Abstract
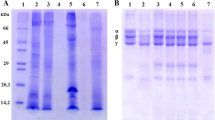
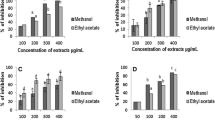
Moringa oleifera Lam., Moringaceae, ethyl acetate soluble fraction has been documented to contain bioactive antivenom chemicals against Bitis arietans venom toxicities. Snake venom metalloproteinase is the principal enzyme of B. arietans venom exhibiting severe haemorrhage and haemostatic disturbances in envenomed victims. In this current study, B. arietans snake venom metalloproteinase was isolated using ion-exchange chromatography and gel filtration while SDS-PAGE was used to determine enzyme molecular weight. M. oleifera ethyl acetate soluble fraction was fractionated using column and thin-layer chromatography into subfractions and tested against B. arietans snake venom metalloproteinase. EchiTab polyvalent antivenom was used as the standard drug. The compounds present in the most active subfraction were identified using GCMS. The molecular weight of isolated enzyme was 43.28 kDa with specific activity of 3.06 μmol/min/mg and percentage yield of 18.5%. A total of 8 subfractions were obtained after fractionation and labelled ethyl acetate subfractions F1-8. All subfractions showed significant (p < 0.05) inhibition against B. arietans snake venom metalloproteinase activity. However, subfraction F5 demonstrated the highest inhibition against B. arietans snake venom metalloproteinase activity with total inhibition at all concentrations compared to other subfractions and antivenom. Subfraction F5 displayed substantial inhibition against the haemorrhagic, haemolytic, and anticoagulant activities of B. arietans venom with a dose-dependent effect. The major bioactive compounds of subfraction F5 were γ-sitosterol (5.92%), kaempferol (5.9%), quercetin (5.28%), n-hexadecanoic acid (5.23%), and n-pentacosane (5.22%) of which some were previously reported as potential snake venom inhibitors. Results showed that subfraction F5 possesses potential antivenom compounds that could be explored for snakebite treatment.
Graphical Abstract




Similar content being viewed by others
Data Availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adeyi AO, Ajisebiola BS, Adeyi OE, Alimba GC, Okorie UG (2020) Antivenom activity of Moringa oleifera leave against pathophysiological alterations, somatic mutation and biological activities of Naja nigricollis venom. Sci Afr 8:e00356. https://doi.org/10.1016/j.sciaf.2020.e00356
Adeyi AO, Adeyemi SO, Effiong EOP, Ajisebiola BS, Adeyi OE, James AS (2021) Moringa oleifera extract extenuates Echis ocellatus venom-induced toxicities, histopathological impairments and inflammation via enhancement of Nrf2 expression in rats. Pathophysiology 28:98–115. https://doi.org/10.3390/biology9100313
Adeyi AO, Mustapha KK, Ajisebiola BS, Adeyi OE, Damilohun SM, Raphael O (2022) Inhibition of Echis ocellatus venom metalloprotease by flavonoid-rich ethyl acetate sub fraction of Moringa oleifera (Lam.) leaves; in vitro and in silico approaches. Toxin Rev 41:476–486. https://doi.org/10.1080/15569543.2021.1893334
Ajisebiola BS, Solomon R, Anwar U, Adeyi AO (2021) Neutralization of Bitis arietans venom-induced pathophysiological disorder, biological activities and genetic alterations by Moringa oleifera leaves. Toxin Rev 40:847–858. https://doi.org/10.1080/15569543.2020.1793780
Ajisebiola BS, Oyinlola RR, Akintunde SO, Paul O, Adeyi AO (2022) Ethyl acetate fraction of Moringa oleifera leaves mollified toxicological activities actuated by Bitis arietans (Puff adder) venom. Nig J Pure Appl Sci 35:4350–4362. https://doi.org/10.48198/NJPAS/22.A08
Alam MI (2014) Inhibition of toxic effects of viper and cobra venom by Indian medicinal plants. Pharmacol Pharm 5:828–837. https://doi.org/10.4236/pp.2014.58093
Baggai BI, Yusuf PO, Alloh FT (2020) Inhibitory studies of Tamarindus indica seed extract and fractions on hematological activities of Bitis arietans venom. J Adv Biol Biotechnol 23:48–57. https://doi.org/10.9734/jabb/2020/v23i430152
