Abstract
Lupus nephritis is a serious condition, for which treatments are limited; hence, there is a need for new cure approaches. The aim of this study was to evaluate the therapeutic effects of calycosin against lupus nephritis induced by lipopolysaccharide (LPS) in human renal cortex proximal convoluted tubule epithelial cells (HK-2). HK-2 cells were stimulated with 1 μg/ml LPS to create a lupus nephritis cell model; the cells were pretreated with calycosin. Cell viability and apoptosis rate were determined using the cell counting kit-8 assay and flow cytometry, respectively. A caspase-3 activity detection kit was used to determine caspase-3 activity. Interleukin (IL)-6, IL-1β, and tumor necrosis factor alpha (TNF-α) levels were determined using enzyme-linked immunosorbent assay kits. Lactate dehydrogenase (LDH) level was determined using an LDH assay kit. Finally, western blotting and reverse transcription-quantitative polymerase chain reaction were performed to determine apoptosis-related protein levels and nuclear factor erythroid 2–related factor 2 (Nrf2)/heme oxygenase-1 (HO-1) signaling. Calycosin had no cytotoxic effects on HK-2 cells. Lipopolysaccharide stimulation significantly inhibited cell viability; increased the IL-6, IL-1β, and TNF-α levels; and elevated apoptosis rate, caspase3 activity, and LDH level in HK-2 cells. The protein level of cleaved caspase3 was also increased in LPS-treated HK-2 cells. In addition, the pattern of Nrf2/HO-1 signaling was disturbed by LPS. These effects were reversed by calycosin treatment. Calycosin could alleviate LPS-induced lupus nephritis and may thus be a novel agent for its treatment.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic lupus erythematosus (SLE) is the most common autoimmune disease in China, with an incidence rate of 30.13–70.41% per 100,000 people. Approximately 40–60% of patients with SLE have lupus nephritis in the initial stage of the disease (Yu et al. 2022). Currently, there is no unified treatment strategy for lupus nephritis; available treatments are mainly aimed at controlling lupus activity, preventing the progression of renal lesions, and minimizing the adverse effects of drug therapy. Individualized treatments should be developed according to clinical manifestations, pathological features, and disease activity (Anders et al. 2020). In 1986, the National Institute of Health of the USA reported that the efficacy of combined immunosuppressant therapy for lupus nephritis was considerably superior to that of conventional hormone therapy, but long-term use of glucocorticoids and/or immunosuppressants such as cyclophosphamide, azathioprine, and cyclosporine A had several adverse effects including severe infection, myelosuppression, gonadal suppression, neuroendocrine disorders, osteoporosis, carcinogenesis, and teratogenesis (Rovin et al. 2022). Consequently, some patients cannot adhere to long-term treatment and are forced to reduce the drug dose or stop the drug. Other approaches such as plasmapheresis, hematopoietic stem cell transplantation, and gene therapy are being investigated (Yu et al. 2017; Zhang et al. 2017). Therefore, the exploration of drug therapies in line with health economics and with less adverse effects has become the focus of current clinical research.
Calycosin (1), a natural isoflavone isolated from members of the genus Astragalus, Fabaceae, has antioxidant, antiradiation, anticancer, antiviral, and lipid-lowering effects. It has been used to treat myocardial fibrosis via the TGFBR1 signaling pathway (Chen et al. 2022). Calycosin also represses the invasion and migration of breast cancer cells (Zhang et al. 2021). It protects against cerebral ischemia–reperfusion via SIRT1 signaling (Yan et al. 2019). However, it is unclear whether calycosin has a preventive effect against lupus nephritis.

