Abstract
Baicalin is one of the bioactive flavonoid glycosides isolated from the dried root of Scutellaria baicalensis Georgi, Lamiaceae, with antiviral properties. In recent years, the antiviral activity of baicalin has been widely investigated to explore its molecular mechanism of action. In this mini-review, the molecular mechanisms of action of baicalin as an antiviral agent are evaluated, which included three categories: the inhibition or stimulation of JAK/STAT, TLRs, and NF-κB pathways; up or down modulation of the expression levels of IFN, IL, SOCS1/3, PKR protein, Mx1 protein, and AP-1 protein; and inhibition of cell apoptosis caused by virus infection. In addition, clinical studies of baicalin are also discussed. This literature search suggested that baicalin can serve as a potential candidate for the development of a novel broad-spectrum antiviral drug.
Graphical abstract

Similar content being viewed by others
Introduction
In recent years, the public health security has attracted wide attention from people all over the world, particularly after COVID-19 pandemic around the world. Given the scarcity of efficacious and safe vaccines or drugs for coronavirus disease 2019, natural products are a non-orthodox therapeutic approach in the management of the disease that has gained attention, especially in developing countries (Chinsembu 2020; Komolafe et al. 2021). Several viruses cause a biochemical imbalance in the infected cell resulting in oxidative stress that promotes cellular damage through protein denaturation, changes in the functions of nucleic acids, lipid peroxidation, and cell death. These effects may be associated with the development of pathologies and worsening of symptoms. Antioxidant properties of polyphenolic compounds (active redox agents), such as flavonoids, have been associated with the presence of aromatic phenolic rings that promote the electron donation and hydrogen atom transfer to free radicals, acting as free radical scavengers, reducing agents, and quenchers of single oxygen formation that can be used as antiviral agents (Diniz et al. 2020).
Baicalin (1) is the main flavonoid glucoside of Scutellaria baicalensis Georgi, Lamiaceae, which has antiviral, bacteriostatic, antitumor, and anti-oxidative effects (Long et al. 2019). This medicinal plant, known with the common name of Baikal skullcap or Chinese skullcap, is native to China, Korea, Mongolia, and in the Russian Far East and Siberia. It is one of the 50 fundamental herbs used in the traditional Chinese medicine (TCM), where it has the name of huáng qín (Chinese: 黄芩). This plant was originally documented in Shennong Bencao Jing, the first Chinese Materia Medica during the Eastern Han Dynasty (25 CE–220 CE) and described the crude dug as the dried root with a bitter taste and cold in nature (Sun and Sun, 1995). Medicinal values of the Chinese skullcap include heat-clearing, elimination of dampness, detoxification, and bleeding prevention (Fang et al. 2020). This plant is used for the prophylaxis and treatment of hepatitis, atherosclerosis, dysentery, ulcerative colitis, inflammations, metabolomic disorders, hypertension, hyperlipidemia, type 2 diabetes, and bacterial and viral infections in the respiratory and gastrointestinal tracts (Dinda et al. 2017; Liang et al. 2019; Fang et al. 2020; Su et al. 2020). Baicalin (1) is regarded as the therapeutic chemical marker for quality control of the S. baicalensis roots (Tong et al. 2012). To improve the therapeutic effects, different herbs are usually combined to produce the TCM formula. The interactions of herbs in the prescription may alter the pharmacokinetics of baicalin. As a major component of TCM formulas, S. baicalensis is widely prescribed with herbs such as Coptidis rhizoma (Lou et al. 2021) and Bupleuri radix (Huang et al. 2019).

Baicalin (1) has been found to have antiviral effect on SARS-CoV-2 (2019-nCoV) in vitro (Chen et al. 2020; Jo et al. 2020; Russo et al. 2020; Su et al. 2020). Baicalin (1) and baicalein, its aglycone, were characterized as the first noncovalent, non-peptidomimetic inhibitors of SARS-CoV-2 3CL protease, which is indispensable for coronavirus replication, and exhibited potent antiviral activities in a cell-based system (Su et al. 2020). With increasing interest in the antiviral effect of baicalin, many studies have been carried out focusing on its pharmacological actions. We have summarized baicalin’s mechanism of action reported in the literature in different cytokines and cell pathways. The pharmacological value of baicalin (1) for the development of a broad-spectrum antiviral drug is further discussed.
Search Strategy
A systematic search of the literature was performed in PubMed, Web of Science, China National Knowledge Infrastructure, Scopus, Embase, Google Scholar, and SciFinder Scholar database. Search on baicalin was done by using combinations of keywords, including “Huang Qin,” “baicalin,” “Scutellaria baicalensis Georgi, Labiatae,” “pharmacological activity,” “Physicochemical Properties,” “Pharmacokinetics,” “toxicity,” “quality control,” and “clinical study.” Considering the language limitation, this review only refers to Chinese and English texts. In the present study, the existing animal and clinical evidence regarding the therapeutic effects of baicalin on antiviral will be reviewed in addition to its possible mechanism.
Discussion
Physicochemical Properties
Baicalin (1) is practically insoluble in water (0.054 g/l) and soluble in alcohol, with a molecular weight and melting point of 446.4 g/mol and 222–224 °C, respectively. The basic structure of baicalin corresponds to a flavonoid, a benzopyran ring with a phenolic moiety, as the aglycone with a glucuronic acid moiety forming an anomeric linkage with the C-7 hydroxyl position of the aglycone which contributes to the solubility of the molecules in polar protic solvents. Log P 1.1 indicates that the molecule is poorly lipophilic. The presence of a di-hydroxylated (catechol) substitution on ring A is the main property of the active redox polyphenolic compounds. Catechols in flavonoids and polyphenols are able to form chelates with metals and are reactive in their oxidized form (like quinonas) for radical scavenging and enzyme inhibition with nucleophiles present in lateral protein chains such as cysteine and lysine.
Baicalin (1) is unstable in the basic pH and stable in weakly acidic pH. Baicalin contains two phenolic hydroxyl and one carboxyl groups. Therefore, the acid property of baicalin was characterized in terms of three acid dissociation constants (Ka): 5.05, 7.6, and 10.1 (Liang et al. 2009). It is reported that baicalin has poor water solubility (67.03 ± 1.60 μg/ml), low permeability (Papp = 0.037 × 10−6 cm/s) and low absolute bioavailability (2.2 ± 0.2%) (Xing et al. 2005; Wu et al. 2014). Thus, one of the challenges with baicalin is to improve its bioavailability.
Quantification
Several analytical methods, mainly HPLC and UPLC coupled to UV, ECD, MS, and MS/MS, were reported for the quantitative determination of baicalin (1) and its related glycosides in biological samples. Li et al. (2011) reported the HPLC–UV method to quantify the content of baicalin, their metabolites in rat’s blood serum after oral administration of the products containing S. baicalensis. It is also reported that the absorption of baicalin occurs at two sites: in upper intestine in the form of glycoside and in the colon in the form of aglycone (Lu et al. 2007). Furthermore, HPLC–ECD (Kotani et al. 2006) and UPLC–MS (Wang et al. 2009) methods were developed for the qualitative and quantitative determination of baicalin, puerarin, wogonoside, and liquiritin in rat’s blood. This method is being used to determine the quality of Gegen-qinlian Tang and other S. baicalensis products. By application of a LC–MS method, it was reported that xiao-chai-hu tang, a herbal formula for chronic hepatitis B containing S. baicalensis (Kong et al. 2018), has a significant influence on the absorption and elimination of baicalin and wogonoside (Zhu et al. 2010). A LC–MS/MS method was reported for determining the analysis of the pharmacokinetics of compounds in rats upon the oral administration of various S. baicalensis preparations (as per Chinese Materia Medica) (Kim et al. 2006; Feng et al. 2010; Ye et al. 2010).