Cuccioloni M, Mozzicafreddo M, Bonfili L, Cecarini V, Eleuteri AM, Angeletti M (2009) Natural occurring polyphenols as template for drug design. Focus on serine proteases. Chem Biol Drug Des 74:1–15. https://doi.org/10.1111/j.1747-0285.2009.00836.x
Currier RB, Harrison RA, Rowley PD, Laing GD, Wagstaff SC (2010) Intra-specific variation in venom of the African puff adder (Bitis arietans): differential expression and activity of snake venom metalloproteinases (SVMPs). Toxicon 55:864–873. https://doi.org/10.1016/j.toxicon.2009.12.009
Escalante T, Rucavado A, Fox JW, Gutiérrez JM (2011) Key events in microvascular damage induced by snake venom hemorrhagic metalloproteinases. J Proteomics 74:1781–1794. https://doi.org/10.1016/j.jprot.2011.03.026
Fernandez MT, Mira ML, Florêncio MH, Jennings KR (2002) Iron and copper chelation by flavonoids: an electrospray mass spectrometry study. J Inorg Biochem 92:105–111. https://doi.org/10.1016/s0162-0134(02)00511-1
Fernandez S, Hodgson W, Chaisakul J, Kornhauser R, Konstantakopoulos N, Smith AL, Kuruppu S (2014) In vitro toxic effects of puff adder (Bitis arietans) venom, and their neutralization by antivenom. Toxins 6:1586–1597. https://doi.org/10.3390/toxins6051586
Girish KS, Kemparaju K (2006) Inhibition of Naja naja venom hyaluronidase: role in the management of poisonous bite. Life Sci 78:1433–1440. https://doi.org/10.1016/j.lfs.2005.07.015
Gomes A, Archita S, Ipshita C, Chakravarty AK (2007) Viper and cobra venom neutralization by sitosterol and stigmasterol isolated from the root extract of Pluchea indica Less. (Asteraceae). Phytomedicine 14:637–643. https://doi.org/10.1016/j.phymed.2006.12.020
Gopi K, Renu K, Jayaraman G (2014) Inhibition of Naja naja venom enzymes by the methanolic extract of Leucas aspera and its chemical profile by GC–MS. Toxicol Rep 1:667–673. https://doi.org/10.1016/j.toxrep.2014.08.012
Habib AG, Musa BM, Iliyasu G, Hamza M, Kuznik A, Chippaux JP (2020) Challenges and prospects of snake antivenom supply in sub-Saharan Africa. PLoS Negl Trop Dis 14:e0008374. https://doi.org/10.1371/journal.pntd.0008374
Jedinák A, Maliar T, Grancai D, Nagy M (2006) Inhibition activities of natural products on serine proteases. Phytother Res 20:214–217. https://doi.org/10.1002/ptr.1836
Kamiguti AS (2005) Platelets as targets of snake venom metalloproteinases. Toxicon 45:1041–1049. https://doi.org/10.1016/j.toxicon.2005.02.026
Megale AAA, Magnoli FC, Kuniyoshi AK, Iwai LK, Tambourgi DV, Portaro FCV, da Silva WD (2018) Kn-Ba: a novel serine protease isolated from Bitis arietans snake venom with fibrinogenolytic and kinin-releasing activities. J Venom Anim Toxins Incl Trop Dis 24:38. https://doi.org/10.1186/s40409-018-0176-5
Megale ÂAA, Magnoli FC, Guidolin FR, Godoi KS, Portaro FCV, Dias-da-Silva W (2021) Bitis arietans snake venom and Kn-Ba, a snake venom serine protease, induce the production of inflammatory mediators in THP-1 macrophages. Toxins 13:906. https://doi.org/10.3390/toxins13120906
Mira L, Fernandez MT, Santos M, Rocha R, Florêncio MH, Jennings KR (2002) Interactions of flavonoids with iron and copper ions: a mechanism for their antioxidant activity. Free Radic Res 36:1199–1208. https://doi.org/10.1080/1071576021000016463
National Research Council (2010) Guide for the care and use of laboratory animals. National Academies Press, Washington, DC, USA
Obert B, Romijn RA, Houllier A, Huizinga EG, Girma JP (2006) Characterization of bitiscetin-2, a second formo of bitiscetin from the venom of Bitis arietans: comparison of its biding site with the collagen-biding site on the von Willebrand fator A3-domain. J Thromb Haemost 4:1596–1601. https://doi.org/10.1111/j.1538-7836.2006.01994.x
Okoro PN, Ibrahim S, Inuwa HM, Irobekhian S, Okoduwa R (2017) Evaluation of mango seed kernel methanolic extract on metalloproteases in carpet viper (Echis ocellatus) venom: an in vitro experiment. Iran J Toxicol 11:23–30
Olaniyan OV, Ebuehi OAT (2022) Isolation and purification of Bitis arietans (puff adder) venom proteins and its effect on blood chemistry of envenomated wistar rats. Ann Psychiatr Clin Neurosci 5:1045