Nuclear factor erythroid-2–related factor 2 (Nrf2), which belongs to the Carney complex (CNC) regulatory protein family, is a transcription factor that regulates cellular REDOX function (Tonelli et al. 2018). Heme oxygenase-1 (HO-1) is a rate-limiting enzyme that catalyzes the degradation of heme to carbonic oxide, biliverdin, and free iron (Wang et al. 2019). Nrf2/HO-1 signaling is involved in various human diseases. Recently, the inhibition of Nrf2/HO-1 has been shown to mediate osteoarthritis (Chen et al. 2019). Nrf2/HO-1 is also involved in hepatic ischemia–reperfusion (Ge et al. 2017). Moreover, Nrf2/HO-1 is a classical therapeutic biomarker for Parkinson (Wang et al. 2021). However, it remains unknown whether calycosin prevents lupus nephritis via the Nrf2/HO-1 pathway. In this study, we aimed to determine the therapeutic effect of calycosin against lupus nephritis induced with lipopolysaccharide (LPS) and illustrate the underlying mechanisms.
Materials and Methods
Model Establishment
Human-derived proximal tubule epithelial cell line HK-2 was obtained from Meisen Bio (Zhejiang, China). The cells were cultured in Ham’s F-12 medium (BI, Israel) supplemented with 1% penicillin and streptomycin (P/I) (BI) and 12% fetal bovine serum (Gibco, NY) in an atmosphere of 5% CO2, at 37 °C. A lupus nephritis model was established using HK-2 cells stimulated with 1 μg/ml LPS (L5293; Merck) for 24 h; the cells were pretreated with calycosin (HY-N0519; MCE). ML385 (1 µM, HY-100523), a Nrf2/HO-1 signaling inhibitor, were procured from MCE. To determine whether calycosin (99.93% purity, Lote S903801, Selleck, China) protects against lupus nephritis via Nrf2/HO-1 signaling, HK-2 cells were treated with calycosin (80 μM) alone or in combination with 1 µM ML385 for 2 h, and then stimulated for 24 h with 1 μg/ml LPS.
Cell-Counting Kit-8 Assay
Cell viability was evaluated using the cell-counting kit-8 (CCK-8) (Fcmacs, Nanjing, China). After LPS stimulation, HK-2 cells were resuspended and seeded in 96-well plates at 2.5 × 103 cells/well and incubated with 10 μl of detection solution for 1.5 h at 37 °C under 5% CO2 in the dark. The optical density (OD) of the samples was measured at 450 nm using an ultraviolet spectrophotometer (Infinite Pro; Tecan).
Lactate Dehydrogenase Assay
Lactate dehydrogenase (LDH) level in the cells was measured following stimulation with LPS using an LDH activity assay kit (ARG81306; Arigo, Taiwan, China). Following the manufacturer’s instructions, the OD of the sample in each well was determined at 490 nm to analyze LDH activity.
Cell Apoptosis
For this assay, 2 × 105 LPS-induced cells were harvested. The cells were incubated with 5 μl of Annexin V-FITC and 5 μl of propidium iodide (Fcmacs, China) at room temperature in the dark for 35 min. Apoptosis rate was analyzed using flow cytometry (C6; Thermo Fisher Scientific, USA).
Caspase-3 Activity Assay
The activity of caspase-3 in cells was determined using a colorimetric kit (ImmunoWay, China). The culture medium and cells were covered with trypsin (BI). The cell samples were then incubated with lysis buffer (Proteintech, China) for 20 min and then centrifuged at 10,000 × g for 1 min. The samples were analyzed using a microplate reader (Tecan, Switzerland).
Enzyme-Linked Immunosorbent Assay
The cell culture supernatant was harvested and used for determining the level of inflammatory cytokines (interleukin (IL)-6, IL-1β, and tumor necrosis factor (TNF)-α). The ELISA kits were obtained from Beyotime (Shanghai, China). All operations were performed according to the manufacturer’s protocol.