Bioavailability and Pharmacokinetics
Bioavailability
Baicalin (1) is categorized as class IV drug under BCS classification. Due to the glycosidic nature of this flavone, baicalin is too polar to cross the lipid bilayer by passive diffusion and is thus poorly absorbed in the intestinal tract. Conversely, baicalein, the aglycone of 1, demonstrates a good permeability due to its great lipophilicity and is well absorbed in the gastrointestinal tract. Baicalin is moderately absorbed in the stomach but poorly in the small intestine and colon, while baicalein had better absorption than baicalin in all segments of the gastrointestinal tract. However, baicalin, rather than baicalein, is the major component in the systemic circulation following oral administration of baicalein (Liu and Jiang 2006).
To improve its bioavailability, several formulations such as cyclodextrin complexes, self-nanoemulsifying systems, micelles, nanocrystals, and gels were developed (Jakab et al. 2019; Xing et al. 2005). It was reported that the efflux of baicalin might be mediated by ATP-binding cassette (ABC) transporters (Kalapos-Kovács et al. 2015). Jakab et al. (2019) and Li et al. (2018) have developed cyclodextrin complexes for baicalin via the inclusion complexation by supercritical fluid encapsulation technique to improve the bioavailability of this flavonoid glucoside. Additionally, owing to hydrogen bonding and van der Waals interactions, the coordination complex of baicalin with the oxovanadium (IV) cation, Na4[VO(baic)2]·6H2O VIVO(baic), has stronger binding with bovine serum albumin than baicalin (Martínez Medina et al. 2017). A self-emulsifying microemulsion drug delivery system (SMEDDS) was developed for baicalin using a phospholipid complex (PC) by a solvent evaporation method. The formulation has improved the bioavailability, and the relative bioavailability of baicalin–PC (1:2)–SMEDDS was 220.37% of baicalin in rats (Wu et al. 2014). Furthermore, the nanoemulsion was reported to improve the absorption of baicalin from the intestinal tract compared to baicalin suspension. The absorption rate constant (Ka) and apparent permeability coefficient (Papp) were increased by 6.14-fold and 6.25-fold, respectively (Wu et al. 2018). In addition, baicalin nanoemulsions were reported to increase the saturation solubility of baicalin and strengthened their interaction with the mucin layer in epithelial tissues leading to an increased uptake in the intestine. The AUC(0-t) and Cmax of baicalin from a lipid-based nanoemulsion was reported to be significantly higher (11.5-fold) than baicalin powder for lymphatic system absorption (Xu et al. 2019). The in vivo disposition of baicalin is affected by combinations of other herbs, and baicalin can interact with other co-administered drugs due to competition between metabolic enzymes and protein binding (Huang et al. 2019). Some other studies have reported the development of nanocapsules and nanocrystals which have improved the absorption and bioavailability of baicalin in pre-clinical studies (El-Gogary et al. 2019; Jin et al. 2014).
Pharmacokinetics
Baicalin (1) is a flavone glycoside mainly absorbed in the stomach but poorly absorbed in the small intestine and colon in comparison to its aglycone, baicalein (Liu and Jiang 2006). Baicalin displays a distinct pharmacokinetic profile including gastrointestinal hydrolysis, enterohepatic recycling, carrier-mediated transport, and complicated metabolism. Numerous studies have reported that the enzymes, β-glucuronidase and UDP-glucuronosyltransferase (UGT), play a vital role in the hydrolysis of baicalin to baicalein (Fong et al. 2012; Kalapos-Kovács et al. 2015; Noh et al. 2016). It is interesting to note that baicalin mainly exist in its glycosidic form in systemic circulation (Li et al. 2014). The studies have observed bimodal and multiple peaks in the absorption profile of baicalin (Zhao et al. 2014; Zhang et al. 2016). These peaks probably arise due to variable gastric emptying time, glucuronidation, and absorption at different sites (Lu et al. 2007; Tong et al. 2012; Song et al. 2017). In addition, after combining with human plasma protein, baicalin was reported to bind with serum albumin quickly and was distributed to various tissues mediated through ABC transporter inhibitors in membranes overexpressing the ATP-dependent MDR1 efflux pump in Caco-2 cell monolayer model (Zhang et al. 2007). Additionally, the concentration of baicalin is different in various visceral organs depending on the formulation and route of administration (Zhu et al. 2013; Wei et al. 2016). The metabolites of baicalin have been observed in various tissues (heart, liver, spleen, lung, kidney, and brain), and the main organs for its metabolism were reported to be the liver and kidney (Zhang et al. 2015). The predominant enzymes involved in metabolism of baicalin are UGT, catechol-O-methyltransferases, β-glucuronidase, and sulfatase and thus causes methoxylation, methylation, hydroxylation, sulfation, glucuronidation, hydrolysis, etc. (Akao et al. 2013; Lu et al. 2012; Wang et al. 2012; Zhang et al. 2015). Baicalin was reported to be predominantly excreted in bile which is mediated by MRP2 (Xing et al. 2005; Akao et al. 2009; Kalapos-Kovács et al. 2015). It was reported that only 7.2% of the baicalin was primarily excreted in urine in the form of sulfated and hydroxylated derivatives and parent drug (Jiang et al. 2014; Lai et al. 2003).
Toxicity
A recent study has reported that baicalin (1) and baicalein inhibit the main protease (3CLpro) of SARS-CoV-2 (Su et al. 2020). Very few studies have been conducted to evaluate the toxicological profile of baicalin and baicalein. Pang et al. (2016) conducted a single-center, double-blind, placebo-controlled, parallel-group clinical study. The dose proportionality constant for AUCss of baicalein and baicalin was found to be 0.922 (90% confidence interval, 0.650–1.195) and 0.942 (90% confidence interval, 0.539–1.345), respectively, and the accumulation index was found to be 1.66–2.07 and 1.68–2.45, respectively. From the results, it was concluded that that a dose of 200–800 mg of baicalein in multiple oral doses is safe and well tolerated. Aqueous extracts of S. baicalensis were reported to beneficial in an antigen-induced ear swelling in rats and decrease the β-hexosaminidase release in rat basophilic leukemia cells (RBL-2H3) without causing dermal irritation/corrosion (Kim et al. 2013). In a 90-day oral toxicity study of UP446 (a combination of baicalin from the roots of Scutellaria baicalensis and (+)-catechin from the heartwoods of Acacia catechu), the ‘No Observed Adverse Effect Level (NOAEL)’ was found to be 1000 mg/kg/day (Yimam et al. 2010). Another study has confirmed that a single-oral dose of 100–2800 mg of baicalein is safe and well tolerated in healthy volunteers (clinical trial registration number: CTR20132944) (Li et al. 2014). However, a recent study has demonstrated that baicalin can activate the transforming growth factor-β (TGF-β)/Smad signaling pathway in a dose-dependent manner, induce the Smad3 acetylation (via interaction of Smad3 with transcriptional coactivator p300), and reduce the phosphorylation of AMPK (a metabolic master kinase) that results in kidney injury and fibrosis at higher doses (Cai et al. 2017). Han et al. (2011) evaluated in vitro and in vivo the pulmonary toxicity of shuang-huang-lian preparation, with a long history for treating respiratory tract infection. A report indicated that the injection of the lyophilized powder of shuang-huang-lian prepared with honeysuckle (Lonicera caprifolium L., Caprifoliaceae), S. baicalensis roots, and the fruits of Forsythia suspensa (Thunb.) Vahl, Oleaceae (Dong et al. 2017), is commonly used to treat bacterial and viral infections in the respiratory tract (Zhang et al. 2013). The in vitro results showed no adverse effects at doses of up to 6 mg/kg and 25 mg/kg for histopathologic study and cell counts experiment, respectively. In vivo results showed no difference in LDH activity, total protein content, nor IL-6, IL-8, and TNF-α levels in bronchoalveolar lavage fluid (BALF). However, inhalation of shuang-huang-lian preparation at higher doses causes lung alveolar fusion, and increased number of monocytes and granulocytes were observed (Han et al. 2011).
