Abstract
Aims
The mechanism behind clock coordination in female reproductive disorders is poorly understood despite the known importance of coordinated and synchronized timing of central and clocks in reproductive organs. We investigated the effect of continuous artificial light (LL) on the central and peripheral reproductive clock gene (Bmal1, Clock, Per1, Per2 and Cry1) and its downstream regulators (Hgf, PR-A and HOXA10) during non-pregnancy and pregnancy phases of female mice.
Main methods
Mice (n = 60) in two sets, were maintained under continuous light (LL) and natural day cycle (LD;12L: 12D) for both non-pregnant and pregnant study. Tissues from hypothalamus-containing SCN, ovary, uterus and serum were collected at different zeitgeber time points (ZT; at 4-h intervals across 24-h periods).
Key findings
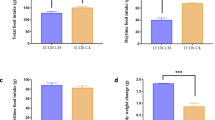
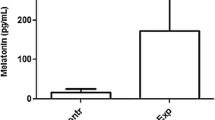
LL exposure desynchronized the expressions of the clock mRNAs (Bmal1, Clock, Per1, Per2 and Cry1) in SCN, ovary, and uterus along with Hgf mRNA rhythm. LL significantly increased the thickness of endometrial tissues. Furthermore, the pregnant study revealed lower serum progesterone level during peri- and post-implantation under LL along with downregulated expression of progesterone receptor (PR) as well as progesterone dependent uterine Homeobox A-10 (Hoxa10) proteins with lowered pregnancy outcomes.
Significance
Our result suggests that LL disrupted the circadian coordination between central and clock genes in reproductive tissue leading to interrupted uterine physiology and altered pregnancy in mice. This led us to propose that duration of light exposure at work-places or home for females is very important in prevention of pregnancy anomalies.
Graphical Abstract







Similar content being viewed by others
References
Plano, S. A., Casiraghi, L. P., Garcia, M. P., Paladino, N., Golombek, D. A., & Chiesa, J. J. (2017). Circadian and metabolic effects of light: implications in weight homeostasis and health. Frontiers in Neurology, 8, 558. https://doi.org/10.3389/fneur.2017.00558
Reppert, S. M., & Weaver, D. R. (2002). Coordination of circadian timing in mammals. Nature, 418(6901), 935–941. https://doi.org/10.1038/nature00965
Tasaki, H., Zhao, L., Isayama, K., Chen, H., Yamauchi, N., Shigeyoshi, Y., Hashimoto, S., & Hattori, M. (2013). Profiling of circadian genes expressed in the uterus endometrial stromal cells of pregnant rats as revealed by DNA microarray coupled with RNA interference. Frontiers in Endocrinology, 4, 82. https://doi.org/10.3389/fendo.2013.00082
Hsu, C. N., & Tain, Y. L. (2020). Light and circadian signaling pathway in pregnancy: programming of adult health and disease. International Journal of Molecular Sciences, 21(6), 2232. https://doi.org/10.3390/ijms21062232
Touitou, Y., Reinberg, A., & Touitou, D. (2017). Association between light at night, melatonin secretion, sleep deprivation, and the internal clock: health impacts and mechanisms of circadian disruption. Life Science, 173, 94–106. https://doi.org/10.1016/j.lfs.2017.02.008
Dolatshad, H., Campbell, E. A., O’Hara, L., Maywood, E. S., Hastings, M. H., & Johnson, M. H. (2006). Developmental and reproductive performance in circadian mutant mice. Human Reproduction, 21(1), 68–79. https://doi.org/10.1093/humrep/dei313
Miller, B. H., Olson, S. L., Turek, F. W., Levine, J. E., Horton, T. H., & Takahashi, J. S. (2004). Circadian clock mutation disrupts estrous cyclicity and maintenance of pregnancy. Current Biology, 14(15), 1367–1373. https://doi.org/10.1016/j.cub.2004.07.055