Reverse Transcription-Quantitative PCR
Following the supplier’s protocol, the total RNA was isolated from the cells using Isolation TRIzol buffer® (Multi Sciences, Hangzhou, China), and cDNA was obtained by reverse transcribing the RNA using a reverse transcription-quantitative polymerase chain reaction (RT-qPCR) kit (Yeasen, China). Furthermore, qRT-PCR was performed using the PerfectStart® SYBR qPCR Mix (Vazyme, Nanjing). The gene expression levels were calculated using the 2−ΔΔCt method. The sequences of primers of Nrf2, HO-1, and β-actin were as follows: β-actin: 5′-CCATCGCCAGTTGCCGATCC-3′ (F) and 5′-GCGAGAGGAGCACAGATACCACCAA-3′ (R); HO-1: 5′-AAGACTGCGTTCCTGCTCAAC-3′ (F) and 5′-AAAGCCCTACAGCAACTGTCG-3′ (R); Nrf2: 5′-TCAGCGACGGAAAGAGTATGA-3′ (F) and 5′-CCACTGGTTTCTGACTGGATGT-3′ (R).
Western Blotting
HK-2 cells were lysed using RIPA buffer (Univ, Shanghai, China). Proteins were resolved using sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred on to PVDF membranes (Whatman, USA). PBST (Univ) and 5% nonfat milk powder (CST, USA) were used to block the PVDF membranes. The membranes were then incubated with primary antibodies against Nrf2, HO-1, cleaved caspase-3, and GAPDH for 12 h. The next day, after incubating with the secondary antibody, the blots were assessed using an image capture system (Wix, USA), and the grayscale value of the target protein was determined using ImageJ.
Statistical Analysis
Data are presented as mean ± standard deviation (SD). Differences among groups were evaluated using a one-way analysis of variance (ANOVA). Results with p < 0.05 were considered statistically significant.
Results and Discussion
HK-2 Cell Growth
The CCK-8 and LDH analyses were performed to explore the treatment effects of 0, 10, 20, 40, 60, and 80 μM calycosin (1) for 24 h in HK-2 cells. In the CCK-8 assay, the OD values indicated that calycosin was not toxic to HK-2 cells (Fig. 1A). Similarly, the LDH activity indicated that calycosin had no negative effects on HK-2 cells (Fig. 1B). These findings indicated that calycosin had no toxic effects on HK-2 cells.
Inhibition of Apoptosis and Inflammation
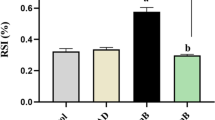
To investigate the effects of calycosin (1) on LPS-induced apoptosis and inflammation, HK-2 cells were treated with calycosin (20, 40, and 80 μM) for 2 h before LPS stimulation. The CCK-8 assay results showed that LPS notably decreased cell viability, whereas calycosin significantly increased the viability of HK-2 cells in a dose-dependent manner (Fig. 2A). Furthermore, the LDH concentration increased in LPS-stimulated HK-2 cells, whereas calycosin reversed these effects (Fig. 2B). The cell apoptotic assay revealed that the apoptosis rate of LPS-stimulated HK-2 cells considerably increased, but it was reduced by calycosin (20, 40, and 80 μM) treatment (Fig. 2C and D). The western blotting results indicated that the cleaved caspase-3 level and ratio of cleaved caspase-3/GAPDH were elevated in LPS-stimulated HK-2 cells, and these changes were reversed by calycosin in a dose-dependent manner (Fig. 2E and F). Consistent with these findings, the caspase-3 activity was increased in the LPS-stimulated group; however, calycosin repressed the effects of LPS in HK-2 cells (Fig. 2G). Moreover, compared with those in the control group, the levels of TNF-α, IL-1β, and IL-6 were increased in the LPS-stimulated group, and they were decreased by calycosin treatment (Fig. 3A–C).