Antiviral Activity
Signaling Pathway
IFN and JAK-STAT
Interferons (IFNs) are a group of signaling proteins of cytokine which are secreted by the human body after virus infection and are named for their ability to “interfere” with viral replication by the regulation of the immune system (Darnell et al. 1994). In pulmonary micro-vascular endothelial cells (PMVEC), the secretion of IFN-α and IFN-γ is significantly increased to reduce the degree of immunological injury (Lin et al. 2009). Therefore, the effect of baicalin (1) on IFN expression has been explored as an important molecular mechanism for the antiviral effect of this flavonoid. The JAK-STAT signaling pathway is involved in processes such as immunity, cell division, cell death, and tumor formation. There are three key parts of JAK-STAT signaling: Janus kinases (JAKs), signal transducer and activator of transcription proteins (STATs), and receptors (which bind the chemical signals). It has been reported that baicalin exhibited antiviral effect as demonstrated by the use of the respiratory syncytial virus infection model via the following mechanism: (1) the increasing expression of IFNs in rats (Zhan et al. 2017), (2) activation of JAK-1 and non-receptor tyrosine-protein kinase (TYK-2) to promote phosphorylation of STAT1 and STAT2, and (3) inhibiting expression of SOCS1 and SOCS3 proteins to activate JAK/STAT pathway (Zhang et al. 2018).
Numerous studies claimed that baicalin (1) could help to induce IFN-γ secreted by CD4+, CD8+ t-lymphocytes, and NK cell to effectively enhance activity of JAK/STAT1 pathway (Chu et al. 2015; Zhi et al. 2019). The main chemical components of the shuanghuanglian injection are chlorogenic acid, baicalin, and forsythia glycosides, which have been found to have the ability of anti-inflammation, improving immunity, and inhibiting the growth of various viruses. Baicalin, as one of the bioactive ingredients, is capable of increasing expression of IFN-γ and effectively inhibiting cell apoptosis in a dose-dependent and time-dependent manner with infection of human adenovirus III (HAdV3) in A549 and Hep-G2 cells (Ma et al. 2017). Oral administration of S. baicalensis root (80% ethanol) effectively protected mice infected with influenza A virus (IVA), increased the survival rate (herbal preparation: 67%; baicalin: 33%), decreased the lung injury index (herbal preparation: 0.90; 1: 1.00), and improved the lung morphology (Fig. 1). This preparation efficiently decreased lung virus titers, reduced hemagglutinin titers, inhibited neuraminidase activities in the lungs of IAV-infected mice, and modulated the inflammatory responses by decreasing the levels of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and monocyte chemotactic protein-1 (MCP-1) and increasing the levels of interferon-γ (IFN-γ) and interleukin-10 (IL-10) in lung tissues (Zhi et al. 2019).

Reproduced from Zhi et al. 2019
Hematoxylin–eosin stained (400 ×) of lung tissues (A) and immunohistochemical expression of C3d in lungs (magnification of alveolus) at day 4 after infection (B). Abbreviations: Model, influenza A virus-infected lung tissues; Ext, extract of Scutellaria baicalensis root in 80% ethanol; 1, baicalin; RB, ribavirin (positive control).
TLRs and NF-κB
NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) is a protein complex that controls transcription of DNA, cytokine production, and cell survival. NF-κB is found in almost all animal cell types and is involved in cellular responses to stimuli such as stress, cytokines, free radicals, heavy metals, ultraviolet irradiation, oxidized LDL, and bacterial or viral antigens (Fig. 2). NF-κB induced immunological injury through regulating the expression of TAB2, NFKB, IRAK1 and RIPK1 (Liu et al. 2016). A report suggested that baicalin (1) inhibit immunological injury through the regulation of TNF-α, IL-1β, IL-6, IL-8, IL-12, IL-18, and CCL5 via specifically inhibiting TLR7/MyD88 pathway. In this pathway, the expression of TLR3, TLR4, and TRIF mRNA were suppressed; the levels of IRAK4 and FOS were reduced; NF-κB and AP-1 protein were activated; and the phosphorylation of NF-κB-65 protein was decreased (Wan et al. 2014). These observed changes suggest that the primary mechanism of baicalin (1) in antiviral in inhibiting immunologic injury involves inhibiting NF-κB and TLR7/MyD88 pathway, followed by reducing the inflammatory cytokine expression.
Mechanism of NF-κB action after activation by a viral infection. The NF-κB heterodimer consisting of Rel and p50 proteins is used as an example. While in an inactivated state, NF-κB is located in the cytosol complexed with the inhibitory protein IκBα. Through the intermediacy of integral membrane receptors, a variety of extracellular signals, as viral agents, can activate the enzyme IκB kinase (IKK). IKK, in turn, phosphorylates the IκBα protein, which results in ubiquitination, dissociation of IκBα from NF-κB, and eventual degradation of IκBα by the proteasome. The activated NF-κB is then translocated into the nucleus where it binds to specific sequences of DNA called response elements (RE). The DNA/NF-κB complex then recruits other proteins such as coactivators and RNA polymerase, which transcribe downstream DNA into mRNA. In turn, mRNA is translated into protein, resulting in a change of cell function
HNFs
HNFs plays a vital role in the proliferation of hepatitis B virus (HBV) and the underlying mechanism involved in up-regulating HNF1α expression, increasing HNF4α expression, and promoting pgRNA biosynthesis and transcription (Huang. 2013). In Hep-G2 cells, baicalin (1) substantially decreased HBsAg and HBeAg expression via reducing the level of HNF1α and HNF4α to inhibit the RNA of HBV proliferation and synthesis (Cheng et al. 2006; Huang et al. 2017b). In addition, a study in exploring the mechanisms involved in the treatment of HBV has been reported for baicalin to reduce proliferation through increasing the physical interaction of HNF4α and ERα/β and decreasing the expression of HNF1α to inhibit the bond between HNFs and cccDNA (Chirumbolo 2018; Xia et al. 2020).
Interferon Regulatory Factors
Influenza A virus (IAV) causes immunologic injury via mediating inflammatory cytokines. The underlying mechanisms probably involved the regulation of interferon factors (IRFs, such as IRF3 and IRF7), NF-κB activation via promoting retinoic acid-induced protein I (RIG-I) and MAVS protein interaction, and increased IFN-α/β and TNF-α secretion. A recent study has reported that baicalin (1) decreases the expression of fundamental factors (IRF3, IRF7, and NF-κB) in RLRs pathway. So, it is suggested that baicalin can suppress innate immune hyperactivity caused by IAV infection (Pang et al. 2018). In ANA-1 cell, the level of macrophage is significantly increased after H1N1 infection, which probably is one of the most important mechanisms of promoting inflammatory reaction occurrence. Thus, decreasing the secretion of macrophage is an effective method to treat inflammatory reaction. A recent study has proven that baicalin increased M1 macrophage ratio, stimulates IFN-β and IRF3 expression, and reduces IL-10, thus inhibiting inflammatory reaction (Geng et al. 2020). Moreover, the chick embryo fibroblast (CEF) cell is reported to overexpress TRF7 after Marek’s disease virus (MDV) infection. Yang et al. (2020) reported that baicalin decreased the IRF7 level in CEF cell, thus exhibiting antiviral effects.