Kaneshi, Y., Ohta, H., Morioka, K., Hayasaka, I., Uzuki, Y., Akimoto, T., Moriichi, A., Nakagawa, M., Oishi, Y., Wakamatsu, H., Honma, N., Suma, H., Sakashita, R., Tsujimura, S., Higuchi, S., Shimokawara, M., Cho, K., & Minakami, H. (2016). Influence of light exposure at nighttime on sleep development and body growth of preterm infants. Scientific Reports, 6, 21680. https://doi.org/10.1038/srep21680
Pietroiusti, A., Neri, A., Somma, G., Coppeta, L., Iavicoli, I., Bergamaschi, A., & Magrini, A. (2010). Incidence of metabolic syndrome among night-shift healthcare workers. Journal of Occupational and Environmental Medicine, 67, 54–57. https://doi.org/10.1136/oem.2009.046797
Zhu, J. L., Hjollund, N. H., Andersen, A. M., & Olsen, J. (2004). Shift work job stress and late fetal loss: The National Birth Cohort in Denmark. Journal of Occupational and Environmental Medicine, 46(11), 1144–1149. https://doi.org/10.1097/01.jom.0000145168.21614.21
Fielder, A. R., & Moseley, M. J. (2000). Environmental light and the preterm infant. Seminars in Perinatology, 24(4), 291–298. https://doi.org/10.1053/sper.2000.8597
Mirmiran, M., & Ariagno, R. L. (2000). Influence of light in the NICU on the development of circadian rhythms in preterm infants. Seminars in Perinatology, 24(4), 247–257. https://doi.org/10.1053/sper.2000.8593
Goldstein, C. A., O’Brien, L. M., Bergin, I. L., & Saunders, T. L. (2018). The effect of repeated light-dark shifts on uterine receptivity and early gestation in mice undergoing embryo transfer. Systems Biology in Reproductive Medicine, 64(2), 103–111. https://doi.org/10.1080/19396368.2017.1408715
Fernandez, R. C., Marino, J. L., Varcoe, T. J., Davis, S., Moran, L. J., Rumbold, A. R., Brown, H. M., Whitrow, M. J., Davies, M. J., & Moore, V. M. (2016). Fixed or rotating night shift work undertaken by women: implications for fertility and miscarriage. Seminars in Reproductive Medicine, 34(2), 74–82. https://doi.org/10.1055/s-0036-1571354
Stocker, L. J., Macklon, N. S., Cheong, Y. C., & Bewley, S. J. (2014). Influence of shift work on early reproductive outcomes: a systematic review and meta-analysis. Obstetrics & Gynecology, 124(1), 99–110. https://doi.org/10.1097/AOG.0000000000000321
Summa, K. C., Vitaterna, M. H., & Turek, F. W. (2012). Environmental perturbation of the circadian clock disrupts pregnancy in the mouse. PLoS ONE, 7(5), e37668. https://doi.org/10.1371/journal.pone.0037668
Valenzuela, F. J., Vera, J., Venegas, C., Pino, F., & Lagunas, C. (2015). Circadian system and melatonin hormone: risk factors for complications during pregnancy. Obstetrics and Gynecology International, 2015, 825802. https://doi.org/10.1155/2015/825802
King, D. P., & Takahashi, J. S. (2000). Molecular genetics of circadian rhythms in mammals. Annual Review of Neuroscience, 23, 713–742. https://doi.org/10.1146/annurev.neuro.23.1.713
Blume, C., Garbazza, C., & Spitschan, M. (2019). Effects of light on human circadian rhythms, sleep and mood. Somnologie, 23(3), 147–156. https://doi.org/10.1007/s11818-019-00215-x
Portaluppi, F., Smolensky, M. H., & Touitou, Y. (2010). Ethics and methods for biological rhythm research on animals and human beings. Chronobiology International, 27(9–10), 1911–1929. https://doi.org/10.3109/07420528.2010.516381
Sinhasane, S. V., & Joshi, B. N. (1998). Impact of aggressive encounters on reproductive behaviour in the Indian desert gerbil, Meriones hurrianae (Jerdon). Journal of Biosciences, 23, 633–639. https://doi.org/10.1007/BF02709176
Quennell, J. H., Howell, C. S., Roa, J., Augustine, R. A., Grattan, D. R., & Anderson, G. M. (2011). Leptin deficiency and diet-induced obesity reduce hypothalamic kisspeptin expression in mice. Endocrinology, 152(4), 1541–1550. https://doi.org/10.1210/en.2010-1100
Feng, T. R., Li, Z., & Li, S. X. (2020). Effects of constant light on the circadian system in rats. Austin Journal of Pharmacology and Therapeutics, 8(2), 1121.