Effect of calycosin (1) on the apoptosis of lipopolysaccharide-induced HK-2 cells. A The CCK-8 assay was carried out to evaluate cell viability. B The lactate dehydrogenase assay was performed to measure LDH activity in calycosin-treated LPS-induced HK-2 cells. C and D Flow cytometry was conducted to assess cell apoptosis. E and F Western blotting was performed to quantify the expression patterns of apoptosis-related protein. G Capase-3 activity assay was carried out to measure caspase-3 activity. **p < 0.01 vs. control; #, ##p < 0.05, 0.01 vs. LPS. Data are presented as mean ± SD of three independent experiments
Inhibition of Nrf2 and HO-1 Expression
To explore the potential mechanisms by which calycosin prevents LPS-induced LN, HK-2 cells were pre-treated with calycosin (20, 40, and 80 μM) for 2 h, and then cultured with 1 μg/ml LPS for 24 h, and the Nrf2/HO-1 pathway was analyzed. Compared with those in the control group, the protein and mRNA levels of Nrf2 and HO-1 were altered in the LPS-stimulated group, and calycosin reversed this phenomenon in a dose-dependent manner (Fig. 4A–C).
ML385 Inhibited Calycosin Protective Effect
To determine whether calycosin protects against lupus nephritis via Nrf2/HO-1 signaling, HK-2 cells were treated with calycosin (80 μM) alone or in combination with 1 µM ML385 for 2 h, and then stimulated for 24 h with 1 μg/ml LPS. The results indicated that the protein and mRNA levels of Nrf2 and HO-1 increased in the LPS + calycosin group, and ML385 suppressed this increase (Fig. 5A–C).
The CCK-8 assay results showed that calycosin enhanced cell growth, which was inhibited by ML385 treatment (Fig. 6A). The LDH activity was significantly suppressed by calycosin compared with that in the LPS group but increased in the LPS + calycosin + ML385 group (Fig. 6B). The flow cytometry assay revealed that calycosin treatment decreased apoptosis compared with that in the LPS group, but this decrease was reversed by ML385 treatment (Fig. 6C and D). Furthermore, the protein level of cleaved caspase3 was decreased in the LPS + calycosin group, and it was increased by ML385 treatment (Fig. 6E and F). Additionally, the caspase-3 activity was substantially decreased by calycosin compared with that in the LPS group, but it was increased by ML385 treatment (Fig. 6G). Finally, the levels of TNF-α, IL-1β, and IL-6 were altered by calycosin treatment, and increased by ML385 (Fig. 7A–C). These results suggest that calycosin alleviates lupus nephritis by activating the Nrf2/HO-1 signaling pathway.
ML385 reversed the effects of calycosin (1) on LPS-induced apoptosis in HK-2 cells. A The CCK-8 assay was performed to determined cell viability. B The LDH assay was carried out to measure LDH activity. C and D Apoptosis rate of HK-2 cells was determined using flow cytometry. E and F The expression pattern of cleaved caspase3 was determined using western blotting. G Capase3 activity assay was used to measure caspase3 activity in HK-2 cells. **p < 0.01 vs. LPS; ##p < 0.01 vs. LPS + calycosin-80. Data are presented as mean ± SD of three independent experiments
Nearly 50% of Chinese patients with systemic lupus erythematosus have LN, which is higher than that in Caucasians with SLE (Li et al. 2019). Lupus nephritis is mainly caused by renal injury due to circulatory or in situ immune complex deposition, whereas systemic lupus erythematosus rarely damages the kidney through non-immune complex pathways (such as lupus interstitial nephritis) or renal vascular lesions (Qi et al. 2018; Liang et al. 2021). The 10-year renal survival rate of patients with lupus nephritis in China is 81–98%, and this disease is one of the common causes of end-stage renal disease and an important cause of death in patients with systemic lupus erythematosus (Quan et al. 2022; Lv et al. 2022). Over the past decade, the lupus nephritis treatment has become more individualized, and the application of new immunosuppression schemes, especially multitarget therapy, has substantially improved the response rate to lupus nephritis treatment.
