cytotoxic, galactagogue, diuretic, lithotriptic, estrogenic, emmenagogue, hepatoprotective, hypotensive, gastroprotective, antithrombotic, antimutagenic and memory enhancing activities agent, after consuming F. vulgare, no serious side effects were observed [66].
6.2 Dill (Anethum graveolens)
Dill, scientifically known as Anethum graveolens, is one of the most prevalent herbaceous plants belonging to the family Apiaceae (Fig. 6). It is one of the annual herbs grown in different regions worldwide. It is used as an herb or spice for food flavor, seeds and leaves are the main parts used for this purpose. Various parts of the Dill plant including seeds, leaves, and stems can be used to distillate the volatile oil. The chemical constituent of volatile oils distilled from Dill seeds, flowering herbs, and vegetative herbs was investigated [67], and they found that Dill seeds oil contains two key molecules of phellandrene (46.33%) and limonene (13.72%), while phellandrene at 11.01% and p-cymene at 17.88% were the key molecules in the oil of Dill vegetative herbs, and the key ingredients of Dill flowering herbs were dill-ether (19.63%), p-cymene (33.42%), and carvone (13.10%). Dill essential oil showed potent antioxidant along with antibacterial capability against Staphylococcus aureus [68], as well as a potent antifungal effect against Saccharomyces cerevisiae [69]. The Dill extracts and volatile oil with their diverse bioactive ingredients exhibited various therapeutic properties including anti-hypercholesterolaemic, antidiabetic, anticancer, antioxidant, anti-inflammatory, insecticidal, antimicrobial, and antispasmodic [70].
7 Conclusions
Essential oils, among natural plant products, need special attention because they are used in a variety of traditional therapeutic systems all over the world. This review attempts to spotlight the nature of volatile oils, their beneficial properties, their ingredients, and various aromatic herbs that produce volatile oils. The findings discussed in this review are proposed to attract the interest of investigators looking for new natural and effective agents working as antimicrobials for emerging resistant microorganisms, in addition to those interested in the therapeutic variety of volatile oils, particularly for treating oxidative stress ailments. The present data refreshes the knowledge of different modes of action of volatile oils as antimicrobials, which will help in new uses in essential oil therapy. Consequently, essential oils and their bioactive ingredients should be subjected to more clinical experiments, to help in developing new therapeutic products from it.
Data availability
Data will be made available on reasonable request.
References
Hassan SA, Al-Thobaiti AT (2015) I. Morphological nutlet characteristics of some Lamiaceae taxa in Saudi Arabia and their taxonomic significance. Pak J Bot 47:1969–1977
El-Said H, Ashgar SS, Bader A et al (2021) Essential oil analysis and antimicrobial evaluation of three aromatic plant species growing in Saudi Arabia. Molecules 26:959
Soliman SS, Alsaadi AI, Youssef EG et al (2017) Calli essential oils synergize with lawsone against multidrug resistant pathogens. Molecules 22:2223
Oliva A, Costantini S, De Angelis M et al (2018) High potency of melaleuca alternifolia essential oil against multi-drug resistant gram-negative bacteria and methicillin-resistant Staphylococcus aureus. Molecules 23:2584
Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils–a review. Food Chem Toxicol 46:446–475
Tisserand R, Young R (2013). Essential oil safety: a guide for health care professionals. Elsevier Health Sciences, Amsterdam
Sirousmehr A, Arbabi J, Asgharipour MR (2014) Effect of drought stress levels and organic manures on yield, essential oil content and some morphological characteristics of sweet basil (Ocimum basilicum). Adv Environ Biol 8:880–885
Prakash B, Kedia A, Mishra PK, Dubey NK (2015) Plant essential oils as food preservatives to control moulds, mycotoxin contamination and oxidative deterioration of agri-food commodities–Potentials and challenges. Food Control 47:381–391
Sharifi-Rad J, Sureda A, Tenore GC et al (2017) Biological activities of essential oils: From plant chemoecology to traditional healing systems. Molecules 22:70