Olaoba OT, Dos Santos PK, Selistre-de-Araujo HS, de Souza DHF (2020) Snake venom metalloproteinases (SVMPs): a structure-function update. Toxicon X 7:100052. https://doi.org/10.1016/j.toxcx.2020.100052
Patiño AC, Pereañez JA, Núñez V, Benjumea DM, Fernandez M, Rucavado A, Sanz L, Calvete JJ (2010) Isolation and biological characterization of Batx-I, a weak hemorrhagic and fibrinogenolytic PI metalloproteinase from Colombian Bothrops atrox venom. Toxicon 56:936–943. https://doi.org/10.1016/j.toxicon.2010.06.016
Preciado LM, Comer J, Núñez V, Rey-Súarez P, Pereañez JA (2018) Inhibition of a snake venom metalloproteinase by the flavonoid myricetin. Molecules 23:2662. https://doi.org/10.3390/molecules23102662
Rajesh KS, Bharath BR, Rao CV, Bhat KI, Bhat CKS, Bhat P (2017) Neutralization of Naja naja venom induced lethality, edema and myonecrosis by ethanolic root extract of Coix lacryma-jobi. Toxicol Rep 4:637–645. https://doi.org/10.1016/j.toxrep.2017.11.005
Rodrigues VM, Soares AM, Guerra-Sá R, Rogrigues V, Fontes MR, Giglio JR (2000) Structural and functional characterization of neuwiedase, a non-hemorrhagic fibrin(ogen)olytic. Arch Biochem Biophys 38:213–224. https://doi.org/10.1006/abbi.2000.1958
Sani I, Umar R, Hassan S, Faruq U, Abdulhamid A, Bello F, Fakai M (2019) Major enzymes from snake venoms: mechanisms of action and pharmacological applications. Asian J Biol Sci 12:396–403. https://doi.org/10.3923/ajbs.2019.396.403
Santhosh MS, Sundaram MS, Sunitha K, Kemparaju K, Girish KS (2013) Viper venom-induced oxidative stress and activation of inflammatory cytokines: a therapeutic approach for overlooked issues of snakebite management. Inflamm Res 62:721–731. https://doi.org/10.1007/s00011-013-0627-y
Singh P, Yasir M, Khare R, Tripathi MK, Shrivastava R (2020) Snake venom phospholipase A2 and its natural inhibitors. Nat Prod Sci 26:259–267. https://doi.org/10.20307/nps.2020.26.4.259
Vulfius CA, Gorbacheva EV, Starkov VG, Osipov AV, Kasheverov IE, Andreeva TV, Astashev ME, Tsetlin VI, Utkin YN (2011) An unusual phospholipase A2 from puff adder Bitis arietans venom—a novel blocker of nicotinic acetylcholine receptors. Toxicon 57:787–793. https://doi.org/10.1016/j.toxicon.2011.02.013
WHO (2019) Snakebite envenoming: a strategy for prevention and control. Geneva: World Health Organization. Licence: CC BY-NC-SA 3.0 IGO
Yusuf PO, Mamman M, Ajagun E, Suleiman MM, Kawu MU, Shittu M, Isa HI, Tauheed M, Yusuf A (2015) Snakes responsible for bites in North-Eastern Nigeria – a hospital based survey. J Environ Sci Toxicol Food Technol 9:118–121. https://doi.org/10.9790/2402-0992118121
Zhu Y, Yin Q, Yang Y (2020) Comprehensive investigation of Moringa oleifera from different regions by simultaneous determination of 11 polyphenols using UPLC-ESI-MS/MS. Molecules 25:676. https://doi.org/10.3390/molecules25030676
Author information
Authors and Affiliations
Contributions
BSA conceptualized and designed the study under the guidance of AOA. BSA and FTB executed the antivenom experiments. PMO did the extract fractionation. BSA, JOO, and AA were responsible for drafting and reviewing the final manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
Animal experimental protocol in this study followed the guidelines of the University of Ibadan-Animal Care and Use Research Ethics Committee (UI-ACUREC) with approved number: UI-ACUREC/19/0030. The animal experimental procedures complied with the National Research Council’s publication on guide for the care and use of laboratory animals (National Research Council 2010).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ajisebiola, B.S., Bello, F.T., Osamudiamen, P.M. et al. Inhibition of Metalloproteinase from Bitis arietans Venom In Vitro by the Ethyl Acetate Fraction of Moringa oleifera and Its Chemical Profile. Rev. Bras. Farmacogn. 33, 822–830 (2023). https://doi.org/10.1007/s43450-023-00416-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00416-4