Recent studies have revealed that calycosin exhibits biological activity against various human diseases. For instance, Huang et al. (2022) reported that calycosin alleviates diabetic kidney disease by modulating ferroptosis. Ma et al. (2022) showed that calycosin improves atherosclerosis by regulating autophagy. Jin et al. (2022) reported that calycosin optimized bone loss in a rat model. Furthermore, a previous study demonstrated a potential therapeutic effect of calycosin against human papillary thyroid cancer (Qu et al. 2022). Elsherbiny et al. (2020) reported that calycosin markedly improved high-fat diet/STZ-induced renal injury and dysfunction by modulating IL33/ST2 signaling, inflammatory cytokine levels, oxidative stress, and fibrotic processes. However, only a few studies have investigated the effects of calycosin against lupus nephritis. In the present study, we found that calycosin alleviated LPS-induced lupus nephritis in vitro.
Nrf2/HO-1 signaling is a critical cytoprotective mechanism. Growing evidence indicates that the Nrf2/HO-1 signaling pathway plays a critical role in human diseases. Yu et al. (2019) suggested that the activation of the Nrf2/HO-1 signaling pathway could improve myocardial ischemia. Nrf2/HO-1 signaling is reportedly involved in the anti-inflammatory response of icariin (El-Shitany and Eid 2019). Wang et al. (2022) demonstrated that Nrf2/HO-1 is related to lung ischemia–reperfusion and may be a potential therapeutic target. Moreover, a previous study indicated that melatonin alleviated nonalcoholic fatty liver disease (NAFLD) by downregulating Nrf2/HO-1 (Joshi et al. 2021). A previous study also indicated that the activation of Nrf2-HO-1 signaling may offer a potential approach for the development of novel therapeutic agents for kidney diseases (Uddin et al. 2021). In our study, we found that calycosin treatment notably inhibited LPS-induced LN via the Nrf2/HO-1 pathway.
Conclusion
Calycosin (1) mitigated LPS-induced lupus nephritis in vitro, as well as decreased inflammation, apoptosis, and the activated Nrf2/HO-1 axis in LPS-stimulated HK-2 cells. Therefore, our results suggest that calycosin is a novel therapeutic agent against lupus nephritis.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anders HJ, Saxena R, Zhao MH, Parodis I, Salmon JE, Mohan C (2020) Lupus Nephritis. Nat Rev Dis Primers 6:7. https://doi.org/10.1038/s41572-019-0141-9
Chen Z, Zhong H, Wei J, Lin S, Zong Z, Gong F, Huang X, Sun J, Li P, Lin H, Wei B, Chu J (2019) Inhibition of Nrf2/HO-1 signaling leads to increased activation of the NLRP3 inflammasome in osteoarthritis. Arthritis Res Ther 21:300. https://doi.org/10.1186/s13075-019-2085-6
Chen G, Xu H, Xu T, Ding W, Zhang G, Hua Y, Wu Y, Han X, Xie L, Liu B, Zhou Y (2022) Calycosin reduces myocardial fibrosis and improves cardiac function in post-myocardial infarction mice by suppressing TGFBR1 signaling pathways. Phytomedicine 104:154277. https://doi.org/10.1016/j.phymed.2022.154277
Elsherbiny NM, Said E, Atef H, Zaitone SA (2020) Renoprotective effect of calycosin in high fat diet-fed/STZ injected rats: effect on IL-33/ST2 signaling, oxidative stress and fibrosis suppression. Chem Biol Interact 315:108897. https://doi.org/10.1016/j.cbi.2019.108897