Swamy MK, Sinniah UR, Akhtar M (2015) In vitro pharmacological activities and GC-MS analysis of different solvent extracts of Lantana camara leaves collected from tropical region of Malaysia. Evid Based Comple Alternat Med. https://doi.org/10.1155/2015/506413
Arumugam G, Swamy MK, Sinniah UR (2016) Plectranthus amboinicus (Lour.) Spreng: botanical, phytochemical, pharmacological and nutritional significance. Molecules 21:369. https://doi.org/10.3390/molecules21040369
Baser KHC, Buchbauer G (2009) Handbook of essential oils: science, technology, and applications. CRC Press
Zuzarte M, Salgueiro L (2015) Essential oils chemistry. In Bioactive essential oils and cancer. Springer, Cham, p 19–61. https://doi.org/10.1007/978-3-319-19144-7_2
Tohidi B, Rahimmalek M, Trindade H (2019) Review on essential oil, extracts composition, molecular and phytochemical properties of Thymus species in Iran. Ind Crops Prod 134:89–99
Sharma SH, Thulasingam S, Nagarajan S (2017) Terpenoids as anti-colon cancer agents–A comprehensive review on its mechanistic perspectives. Eur J Pharmacol 795:169–178
Gyawali R, Ibrahim SA (2014) Natural products as antimicrobial agents. Food Control 46:412–429
Hyldgaard M, Mygind T, Meyer RL (2012) Essential oils in food preservation: mode of action, synergies, and interactions with food matrix components. Front Microbiol 3:12
Megawati FD, Sediawan WB, Hisyam A (2019) Kinetics of mace (Myristicae arillus) essential oil extraction using microwave assisted hydrodistillation: effect of microwave power. Ind Crops Prod 131:315–322
Chouhan S, Sharma K, Guleria S (2017) Antimicrobial activity of some essential oils—present status and future perspectives. Medicines 4:58
Farhat A, Fabiano-Tixier A-S, Visinoni F et al (2010) A surprising method for green extraction of essential oil from dry spices: microwave dry-diffusion and gravity. J Chromatogr A 1217:7345–7350
Sadeh D, Nitzan N, Chaimovitsh D et al (2019) Interactive effects of genotype, seasonality and extraction method on chemical compositions and yield of essential oil from rosemary (Rosmarinus officinalis L.). Ind Crops Prod 138:111419
Pavlić B, Vidović S, Vladić J et al (2015) Isolation of coriander (Coriandrum sativum L.) essential oil by green extractions versus traditional techniques. J Supercrit Fluids 99:23–28
Taban A, Saharkhiz MJ, Niakousari M (2018) Sweet bay (Laurus nobilis L.) essential oil and its chemical composition, antioxidant activity and leaf micromorphology under different extraction methods. Sustain Chem Pharm 9:12–18
Butnariu M, Sarac I (2018) Essential Oils from Plants. JBBS 1:35–43. https://doi.org/10.14302/issn.2576-6694.jbbs-18-2489
Raut JS, Karuppayil SM (2014) A status review on the medicinal properties of essential oils. Ind Crops Prod 62:250–264
Swamy MK, Sinniah UR (2015) A comprehensive review on the phytochemical constituents and pharmacological activities of Pogostemon cablin Benth.: an aromatic medicinal plant of industrial importance. Molecules 20:8521–8547
Swamy MK, Akhtar MS, Sinniah UR (2016) Antimicrobial properties of plant essential oils against human pathogens and their mode of action: an updated review. Evid Based Complement Alternat Med. https://doi.org/10.1155/2016/3012462
Lanigan RS, Yamarik TA (2002) Final report on the safety assessment of BHT (1). Int J Toxicol 21:19–94
Firuzi O, Miri R, Tavakkoli M, Saso L (2011) Antioxidant therapy: current status and future prospects. Curr Med Chem 18:3871–3888
Liang N, Kitts DD (2014) Antioxidant property of coffee components: assessment of methods that define mechanisms of action. Molecules 19:19180–19208
Edris AE (2007) Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: a review. Phytother Res 21:308–323
Miguel MG (2010) Antioxidant and anti-inflammatory activities of essential oils: a short review. Molecules 15:9252–9287
Tripathi R, Mohan H, Kamat JP (2007) Modulation of oxidative damage by natural products. Food Chem 100:81–90
Aebisher D, Cichonski J, Szpyrka E et al (2021) Essential oils of seven lamiaceae plants and their antioxidant capacity. Molecules 26:3793
Gulluce M, Sahin F, Sokmen M et al (2007) Antimicrobial and antioxidant properties of the essential oils and methanol extract from Mentha longifolia L. ssp. longifolia. Food Chem 103:1449–1456
Hussain AI, Anwar F, Sherazi STH, Przybylski R (2008) Chemical composition, antioxidant and antimicrobial activities of basil (Ocimum basilicum) essential oils depends on seasonal variations. Food Chem 108:986–995