Interleukin (IL) and TNF-α
When the virus invades the host body, a series of physiological reactions occur in the host body, including the activation of the immune response and the secretion of a variety of inflammatory factors. IL-6, a common inflammatory cytokine, plays a vital role in increasing the expression of STAT3 protein and SOCS-1 gene and inhibiting the differentiation of Th1 cell, thus down-regulating the JAK/STAT pathway (Zhang et al. 2018). Zhang et al. (2020) reported that baicalin (1) has a shown a significant effect in relieving oxidative stress by reducing the level of IL-17A, IL-22, IL-6, and IL-1β in chicks infected with H9N2. Treatment of baicalin lowered the TNF-α and IL-2 level and reduced the degree of inflammatory cell infiltration in RSV-infected rat (Cheng et al. 2014). Moreover, inflammatory injury is caused by human adenovirus III in A549 and Hep-G2 cell. After being treated by shuanghuanglian injection lyophilized powder, the symptom was significantly reduced. The underlying mechanisms were reported to the inhibition of plaque formation and decreasing the levels of TNF-α, IL-1β, and IL-6 (Ma et al. 2017).
Apoptosis-Related Genes
The third phase of cell apoptosis, center control and effective phase, is the phase of cysteine aspirate protease activated by apoptosis signal to performed programmed cell death. The apoptosis pathways include caspase-8-mediated extrinsic pathway and the intrinsic pathway (caspase-9-mediated mitochondria apoptosis pathway and endoplasmic reticulum pathway) (Long et al. 2004; Nomura et al. 2005). In recent years, studies reported that baicalin (1) exhibits the inhibition of cell apoptosis via (1) decreased expression of membrane receptor Fas; (2) reduced level of caspases; (3) increased expression of Bcl-xl, FLIP, and IAP; (4) inhibition of protein phosphatase 3 that catalyzes the subunit alpha-subtype gene (Cn); and (5) inhibition of apoptosis signal transduction. Therefore, the result suggested that baicalin exhibited inhibition of cell apoptosis through regulating the relevant protein of caspase-8 expression mediated by extrinsic pathway and endoplasmic reticulum pathway. In addition, in the cell infected with EV71 and CHIKV, the expression of pro-apoptotic genes, Fas and caspase-3, is significantly decreased to inhibit cellular apoptosis. Therefore, the effect of baicalin (1) on cellular apoptosis has been explored as a vital underlying mechanism for the antiviral effect of this flavonoid (Li et al. 2015b; Oo et al. 2018).
Immune Cells
The activity of the T cell subset is an index to determine the cellular immune function of patients. Recent studies have indicated that baicalin (1) can improve patient immune function. In infected patients with HBV and RSV, baicalin increased CD4+, CD8+ levels, CD4+/CD8+ ratio, and the activity of NK cells (Shi et al. 2016). The IAV infection was reported to increase elevated level of pro-inflammatory response factors (Th1 and Th17). Baicalin was reported to inhibit the inflammatory reaction via increased up-regulation of the Th2 and Treg expression and decreased Th1/Th2 and Th17/Treg ratios. The results revealed that baicalin has blocked the Th1 and Th17 type inflammatory reaction through regulating the differentiation of CD4+ (Pang et al. 2018). Baicalin was reported to inhibit H1N1 infection via stimulation of CD4+, CD8+, and NK cells, increased expression of IFN-γ, and activation of JAK/STAT1 pathway in human peripheral blood monocyte cells (HPBMC) (Chu et al. 2015).
Others
In Hela cell, Coxsackie Virus B3-induced cell autophagy is mediated via (1) increasing the content of free fatty acid (FFA) and cholesterol (CHO), (2) up-regulating the expression of fatty acid synthase (Fasn) and acetyl-Coa carboxylase mRNA, and (3) stimulating the generation of cytolysosome. The treatment of baicalin (1) reduced the level of FFA, CHO, Fasn acetyl-CoA carboxylase, and LC3-II (an autophagosome marker). Therefore, the result has concluded that the observed antiviral effect of baicalin on CVB3 might be mediated via suppressing lipid synthesis and inhibiting autophagosome generation (Wang et al. 2020). The mechanisms implicated in the inhibitory effects of baicalin on viral pneumonia were also reported in the literature. In FM1-infected mouse model, baicalin was reported to down-regulate the MDA and total NO synthase levels in the lung tissues, increase the glutathione peroxidase (GSH-PX) activity, reduce endothelial cell dysfunction, reduce the vascular overexpansion, and reduce the tissue injury (Wan et al. 2011). Finally, the oral liquid shuang-huang-lian formulation showed a dose-dependently inhibited SARS-CoV-2 3CLpro as well as the replication of SARS-CoV-2 in Vero E6 cells. Baicalin and baicalein were characterized as the first noncovalent, nonpeptidomimetic inhibitors of SARS-CoV-2 3CLpro and exhibited potent antiviral activities in a cell-based system (Su et al. 2020).
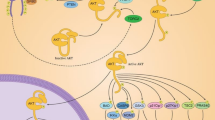
To sum up, the most significant antiviral molecular mechanisms of baicalin (1) are the increased expression of IFNs; activation of JAK/STAT pathway; suppression of TLRs activity; suppression of NF-κB pathway; reduction of HNFs, IRFs, TNF-α, and IL levels; inhibition of cysteine aspirate protease; inhibition of pro-apoptotic genes; and improvement of immune function. The antiviral molecular mechanisms of baicalin are summarized in Table S1, and the molecular pathways for the antiviral properties of baicalin are illustrated in Fig. 3.
Clinical Studies
With the rising interest in the antiviral effect of baicalin (1), numerous studies have been carried out focusing on its clinical applications. Baicalin could be combined with other medicines to treat viral diseases such as chronic hepatitis B and A H1N1 influenza. The possible underlying molecular mechanisms involved in its beneficial action are improving the immunity in patients via regulation of the T-lymphocyte subset levels and up-regulation of CD3+, CD4+, and CD4+/CD8+ levels in patients infected with A H1N1 influenza (Su and Xiang 2014). In addition, a study has reported that baicalin reduces the level of alanine aminotransferase (ALT), aspartate transaminase (AST), and total bilirubin (TBil), and HBV-DNA were reduced and reduced the hepatic fibrosis in chronic hepatitis B patients (Huang et al. 2017a). The details about drugs and studies are summarized in Table S2.
However, the reported findings in the literature are obtained from retrospective studies, and thus there might be a component of bias in reporting the efficacy. The bias could arise from the subjectivity of the clinician, experimental methods, sample size, and the classification of disease, which are parameters difficult to control in clinical studies (Li et al. 2020, 2021).