El-Shitany NA, Eid BG (2019) Icariin modulates carrageenan-induced acute inflammation through HO-1/Nrf2 and NF-kB signaling pathways. Biomed Pharmacother 120:109567. https://doi.org/10.1016/j.biopha.2019.109567
Ge M, Yao W, Yuan D, Zhou S, Chen X, Zhang Y, Li H, Xia Z, Hei Z (2017) Brg1-mediated Nrf2/HO-1 pathway activation alleviates hepatic ischemia-reperfusion injury. Cell Death Dis 8:e2841. https://doi.org/10.1038/cddis.2017.236
Huang D, Shen P, Wang C, Gao J, Ye C, Wu F (2022) Calycosin plays a protective role in diabetic kidney disease through the regulation of ferroptosis. Pharm Biol 60:990–996. https://doi.org/10.1080/13880209.2022.2067572
Jin X, Wang H, Liang X, Ru K, Deng X, Gao S, Qiu W, Huai Y, Zhang J, Lai L, Li F, Miao Z, Zhang W, Qian A (2022) Calycosin prevents bone loss induced by hindlimb unloading. NPJ Microgravity 8:23. https://doi.org/10.1038/s41526-022-00210-x
Joshi A, Upadhyay KK, Vohra A, Shirsath K, Devkar R (2021) Melatonin induces Nrf2-HO-1 reprogramming and corrections in hepatic core clock oscillations in non-alcoholic fatty liver disease. FASEB J 35:e21803. https://doi.org/10.1096/fj.202002556RRR
Li D, Shi G, Wang J, Zhang D, Pan Y, Dou H, Hou Y (2019) Baicalein ameliorates pristane-induced lupus nephritis via activating Nrf2/HO-1 in myeloid-derived suppressor cells. Arthritis Res Ther 21:105. https://doi.org/10.1186/s13075-019-1876-0
Liang CL, Jiang H, Feng W, Liu H, Han L, Chen Y, Zhang Q, Zheng F, Lu CJ, Dai Z (2021) Total glucosides of paeony ameliorate pristane-induced lupus nephritis by inducing PD-1 ligands+ macrophages via activating IL-4/STAT6/PD-L2 signaling. Front Immunol 12:683249. https://doi.org/10.3389/fimmu.2021.683249
Lv F, He Y, Xu H, Li Y, Han L, Yan L, Lang H, Zhao Y, Zhao Z, Qi Y (2022) CD36 aggravates podocyte injury by activating NLRP3 inflammasome and inhibiting autophagy in lupus nephritis. Cell Death Dis 13:729. https://doi.org/10.1038/s41419-022-05179-9
Ma C, Wu H, Yang G, Xiang J, Feng K, Zhang J, Hua Y, Kang L, Fan G, Yang S (2022) Calycosin ameliorates atherosclerosis by enhancing autophagy via regulating the interaction between KLF2 and MLKL in apolipoprotein E gene-deleted mice. Br J Pharmacol 179:252–269. https://doi.org/10.1111/bph.15720
Qi YY, Zhou XJ, Cheng FJ, Hou P, Ren YL, Wang SX, Zhao MH, Yang L, Martinez J, Zhang H (2018) Increased autophagy is cytoprotective against podocyte injury induced by antibody and interferon-α in lupus nephritis. Ann Rheum Dis 77:1799–1809. https://doi.org/10.1136/annrheumdis-2018-213028
Qu N, Qu J, Huang N, Zhang K, Ye T, Shi J, Chen B, Kan C, Zhang J, Han F, Hou N, Sun X, Pan R (2022) Calycosin induces autophagy and apoptosis via Sestrin2/AMPK/mTOR in human papillary thyroid cancer cells. Front Pharmacol 13:1056687. https://doi.org/10.3389/fphar.2022.1056687
Quan XY, Chen HT, Liang SQ, Yang C, Yao CW, Xu YZ, Liu HF, An N (2022) Revisited cyclophosphamide in the treatment of lupus nephritis. Biomed Res Int 2022:8345737. https://doi.org/10.1155/2022/8345737
Rovin BH, Furie R, Teng YKO, Contreras G, Malvar A, Yu X, Ji B, Green Y, Gonzalez-Rivera T, Bass D, Gilbride J, Tang CH, Roth DA (2022) A secondary analysis of the Belimumab International Study in lupus nephritis trial examined effects of belimumab on kidney outcomes and preservation of kidney function in patients with lupus nephritis. Kidney Int 101:403–413. https://doi.org/10.1016/j.kint.2021.08.027