Romeilah RM, Fayed SA, Mahmoud GI (2010) Chemical compositions, antiviral and antioxidant activities of seven essential oils. J Appl Sci Res 6:50–62
Tomaino A, Cimino F, Zimbalatti V et al (2005) Influence of heating on antioxidant activity and the chemical composition of some spice essential oils. Food Chem 89:549–554
Lang G, Buchbauer G (2012) A review on recent research results (2008–2010) on essential oils as antimicrobials and antifungals. A Rev Flavour Fragr J 27:13–39
Sánchez-González L, Vargas M, González-Martínez C et al (2011) Use of essential oils in bioactive edible coatings: a review. Food Eng Rev 3:1–16
Petretto GL, Chessa M, Piana A et al (2013) Chemical and biological study on the essential oil of Artemisia caerulescens L. ssp. densiflora (Viv.). Nat Prod Res 27:1709–1715
Bhavaniramya S, Vishnupriya S, Al-Aboody MS et al (2019) Role of essential oils in food safety: antimicrobial and antioxidant applications. Grain Oil Sci Technol 2:49–55
Hammer KA, Carson CF, Riley TV (1999) Antimicrobial activity of essential oils and other plant extracts. J Appl Microbiol 86:985–990
Hayes AJ, Markovic B (2003) Toxicity of Australian essential oil Backhousia citriodora (lemon myrtle). Part 2. absorption and histopathology following application to human skin. Food Chem Toxicol 41:1409–1416
Maurya S, Kushwaha AK, Singh G (2013) Biological significance of spicy essential oils. Adv Nat Sci 6:84–95
Mota AS, Martins MR, Arantes S et al (2015) Antimicrobial activity and chemical composition of the essential oils of Portuguese Foeniculum vulgare fruits. Nat Prod Commun. https://doi.org/10.1177/1934578X1501000437
Carson CF, Hammer KA, Riley TV (2006) Melaleuca alternifolia (tea tree) oil: a review of antimicrobial and other medicinal properties. Clin Microbiol Rev 19:50–62
Behbahani BA, Noshad M, Falah F (2019) Cumin essential oil: Phytochemical analysis, antimicrobial activity and investigation of its mechanism of action through scanning electron microscopy. Microb Pathog 136:103716
Huang Z, Jia S, Zhang L et al (2019) Inhibitory effects and membrane damage caused to fish spoilage bacteria by cinnamon bark (Cinnamomum tamala) oil. LWT-Food Sci Technol 112:108195
Xiang F, Bai J, Tan X et al (2018) Antimicrobial activities and mechanism of the essential oil from Artemisia argyi Levl. et Van. var. argyi cv. Qiai Ind Crops Prod 125:582–587
Guo J, Gao Z, Li G et al (2019) Antimicrobial and antibiofilm efficacy and mechanism of essential oil from Citrus Changshan-huyou YB chang against Listeria monocytogenes. Food Control 105:256–264
Churklam W, Chaturongakul S, Ngamwongsatit B, Aunpad R (2020) The mechanisms of action of carvacrol and its synergism with nisin against Listeria monocytogenes on sliced bologna sausage. Food Control 108:106864
Hammer KA, Carson CF (2011) Antibacterial and antifungal activities of essential oils. Lipids Essent Oils Antimicrob Agents 77:256–293
Irkin R, Korukluoglu M (2009) Effectiveness of Cymbopogon citratus L. essential oil to inhibit the growth of some filamentous fungi and yeasts. J Med Food 12:193–197
Pragadheesh VS, Saroj A, Yadav A et al (2013) Chemical characterization and antifungal activity of Cinnamomum camphora essential oil. Ind Crops Prod 49:628–633
Viuda-Martos M, Ruiz-Navajas Y, Fernández-López J, Pérez-Álvarez J (2008) Antifungal activity of lemon (Citrus lemon L.), mandarin (Citrus reticulata L.), grapefruit (Citrus paradisi L.) and orange (Citrus sinensis L.) essential oils. Food Control 19:1130–1138
Raut JS, Shinde RB, Chauhan NM, Mohan Karuppayil S (2013) Terpenoids of plant origin inhibit morphogenesis, adhesion, and biofilm formation by Candida albicans. Biofouling 29:87–96
Zore GB, Thakre AD, Jadhav S, Karuppayil SM (2011) Terpenoids inhibit Candida albicans growth by affecting membrane integrity and arrest of cell cycle. Phytomedicine 18:1181–1190
Rao A, Zhang Y, Muend S, Rao R (2010) Mechanism of antifungal activity of terpenoid phenols resembles calcium stress and inhibition of the TOR pathway. Antimicrob Agents Chemother 54:5062–5069
World Health Organization. (2019). WHO global report on traditional and complementary medicine 2019. World Health Organization. https://apps.who.int/iris/handle/10665/312342