Perspective on Future Directions
Some clinical trials have suggested that the potent inhibitory activity of baicalin (1) on influenza virus make it worthwhile to be subjected to further clinical trials, particularly for COVID-19 treatment (Su et al. 2020). Low aqueous solubility and low bioavailability of baicalin (1) are the major challenges for its use in clinic. Encouraging results in terms of improving the bioavailability are obtained from cyclodextrin complexes, self-nanoemulsifying systems, micelle, nanocrystals, and gelling system, inter alia. However, development of an ideal formulation is still far away. In the literature, very limited studies were reported on the safety and efficacy of baicalin. Also, a few clinical studies have reported the beneficial effect of baicalin in improving the antiviral activity of entecavir, lamivudine, and oseltamivir, which has to be further evaluated for the treatment of viral infections in the respiratory tract and combat COVID-19. There were only very few clinical studies in which the antiviral activity of baicalin was investigated. Therefore, the future research studies must be focused on evaluating the clinical efficacy of baicalin in treating viral infections. Therefore, more rigorous scientific studies should be carried out on baicalin and its plant sources.
Conclusion
In this review, we have discussed the latest research progress on baicalin (1) regarding its physicochemical properties, toxicity, bioavailability, pharmacokinetics, antiviral activities, and clinical studies. In recent years, the number of studies on baicalin was increased, and most of the studies have been concentrated on the direction of the clinical studies for its effect as an antiviral activity. Additionally, despite the availability of many reported formulations on baicalin to improve its bioavailability, the ideal formulation is yet to be identified. Only a few clinical trials with small number of volunteers have been conducted to evaluate the safety of baicalin, and thus utmost care for the administration of baicalin should be paid. In this review, we also summarized the antiviral molecular mechanisms of baicalin. In recent years, the antiviral effect of baicalin against IVA, RSV, HV, CVB3, etc. has been widely investigated. The main molecular mechanisms reported were regulation of JAK/STAT, TLRs, and NF-κB pathways. Baicalin inhibits inflammations via regulating the IFN, IL, SOCS1/3, PKR protein, Mx1 protein, and AP-1 protein levels. The antiviral activity of baicalin was also reported to be exerted through inhibition of apoptosis, decreased expression of FAS protein, inhibition of caspase-8 pathway, and decreased expression of Bax protein. These information present in this review will be helpful in guiding future research studies. Further studies must be carried out to explore the possibility of involvement of additional mechanisms.
References
Akao T, Sato K, Hanada M (2009) Hepatic contribution to a marked increase in the plasma concentration of baicalin after oral administration of its aglycone, baicalein, in multidrug resistance-associated protein 2-deficient rat. Biol Pharm Bull 32:2079–2082. https://doi.org/10.1248/bpb.32.2079
Akao T, Sato K, He JX, Ma CM, Hattori M (2013) Baicalein 6-O-β-D-glucopyranuronoside is a main metabolite in the plasma after oral administration of baicalin, a flavone glucuronide of scutellariae radix, to rats. Biol Pharm Bull 36:748–753. https://doi.org/10.1248/bpb.b12-00850
Cai Y, Ma W, Xiao Y, Wu B, Li X, Liu F, Qiu J, Zhang G (2017) High doses of baicalin induces kidney injury and fibrosis through regulating TGF-β/Smad signaling pathway. Toxicol Appl Pharmacol 333:1–9. https://doi.org/10.1016/j.taap.2017.08.003
Chen Y, Yuan WJ, Yang YH, Yao FK, Ming K, Liu JG (2018) Inhibition mechanisms of baicalin and its phospholipid complex against DHAV-1 replication. Poult Sci 97:3816–3825. https://doi.org/10.3382/ps/pey255
Chen Y, Yao FK, Ming K, Shi JT, Zeng L, Wang DY, Wu Y, Hu YL, Liu JG (2019) Assessment of the effect of baicalin on duck virus hepatitis. Curr Mol Med 19:376–386. https://doi.org/10.2174/1566524019666190405095301
Chen KH, Wang SF, Wang SY, Yang YP, Wang ML, Chiou SH, Chang YL (2020) Pharmacological development of the potential adjuvant therapeutic agents against coronavirus disease 2019. J Chin Med Assoc 83:817–821. https://doi.org/10.1097/JCMA.0000000000000375
Cheng Y, Ping J, Xu HD, Fu HJ, Zhou ZH (2006) Synergistic effect of a novel oxymatrine-baicalin combination against hepatitis B virus replication, alpha smooth muscle actin expression and type I collagen synthesis in vitro. World J Gastroenterol 12:5153–5159. https://doi.org/10.3748/wjg.v12.i32.5153
Cheng KB, Wu ZH, Gao BL, Xu JF (2014) Analysis of influence of baicalin joint resveratrol retention enema on the TNF-α, SIgA, IL-2, IFN-γ of rats with respiratory syncytial virus infection. Cell Biochem Biophys 70:1305–1309. https://doi.org/10.1007/s12013-014-0055-9
Chinsembu KC (2020) Coronaviruses and nature’s pharmacy for the relief of coronavirus disease 2019. Rev Bras Farmacogn 30:603–621. https://doi.org/10.1007/s43450-020-00104-7
Chirumbolo S (2018) Baicalin in flavocoxid may act against hepatitis B virus via a pro-inflammatory pathway. Inflamm Res 67:203–205. https://doi.org/10.1007/s00011-017-1111-x
Chu M, Xu L, Zhang MB, Chu ZY, Wang YD (2015) Role of baicalin in anti-influenza virus A as a potent inducer of IFN-Gamma. Biomed Res Int 2015:263630. https://doi.org/10.1155/2015/263630
Darnell JE Jr, Kerr IM, Stark GR (1994) Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 264:1415–1421. https://doi.org/10.1126/science.8197455
Dinda B, Dinda S, DasSharma S, Banik R, Chakraborty A, Dinda M (2017) Therapeutic potentials of baicalin and its aglycone, baicalein against inflammatory disorders. Eur J Med Chem 131:68–80. https://doi.org/10.1016/j.ejmech.2017.03.004
Diniz LRL, Souza MTS, Duarte ABS, Sousa DP (2020) Mechanistic aspects and therapeutic potential of quercetin against COVID-19-associated acute kidney injury. Molecules 25:5772. https://doi.org/10.3390/molecules25235772
Dong Z, Lu X, Tong X, Dong Y, Tang L, Liu M (2017) Forsythiae fructus: a review on its phytochemistry, quality control, pharmacology and pharmacokinetics. Molecules 22:1466. https://doi.org/10.3390/molecules22091466