Hardy, D. F. (1970). The effect of constant light on the estrous cycle and behavior of the female rat. Physiology & Behavior, 5(4), 421–425. https://doi.org/10.1016/0031-9384(70)90246-5
Paccola, C. C., Resende, C. G., Stumpp, T., Miraglia, S. M., & Cipriano, I. (2013). The rat estrous cycle revisited: a quantitative and qualitative analysis. Animal Reproduction, 10, 677–683.
Shukla, D., Das, M., Kasade, D., Pandey, M., Dubey, A. K., Yadav, S. K., & Parmar, A. S. (2020). Sandalwood-derived carbon quantum dots as bioimaging tools to investigate the toxicological effects of malachite green in model organisms. Chemosphere, 248, 125998. https://doi.org/10.1016/j.chemosphere.2020.125998
Pakrasi, P. L., & Jain, A. K. (2008). Cyclooxygenase-2 derived PGE2 and PGI2 play an important role via EP2 and PPAR delta receptors in early steps of oil induced decidualization in mice. Placenta, 29(6), 523–530. https://doi.org/10.1016/j.placenta.2008.03.001
Wharfe, M. D., Mark, P. J., Wyrwoll, C. S., Smith, J. T., Yap, C., Clarke, M. W., & Waddell, B. J. (2016). Pregnancy-induced adaptations of the central circadian clock and maternal glucocorticoids. Journal of Endocrinology, 228(3), 135–47. https://doi.org/10.1530/JOE-15-0405
Mishra, I., Knerr, R. M., Stewart, A. A., Payette, W. I., Richter, M. M., & Ashley, N. T. (2019). Light at night disrupts diel patterns of cytokine gene expression and endocrine profiles in zebra finch (Taeniopygia guttata). Scientific Reports, 9(1), 15833. https://doi.org/10.1038/s41598-019-51791-9
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods, 25(4), 402–408. https://doi.org/10.1006/meth.2001.1262
Chowdhury, J. P., & Haldar, C. (2018). Photoperiodic regulation of ovarian steroidogenesis in a tropical rodent, Funambulus pennanti: role of melatonin and MT1. Biological Rhythm Research, 51, 102–119. https://doi.org/10.1080/09291016.2018.1525134
Yadav, S. K., Haldar, C., & Singh, S. S. (2011). Variation in melatonin receptors (Mel1a and Mel1b) and androgen receptor (AR) expression in the spleen of a seasonally breeding bird, Perdicula asiatica. Journal of Reproductive Immunology, 92(1–2), 54–61. https://doi.org/10.1016/j.jri.2011.08.003
Amano, T., Ripperger, J. A., & Albrecht, U. (2020). Changing the light schedule in late pregnancy alters birth timing in mice. Theriogenology, 154, 212–222. https://doi.org/10.1016/j.theriogenology.2020.05.032
Pawlik, T. M., Hawke, D. H., Liu, Y., Krishnamurthy, S., Fritsche, H., Hunt, K. K., & Kuerer, H. M. (2006). Proteomic analysis of nipple aspirate fluid from women with early-stage breast cancer using isotope-coded affinity tags and tandem mass spectrometry reveals differential expression of vitamin D binding protein. BMC Cancer, 6, 68. https://doi.org/10.1186/1471-2407-6-68
Nakamura, T. J., Moriya, T., Inoue, S., Shimazoe, T., Watanabe, S., Ebihara, S., & Shinohara, K. (2005). Estrogen differentially regulates expression of Per1 and Per2 genes between central and peripheral clocks and between reproductive and nonreproductive tissues in female rats. Journal of Neuroscience Research, 82(5), 622–30. https://doi.org/10.1002/jnr.20677