Tonelli C, Chio IIC, Tuveson DA (2018) Transcriptional regulation by Nrf2. Antioxid Redox Signal 29:1727–1745. https://doi.org/10.1089/ars.2017.7342
Uddin MJ, Kim EH, Hannan MA, Ha H (2021) Pharmacotherapy against oxidative stress in chronic kidney disease: promising small molecule natural products targeting Nrf2-HO-1 signaling. Antioxidants 10:258. https://doi.org/10.3390/antiox10020258
Wang Y, Yang C, Elsheikh NAH, Li C, Yang F, Wang G, Li L (2019) HO-1 reduces heat stress-induced apoptosis in bovine granulosa cells by suppressing oxidative stress. Aging 11:5535–5547. https://doi.org/10.18632/aging.102136
Wang Y, Gao L, Chen J, Li Q, Huo L, Wang Y, Wang H, Du J (2021) Pharmacological modulation of Nrf2/HO-1 signaling pathway as a therapeutic target of Parkinson’s disease. Front Pharmacol 12:757161. https://doi.org/10.3389/fphar.2021.757161
Wang Y, Dong Z, Zhang Z, Wang Y, Yang K, Li X (2022) Postconditioning with irisin attenuates lung ischemia/reperfusion injury by suppressing ferroptosis via induction of the Nrf2/HO-1 signal axis. Oxid Med Cell Longev 2022:9911167. https://doi.org/10.1155/2022/9911167
Yan X, Yu A, Zheng H, Wang S, He Y, Wang L (2019) Calycosin-7-O-β-D-glucoside attenuates OGD/R-induced Ddmage by preventing oxidative stress and neuronal apoptosis via the SIRT1/FOXO1/PGC-1α pathway in HT22 cells. Neural Plast 2019:8798069. https://doi.org/10.1155/2019/8798069
Yu F, Haas M, Glassock R, Zhao MH (2017) Redefining lupus nephritis: clinical implications of pathophysiologic subtypes. Nat Rev Nephrol 13:483–495. https://doi.org/10.1038/nrneph.2017.85
Yu H, Chen B, Ren Q (2019) Baicalin relieves hypoxia-aroused H9c2 cell apoptosis by activating Nrf2/HO-1-mediated HIF1α/BNIP3 pathway. Artif Cells Nanomed Biotechnol 47:3657–3663. https://doi.org/10.1080/21691401.2019.1657879
Yu C, Li P, Dang X, Zhang X, Mao Y, Chen X (2022) Lupus nephritis: new progress in diagnosis and treatment. J Autoimmun 132:102871. https://doi.org/10.1016/j.jaut.2022.102871
Zhang H, Liu Z, Zhou M, Liu Z, Chen J, Xing C, Lin H, Ni Z, Fu P, Liu F, Chen N, He Y, Liu J, Zeng C, Liu Z (2017) Multitarget therapy for maintenance treatment of lupus nephritis. J Am Soc Nephrol 28:3671–3678. https://doi.org/10.1681/ASN.2017030263
Zhang Z, Lin M, Wang J, Yang F, Yang P, Liu Y, Chen Z, Zheng Y (2021) Calycosin inhibits breast cancer cell migration and invasion by suppressing EMT via BATF/TGF-β1. Aging 13:16009–16023. https://doi.org/10.18632/aging.203093
Funding
This study was supported by Wuhan Medical Research Project (grant no. WX21D37).
Author information
Authors and Affiliations
Contributions
YY contributed to data collection, statistical analysis, data interpretation, and manuscript preparation. PS contributed to data collection and manuscript preparation. All authors have read and approved the final manuscript.
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, Y., Sun, P. Calycosin Alleviates Lupus Nephritis by Activating the Nrf2/HO-1 Signaling Pathway. Rev. Bras. Farmacogn. 33, 1052–1059 (2023). https://doi.org/10.1007/s43450-023-00435-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00435-1