El-Gogary Gaber Nasr RISAAM (2019) Polymeric nanocapsular baicalin: chemometric optimization, physicochemical characterization and mechanistic anticancer approaches on breast cancer cell lines. Sci Rep 9:11064. https://doi.org/10.1038/s41598-019-47586-7
Fang P, Yu M, Shi M, Bo P, Gu X, Zhang Z (2020) Baicalin and its aglycone: a novel approach for treatment of metabolic disorders. Pharmacol Rep 72:13–23. https://doi.org/10.1007/s43440-019-00024-x
Feng J, Xu W, Tao X, Wei H, Cai F, Jiang B, Chen W (2010) Simultaneous determination of baicalin, baicalein, wogonin, berberine, palmatine and jatrorrhizine in rat plasma by liquid chromatography-tandem mass spectrometry and application in pharmacokinetic studies after oral administration of traditional Chinese medicinal preparations containing scutellaria-coptis herb couple. J Pharm Biomed Anal 53:591–598. https://doi.org/10.1016/j.jpba.2010.04.002
Fong YK, Li CR, Wo SK, Wang S, Zhou L, Zhang L, Lin G, Zuo Z (2012) In vitro and in situ evaluation of herb-drug interactions during intestinal metabolism and absorption of baicalein. J Ethnopharmacol 141:742–753. https://doi.org/10.1016/j.jep.2011.08.042
Geng P, Zhu HY, Zhou W, Su C, Chen MC, Huang CG, Xia CJ, Huang H, Cao YO, Shi XL (2020) Baicalin inhibits influenza A virus infection via promotion of M1 macrophage polarization. Front Pharmacol 11:01298. https://doi.org/10.3389/fphar.2020.01298
Guo XS, Wei N, Wang YX (2016) Observation on the effect of baicalin combined with antiviral drugs on severe influenza. Women’s Health Res 18:186–193
Han R, Ye JX, Quan LH, Liu CY, Yang M, Liao YH (2011) Evaluating pulmonary toxicity of Shuang-Huang-Lian in vitro and in vivo. J Ethnopharmacol 135:522–529. https://doi.org/10.1016/j.jep.2011.03.060
Huang H (2013) Study on the anti-influenza virus mechanism of GM-CSF and anti-HBV effects of entecavir-baicalin combination. Fudan University. https://t.cnki.net/kcms/detail?v=3uoqIhG8C447WN1SO36whLpCgh0R0Z-iv9r0YoQXiId4v9BfOE9rDiZFolQwPaJdluhFmc4M6iWyCgUbDFMTXo9y4RhIVobL
Huang XZ, Ke WB, Zhan GX, Sai J, Ji D (2017a) Clinical study on sugan capsules combined with baicalin capsules in treatment of chronic hepatitis B. Drugs & Clinic 32:271–274
Huang H, Zhou W, Zhu HY, Zhou P, Shi XL (2017b) Baicalin benefits the anti-HBV therapy via inhibiting HBV viral RNAs. Toxicol Appl Pharmacol 323:36–43. https://doi.org/10.1016/j.taap.2017.03.016
Huang T, Liu Y, Zhang C (2019) Pharmacokinetics and bioavailability enhancement of baicalin: a review. Eur J Drug Metab Pharmacokinet 44:159–168. https://doi.org/10.1007/s13318-018-0509-3
Jakab G, Bogdán D, Mazák K, Deme R, Mucsi Z, Mándity IM, Noszál B, Kállai-Szabó N, Antal I (2019) Physicochemical profiling of baicalin along with the development and characterization of cyclodextrin inclusion complexes. AAPS PharmSciTech 20:314. https://doi.org/10.1208/s12249-019-1525-6
Ji S, Li R, Wang Q, Miao WJ, Li ZW, Si LL, Qiao X, Yu SW, Zhou DM, Ye M (2015) Anti-H1N1 virus, cytotoxic and Nrf2 activation activities of chemical constituents from Scutellaria baicalensis. J Ethnopharmacol 176:475–484. https://doi.org/10.1016/j.jep.2015.11.018
Jia Y, Xu RG, Hu YC, Zhu TL, Ma TT, Wu HH, Hu LW (2016) Anti-NDV activity of baicalin from a traditional Chinese medicine in vitro. J Vet Med Sci 78:819–824. https://doi.org/10.1292/jvms.15-0572
Jiang S, Xu J, Qian DW, Shang EX, Liu P, Su SL, Leng XJ, Guo JM, Duan JA, Du L, Zhao M (2014) Comparative metabolites in plasma and urine of normal and type 2 diabetic rats after oral administration of the traditional Chinese scutellaria-coptis herb couple by ultra-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 965:27–32. https://doi.org/10.1016/j.jchromb.2014.05.028
Jin SY, Han J, Jin SX, Lv QY, Bai JX, Chen HG, Li RS, Wu W, Yuan HL (2014) Characterization and evaluation in vivo of baicalin-nanocrystals prepared by an ultrasonic-homogenization-fluid bed drying method. Chin J Nat Med 12:71–80. https://doi.org/10.1016/S1875-5364(14)60012-1
Jo S, Kim S, Kim DY, Kim MS, Shin DH (2020) Flavonoids with inhibitory activity against SARS-CoV-2 3CLpro. J Enzyme Inhib Med Chem 35:1539–1544. https://doi.org/10.1080/14756366.2020.1801672
Kalapos-Kovács B, Magda B, Jani M, Fekete Z, Szabó PT, Antal I, Krajcsi P, Klebovich I (2015) Multiple ABC transporters efflux baicalin. Phytother Res 29:1987–1990. https://doi.org/10.1002/ptr.5477
Kim YH, Jeong DW, Paek IB, Ji HY, Kim YC, Sohn DH, Lee HS (2006) Liquid chromatography with tandem mass spectrometry for the simultaneous determination of baicalein, baicalin, oroxylin A and wogonin in rat plasma. J Chromatogr B Analyt Technol Biomed Life Sci 844:261–267. https://doi.org/10.1016/j.jchromb.2006.07.021
Kim TW, Song IB, Lee HK, Kim MS, Ham SH, Cho JH, Lim JH, Yun HI (2013) Assessment of dermal safety of Scutellaria baicalensis aqueous extract topical application on skin hypersensitivity. Planta Med 79:959–962. https://doi.org/10.1055/s-0032-1328714
Komolafe K, Komolafe TR, Fatoki TH, Akinmoladun AC, Brai BIC, Olaleye MT, Akindahunsi AA (2021) Coronavirus disease 2019 and herbal therapy: pertinent issues relating to toxicity and standardization of phytopharmaceuticals. Rev Bras Farmacogn 31:142–161. https://doi.org/10.1007/s43450-021-00132-x
Kong D, Liang N, Liu J, Nikolova D, Jakobsen J, Gluud C (2018) Xiao Chai Hu Tang, a Chinese herbal medicine formula, for chronic hepatitis B. Cochrane Database Syst Rev 2018:8. https://doi.org/10.1002/14651858.CD013090.pub2
Kotani A, Kojima S, Hakamata H, Kusu F (2006) HPLC with electrochemical detection to examine the pharmacokinetics of baicalin and baicalein in rat plasma after oral administration of a Kampo medicine. Anal Biochem 350:99–104. https://doi.org/10.1016/j.ab.2005.11.007
Lai MY, Hsiu SL, Chen CC, Hou YC, Chao PD (2003) Urinary pharmacokinetics of baicalein, wogonin and their glycosides after oral administration of Scutellariae Radix in humans. Biol Pharm Bull 26:79–83. https://doi.org/10.1248/bpb.26.79
Li R, Wang LX (2019) Baicalin inhibits influenza virus A replication via activation of type I IFN signaling by reducing miR-146a. Mol Med Rep 20:5041–5049. https://doi.org/10.3892/mmr.2019.10743