Sudo, M., Sasahara, K., Moriya, T., Akiyama, M., Hamada, T., & Shibata, S. (2003). Constant light housing attenuates circadian rhythms of mPer2 mRNA and mPER2 protein expression in the suprachiasmatic nucleus of mice. Neuroscience, 121(2), 493–499. https://doi.org/10.1016/s0306-4522(03)00457-3
Wang, Y., Chen, M., Xu, J., Liu, X., Duan, Y., Zhou, C., & Xu, Y. (2020). Core clock gene Bmal1 deprivation impairs steroidogenesis in mice luteinized follicle cells. Reproduction, 160(6), 955–967. https://doi.org/10.1530/REP-20-0340
Nakamura, T. J., Sellix, M. T., Kudo, T., Nakao, N., Yoshimura, T., Ebihara, S., Colwell, C. S., & Block, G. D. (2010). Influence of the estrous cycle on clock gene expression in reproductive tissues: effects of fluctuating ovarian steroid hormone levels. Steroids, 75(3), 203–12. https://doi.org/10.1016/j.steroids.2010.01.007
He, P. J., Hirata, M., Yamauchi, N., & Hattori, M. A. (2007). Up-regulation of Per1 expression by estradiol and progesterone in the rat uterus. Journal of Endocrinology, 194(3), 511–519. https://doi.org/10.1677/JOE-07-0172
Karman, B. N., & Tischkau, S. A. (2006). Circadian clock gene expression in the ovary: effects of luteinizing hormone. Biology of reproduction, 75(4), 624–32. https://doi.org/10.1095/biolreprod.106.050732
Nakamura, T. (1991). Structure and function of hepatocyte growth factor. Progress in Growth Factor Research, 3(1), 67–85. https://doi.org/10.1016/0955-2235(91)90014-u
Sen, A., & Hoffmann, H. M. (2020). Role of core circadian clock genes in hormone release and target tissue sensitivity in the reproductive axis. Molecular and Cellular Endocrinology, 501, 110655. https://doi.org/10.1016/j.mce.2019.110655
Shiraga, M., Komatsu, N., Teshigawara, K., Okada, A., Takeuchi, S., Fukamachi, H., & Takahashi, S. (2000). Epidermal growth factor stimulates proliferation of mouse uterine epithelial cells in primary culture. Zoological Science, 17(5), 661–666. https://doi.org/10.2108/zsj.17.661
Nicolaides, N. C., Charmandari, E., Chourousos, G. P., & Kino, T. (2014). Circadian endocrine rhythms: the hypothalamic–pituitary–adrenal axis and its actions. Annals of the New York Academy, 1318, 71–80. https://doi.org/10.1111/nyas.12464
Islam, M. R., Yamagami, K., Yoshii, Y., & Yamauchi, N. (2016). Growth factor induced proliferation, migration, and lumen formation of rat endometrial epithelial cells in vitro. Journal of Reproduction and Development, 62(3), 271–278. https://doi.org/10.1262/jrd.2015-158
Brandon, D. H., Holditch, D. D., & Belyea, M. (2002). Preterm infants born at less than 31 weeks’ gestation have improved growth in cycled light compared with continuous near darkness. The Journal of Pediatrics, 140(2), 192–199. https://doi.org/10.1067/mpd.2002.121932
Matsumoto, K., & Nakamura, T. (1993). Roles of HGF as a pleiotropic factor in organ regeneration. EXS, 65, 225–249.