Swamy MK, Sinniah UR (2016) Patchouli (Pogostemon cablin Benth.): botany, agrotechnology and biotechnological aspects. Ind Crops Prod 87:161–176
Jugreet BS, Suroowan S, Rengasamy RK, Mahomoodally MF (2020) Chemistry, bioactivities, mode of action and industrial applications of essential oils. Trends Food Sci Technol 101:89–105
Bedi S, Vyas SP (2008) A handbook of aromatic and essential oil plants: cultivation, chemistry, processing and uses. In: A Handbook of aromatic and essential oil plants: cultivation, chemistry, processing and uses. pp 598–598
Rather MA, Dar BA, Sofi SN et al (2016) Foeniculum vulgare: A comprehensive review of its traditional use, phytochemistry, pharmacology, and safety. Arab J Chem 9:S1574–S1583
Khaleil MM, Alnoman MM, Elrazik ESA et al (2021) Essential Oil of Foeniculum vulgare Mill. As a green fungicide and defense-inducing agent against fusarium root rot disease in Vicia faba L. Biology 10:696
Rahimi R, Ardekani MRS (2013) Medicinal properties of Foeniculum vulgare Mill. in traditional Iranian medicine and modern phytotherapy. Chin J Integr Med 19:73–79
Said-Al Ahl HA, Sarhan AM, Dahab A et al (2015) Essential oils of Anethum graveolens L.: Chemical composition and their antimicrobial activities at vegetative, flowering and fruiting stages of development. Int J Plant Sci Ecol 1:98–102
Singh G, Maurya S, De Lampasona MP, Catalan C (2005) Chemical constituents, antimicrobial investigations, and antioxidative potentials of Anethum graveolens L. essential oil and acetone extract: Part 52. J Food Sci 70:M208–M215
Jirovetz L, Buchbauer G, Stoyanova AS et al (2003) Composition, quality control, and antimicrobial activity of the essential oil of long-time stored dill (Anethum graveolens L.) seeds from Bulgaria. J Agric Food Chem 51:3854–3857
Chahal KK, Kumar A, Bhardwaj U, Kaur R (2017) Chemistry and biological activities of Anethum graveolens L. (dill) essential oil: A review. J Pharmacogn Phytochem 6:295–306
Pejčić M, Stojanović-Radić Z, Genčić M et al (2020) Anti-virulence potential of basil and sage essential oils: Inhibition of biofilm formation, motility and pyocyanin production of Pseudomonas aeruginosa isolates. Food Chem Toxicol 141:111431
Hadidi M, Pouramin S, Adinepour F et al (2020) Chitosan nanoparticles loaded with clove essential oil: characterization, antioxidant and antibacterial activities. Carbohydr Polym 236:116075
Micić D, Ostojić S, Pezo L et al (2019) Essential oils of coriander and sage: Investigation of chemical profile, thermal properties and QSRR analysis. Ind Crops Prod 138:111438
Esmaeili H, Cheraghi N, Khanjari A et al (2020) Incorporation of nanoencapsulated garlic essential oil into edible films: A novel approach for extending shelf life of vacuum-packed sausages. Meat Sci 166:108135
Cui H, Zhang C, Li C, Lin L (2018) Antimicrobial mechanism of clove oil on Listeria monocytogenes. Food Control 94:140–146
Liu T, Kang J, Liu L (2021) Thymol as a critical component of Thymus vulgaris L. essential oil combats Pseudomonas aeruginosa by intercalating DNA and inactivating biofilm. LWT 136:110354
Politeo O, Jukić M, Miloš M (2006) Chemical composition and antioxidant activity of essential oils of twelve spice plants. Croat Chem Acta 79:545–552
Burits M, Bucar F (2000) Antioxidant activity of Nigella sativa essential oil. Phytother Res 14:323–328
Farag RS, Badei A, Hewedi FM, El-Baroty GSA (1989) Antioxidant activity of some spice essential oils on linoleic acid oxidation in aqueous media. JAOCS 66:792–799
Teissedre PL, Waterhouse AL (2000) Inhibition of oxidation of human low-density lipoproteins by phenolic substances in different essential oils varieties. J Agric Food Chem 48:3801–3805
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
AAM collecting previous literatures—reviewing and continuous follow-up of all parts of the research. BMA participation in determining the title of research—collecting previous studies—writing research content—identifying and organizing references—adjusting research formats in line with the journal.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohamed, A.A., Alotaibi, B.M. Essential oils of some medicinal plants and their biological activities: a mini review. J.Umm Al-Qura Univ. Appll. Sci. 9, 40–49 (2023). https://doi.org/10.1007/s43994-022-00018-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43994-022-00018-1