Li BQ, Fu T, Dongyan Y, Mikovits JA, Ruscetti FW, Wang JM (2000) Flavonoid baicalin inhibits HIV-1 infection at the level of viral entry. Biochem Biophys Res Commun 276:534–538. https://doi.org/10.1006/bbrc.2000.3485
Li C, Zhang L, Lin G, Zuo Z (2011) Identification and quantification of baicalein, wogonin, oroxylin A and their major glucuronide conjugated metabolites in rat plasma after oral administration of Radix scutellariae product. J Pharm Biomed Anal 54:750–758. https://doi.org/10.1016/j.jpba.2010.10.005
Li M, Shi A, Pang H, Xue W, Li Y, Cao G, Yan B, Dong F, Li K, Xiao W, He G, Du G, Hu X (2014) Safety, tolerability, and pharmacokinetics of a single ascending dose of baicalein chewable tablets in healthy subjects. J Ethnopharmacol 156:210–215. https://doi.org/10.1016/j.jep.2014.08.031
Li XH, Huang SD, Yang J (2015a) Effect of baicalin combined with lamivudine in the treatment of chronic hepatitis B on cellular immunity. Pharm Clin Chin Materi Medica 31:218–220
Li X, Liu YY, Wu TT, Jin Y, Cheng JP, Wan CB, Qian WH, Xing F, Shi WF (2015b) The antiviral effect of baicalin on enterovirus 71 in vitro. Viruses 7:4756–4771. https://doi.org/10.3390/v7082841
Li Y, He ZD, Zheng QE, Hu C, Lai WF (2018) Hydroxypropyl-β-cyclodextrin for delivery of baicalin via inclusion complexation by supercritical fluid encapsulation. Molecules 23:1169. https://doi.org/10.3390/molecules23051169
Li KW, Liang YY, Xie SM, Niu FJ, Guo LY, Liu ZH, Zhou CZ, Wang LZ (2020) Ixeris sonchifolia: a review of its traditional uses, chemical constituents, pharmacology and modern applications. Biomed Pharmacother 125:109869. https://doi.org/10.1016/j.biopha.2020.109869
Li KW, Liang YY, Wang Q, Li Y, Zhou SJ, Wei HC, Zhou CZ, Wan XH (2021) Brucea javanica: a review on anticancer of its pharmacological properties and clinical researches. Phytomedicine 86:153560. https://doi.org/10.1016/j.phymed.2021.153560
Liang R, Han RM, Fu LM, Ai XC, Zhang JP, Skibsted LH (2009) Baicalin in radical scavenging and its synergistic effect with beta-carotene in antilipoxidation. J Agric Food Chem 57:7118–7124. https://doi.org/10.1021/jf9013263
Liang S, Deng X, Lei L, Zheng Y, Ai J, Chen L, Xiong H, Mei Z, Cheng YC, Ren Y (2019) The comparative study of the therapeutic effects and mechanism of baicalin, baicalein, and their combination on ulcerative colitis rat. Front Pharmacol 10:1466. https://doi.org/10.3389/fphar.2019.01466
Lin H, Hu G, Yi PF, Jiang XL, Mu X (2009) Effect of baicalin and forsythoside on the excretion of IFN-α and IFN-γ in pulmonary microvascular endothelial cells. China Animal Hus Vet Med 36:30–33
Liu TM, Jiang XH (2006) Investigation of the absorption mechanisms of baicalin and baicalein in rats. J Pharm Sci 95:1326–1333. https://doi.org/10.1002/jps.20593
Liu XT, Gu LG, Deng DY, Yu ZN, Wang YQ, Wu J, Qiu ZJ (2016) Effects of baicalin and luteolin-7-O-glucoside on NF-κB signaling pathway in A549 cells infected with influenza virus H1N1 in vitro. China J Tradit Chin Med Pharm 31:1937–1941
Long S, Wilson M, Bengtén R, Clem LW, Miller NW, Chinchar VG (2004) Identification and characterization of a FasL-like protein and cDNAs encoding the channel catfish death-inducing signaling complex. Immunogenetics 56:518–530. https://doi.org/10.1007/s00251-004-0701-2
Long Y, Xiang Y, Tan YJ, Li N, Yang QY (2019) Research progress on pharmacological action and new dosage forms of baicalin. Chin Tradit Herbal Drugs 50:6142–6148
Lou G, Xiong H, Gan Q, Hu J, Peng C, Yan Z, Yan H, Huang Q (2021) UPLC-Q-Orbitrap HRMS analysis of Coptis chinensis aerial parts and its regulatory activity on glucose-lipid metabolism. Rev Bras Farmacogn 31:24–31. https://doi.org/10.1007/s43450-020-00124-3
Lu T, Song J, Huang F, Deng Y, Xie L, Wang G, Liu X (2007) Comparative pharmacokinetics of baicalin after oral administration of pure baicalin, Radix scutellariae extract and Huang-Lian-Jie-Du-Tang to rats. J Ethnopharmacol 110:412–418. https://doi.org/10.1016/j.jep.2006.09.036
Lu QY, Zhang L, Moro A, Chen MC, Harris DM, Eibl G, Go VL (2012) Detection of baicalin metabolites baicalein and oroxylin-a in mouse pancreas and pancreatic xenografts. Pancreas 41:571–576. https://doi.org/10.1097/MPA.0b013e318232e130
Ma Q, Liang D, Song S, Yu Q, Shi C, Xing X, Luo JB (2017) Comparative study on the antivirus activity of shuang-huang-lian injectable powder and its bioactive compound mixture against human adenovirus III in vitro. Viruses 9:79. https://doi.org/10.3390/v9040079
Martínez Medina JJ, Naso LG, Pérez AL, Rizzi A, Ferrer EG, Williams PAM (2017) Antioxidant and anticancer effects and bioavailability studies of the flavonoid baicalin and its oxidovanadium(IV) complex. J Inorg Biochem 166:150–161. https://doi.org/10.1016/j.jinorgbio.2016.11.005
Nayak MK, Agrawal AS, Bose S, Naskar S, Bhowmick R, Chakrabarti S, Sarkar S, Chawla-Sarkar M (2014) Antiviral activity of baicalin against influenza virus H1N1-pdm09 is due to modulation of NS1-mediated cellular innate immune responses. J Antimicrob Chemother 69:1298–1310. https://doi.org/10.1093/jac/dkt534
Noh K, Kang Y, Nepal MR, Jeong KS, Oh DG, Kang MJ, Lee S, Kang W, Jeong HG, Jeong TC (2016) Role of intestinal microbiota in baicalin-induced drug interaction and its pharmacokinetics. Molecules 21:337. https://doi.org/10.3390/molecules2103033
Nomura J, Matsumoto K, Iguchi-Ariga SM, Ariga H (2005) Mitochondria-independent induction of Fas-mediated apoptosis by MSSP. Oncol Rep 14:1305–1309
Oo A, Rausalu K, Merits A, Higgs S, Vanlandingham D, Bakar SA, Zandi K (2018) Deciphering the potential of baicalin as an antiviral agent for Chikungunya virus infection. Antiviral Res 150:101–111. https://doi.org/10.1016/j.antiviral.2017.12.012
Pang H, Xue W, Shi A, Li M, Li Y, Cao G, Yan B, Dong F, Xiao W, He G, Du G, Hu X, Cheng G (2016) Multiple-ascending-dose pharmacokinetics and safety evaluation of baicalein chewable tablets in healthy Chinese volunteers. Clin Drug Investig 36:713–724. https://doi.org/10.1007/s40261-016-0418-7
Pang P, Zheng K, Wu SZ, Xu HC, Deng L, Shi YC, Chen XY (2018) Baicalin downregulates RLRs signaling pathway to control influenza A virus infection and improve the prognosis. Evid Based Complement Alternat Med 2018:4923062. https://doi.org/10.1155/2018/4923062