Serber, D. W., Rogala, A., Makarem, M., Rosson, G. B., Simin, K., Godfrey, V., Van Dyke, T., Eaves, C. J., & Bultman, S. J. (2012). The BRG1 chromatin remodeler protects against ovarian cysts, uterine tumors, and mammary tumors in a lineage-specific manner. PLoS ONE, 7(2), e31346. https://doi.org/10.1371/journal.pone.0031346
Folkins, A. K., Nevadunsky, N. S., Saleemuddin, A., Jarboe, E. A., Muto, M. G., Feltmate, C. M., Crum, C. P., & Hirsch, M. S. (2010). Evaluation of vascular space involvement in endometrial adenocarcinomas: laparoscopic vs abdominal hysterectomies. Modern Pathology, 23(8), 1073–1079. https://doi.org/10.1038/modpathol.2010.91
Evans, G. S., Gibson, D. F., Roberts, S. A., Hind, T. M., & Potten, C. S. (1990). Proliferative changes in the genital tissue of female mice during the oestrous cycle. Cell and tissue kinetics, 23(6), 619–635. https://doi.org/10.1111/j.1365-2184.1990.tb01350.x
Godbole, G., Suman, P., Malik, A., Galvankar, M., Joshi, N., Fazleabas, A., Gupta, S. K., & Modi, D. (2017). Decrease in expression of HOXA10 in the decidua after embryo implantation promotes trophoblast invasion. Endocrinology, 158(8), 2618–2633. https://doi.org/10.1210/en.2017-00032
Yin, Y., Lin, V., Sawalha, D., Bany, B. M., & Ma, L. (2011). Molecular analysis of implantation defects in homeobox gene HOXA10-deficient mice. Reproductive System and Sexual Disorders, S1, 001. https://doi.org/10.4172/2161-038X.S1-001
Lee, K. Y., Jeong, J. W., Tsai, S. Y., Lydon, J. P., & DeMayo, F. J. (2007). Mouse models of implantation. TEM, 18(6), 1043–2760. https://doi.org/10.1016/j.tem.2007.06.002
Mulac-Jericevic, B., Mullinax, R. A., DeMayo, F. J., Lydon, J. P., & Conneely, O. M. (2000). Subgroup of reproductive functions of progesterone mediated by progesterone receptor-B isoform. Science, 289(5485), 1751–1754. https://doi.org/10.1126/science.289.5485.1751
Lim, H., Ma, L., Ma, W. G., Maas, R. L., & Dey, S. K. (1999). HOXA-10 regulates uterine stromal cell responsiveness to progesterone during implantation and decidualization in the mouse. Molecular Endocrinology, 13(6), 1005–1017. https://doi.org/10.1210/mend.13.6.0284
Taylor, H., Arici, A., Olive, D., & Igarashi, P. (1998). HOXA10 is expressed in response to sex steroids at the time of implantation in the human endometrium. Journal of Clinical Investigation, 101(7), 1379–1384. https://doi.org/10.1172/JCI1057
Lydon, J. P., DeMayo, F. J., Funk, C. R., Mani, S. K., Hughes, A. R., Montgomery, C. A., Shyamala, G., Conneely, O. M., & O’Malley, B. W. (1995). Mice lacking progesterone receptor exhibit pleiotropic reproductive abnormalities. Genes & Development, 9(18), 2266–2278. https://doi.org/10.1101/gad.9.18.2266
Ohara, T., Nakamura, T. J., Nakamura, W., & Tokuda, I. T. (2020). Modeling circadian regulation of ovulation timing: age-related disruption of estrous cyclicity. Scientific Reports, 10, 16767. https://doi.org/10.1038/s41598-020-73669-x
Miller, B. H., & Takahashi, J. S. (2014). Central circadian control of female reproductive function. Frontiers in endocrinology, 4, 195. https://doi.org/10.3389/fendo.2013.00195
Acknowledgements
The authors would like to thank UGC-Non-NET fellowship (UGC-RESEARCH-FELLOW2017-18/41147) and ICMR-SRF fellowship (Award No RBMH/FW/2019/6) to Ms. Megha Das, ICMR adhoc project (P-14/267) research grant to Dr. S. K Yadav, Centre for Advance Studies (CAS) facilities to Department of Zoology and Institute of Life Science (ISLS) for permission to use Real-Time PCR facility and Nanodrop Facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest that could be distinguished as discriminating the impartiality of the review reported.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Das, M., Minocha, T., Kumar, D. et al. Continuous artificial light potentially disrupts central and peripheral reproductive clocks leading to altered uterine physiology and reduced pregnancy success in albino mice. Photochem Photobiol Sci 21, 1217–1232 (2022). https://doi.org/10.1007/s43630-022-00210-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43630-022-00210-6