Pollicino T, Musolino C, Irrera N, Bitto A, Lombardo D, Timmoneri M, Minutoli L, Raimondo G, Squadrito G, Squadrito F, Altavilla D (2018) Flavocoxid exerts a potent antiviral effect against hepatitis B virus. Inflamm Res 67:89–103. https://doi.org/10.1007/s00011-017-1099-2
Qian K, Kong ZR, Zhang J, Cheng XW, Wu ZY, Gu CX, Shao HX, Qin AJ (2018) Baicalin is an inhibitor of subgroup J avian leukosis virus infection. Virus Res 248:63–70. https://doi.org/10.1016/j.virusres.2018.02.017
Russo M, Moccia S, Spagnuolo C, Tedesco I, Russo GL (2020) Roles of flavonoids against coronavirus infection. Chem Biol Interact 328:109211. https://doi.org/10.1016/j.cbi.2020.109211
Shi HF, Ren K, Lv BJ, Zhang W, Zhao Y, Tan RX, Li EG (2016) Baicalin from Scutellaria baicalensis blocks respiratory syncytial virus (RSV) infection and reduces inflammatory cell infiltration and lung injury in mice. Sci Rep 6:35851. https://doi.org/10.1038/srep35851
Song JZ, Li LJ, Ji L, Shun L, Rui Y (2017) The pharmacokinetics of Tiangou antihypertensive capsule in rat in vivo. Biomed Rep 6:113–119. https://doi.org/10.3892/br.2016.810
Su Y, Xiang YM (2014) Clinical efficacy of baicalin combined with antivirals in the treatment of severe influenza A HIN1 influenza. Chin Hosp Pharm J 34:306–308
Su HX, Yao S, Zhao WF, Li MJ, Liu J, Shang WJ, Xie H, Ke CQ, Hu HC, Gao MN, Yu KQ, Liu H, Shen JS, Tang W, Zhang LK, Xiao GF, Ni L, Wang DW, Zuo JP, Jiang HL, Bai F, Wu Y, Ye Y, Xu YC (2020) Anti-SARS-CoV-2 activities in vitro of Shuanghuanglian preparations and bioactive ingredients. Acta Pharmacol Sin 41:1167–1177. https://doi.org/10.1038/s41401-020-0483-6
Sun XY (1995) Sun FY (1995) Shennong Bencao Jing. The Commercial Press, Peking
Tong L, Wan M, Zhang L, Zhu Y, Sun H, Bi K (2012) Simultaneous determination of baicalin, wogonoside, baicalein, wogonin, oroxylin A and chrysin of Radix scutellariae extract in rat plasma by liquid chromatography tandem mass spectrometry. J Pharm Biomed Anal 70:6–12. https://doi.org/10.1016/j.jpba.2012.03.051
Wan QF, Gu LG, Yin SJ, Qi GJ, Zhang S, Li GM, Ge DY (2011) Protection effect of baicalin on lung injury of mice infected with influenza FM1. Chin Tradit Patent Med 26:2848–2851
Wan QF, Wang H, Han XB, Lin Y, Yang YH, Gu LG, Zhao JQ, Wang L, Huang L, Li YB, Yang YR (2014) Baicalin inhibits TLR7/MYD88 signaling pathway activation to suppress lung inflammation in mice infected with influenza A virus. Biomed Rep 2:437–441. https://doi.org/10.3892/br.2014.253
Wang Q, Wang YT, Pu SP, Zheng YT (2004) Zinc coupling potentiates anti-HIV-1 activity of baicalin. Biochem Biophys Res Commun 324:605–610. https://doi.org/10.1016/j.bbrc.2004.09.093
Wang Y, Yao Y, An R, You L, Wang X (2009) Simultaneous determination of puerarin, daidzein, baicalin, wogonoside and liquiritin of GegenQinlian decoction in rat plasma by ultra-performance liquid chromatography-mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 877:1820–1826. https://doi.org/10.1016/j.jchromb.2009.05.035
Wang Y, Yang J, Li X, Wang J (2012) The metabolism of baicalin in rat and the biological activities of the metabolites. Evid Based Complement Alternat Med 2012:404529. https://doi.org/10.1155/2012/404529
Wang MJ, Yang CH, Jin Y, Wan CB, Qian WH, Xing F, Li X, Liu YY (2020) Baicalin inhibits coxsackievirus B3 replication by reducing cellular lipid synthesis. Am J Chin Med 48:143–160. https://doi.org/10.1142/S0192415X20500081
Wei Y, Pi C, Yang G, Xiong X, Lan Y, Yang H, Zhou Y, Ye Y, Zou Y, Zheng W, Zhao L (2016) LC-UV Determination of baicalin in rabbit plasma and tissues for application in pharmacokinetics and tissue distribution studies of baicalin after intravenous administration of liposomal and injectable formulations. Molecules 21:444. https://doi.org/10.3390/molecules21040444
Wu H, Long X, Yuan F, Chen L, Pan S, Liu Y, Stowell Y, Li X (2014) Combined use of phospholipid complexes and self-emulsifying microemulsions for improving the oral absorption of a BCS class IV compound, baicalin. Acta Pharm Sin B 4:217–226. https://doi.org/10.1016/j.apsb.2014.03.002
Wu L, Bi Y, Wu H (2018) Formulation optimization and the absorption mechanisms of nanoemulsion in improving baicalin oral exposure. Drug Dev Ind Pharm 44:266–275. https://doi.org/10.1080/03639045.2017.1391831
Xia CJ, Tang WY, Geng P, Zhu HY, Zhou W, Huang H, Zhou P, Shi XL (2020) Baicalin down-regulating hepatitis B virus transcription depends on the liver-specific HNF4α-HNF1α axis. Toxicol Appl Pharm 403:115131. https://doi.org/10.1016/j.taap.2020.115131
Xing J, Chen X, Zhong D (2005) Absorption and enterohepatic circulation of baicalin in rats. Life Sci 78:140–146. https://doi.org/10.1016/j.lfs.2005.04.072
Xu Q, Zhou A, Wu H, Bi Y (2019) Development and in vivo evaluation of baicalin-loaded W/O nanoemulsion for lymphatic absorption. Pharm Dev Technol 24:1155–1163. https://doi.org/10.1080/10837450.2019.1646757
Yang F, Feng C, Yao YX, Qin AJ, Shao HX, Qian K (2020) Antiviral effect of baicalin on Marek’s disease virus in CEF cells. BMC Vet Res 16:371. https://doi.org/10.1186/s12917-020-02595-x
Ye JX, Wei W, Quan LH, Liu CY, Chang Q, Liao YH (2010) An LC-MS/MS method for the simultaneous determination of chlorogenic acid, forsythiaside A and baicalin in rat plasma and its application to pharmacokinetic study of shuang-huang-lian in rats. J Pharm Biomed Anal 52:625–630. https://doi.org/10.1016/j.jpba.2010.01.035
Yimam M, Zhao Y, Ma W, Jia Q, Do SG, Shin JH (2010) 90-day oral toxicity study of UP446, a combination of defined extracts of Scutellaria baicalensis and Acacia catechu, in rats. Food Chem Toxicol 48:1202–1209. https://doi.org/10.1016/j.fct.2010.02.011
Zhai XJ (2016) Clinical observation of entecavir dispersing tablet combined with baicalin capsule in treatment of chronic hepatitis B for 24 weeks. J Med Theor Prac 29:53–54
Zhan YC, Qin S, Chen F (2017) Study on effect and mechanism of baicalin on antiviral effects about respiratory syncytial virus. Int J Lab Med 38:1907–1909. https://doi.org/10.3969/j.issn.1673-4130.2017.14.013
Zhang L, Lin G, Kovács B, Jani M, Krajcsi P, Zuo Z (2007) Mechanistic study on the intestinal absorption and disposition of baicalein. Eur J Pharm Sci 31:221–231. https://doi.org/10.1016/j.ejps.2007.04.001
Zhang H, Chen Q, Zhou W, Gao S, Lin H, Ye S, Xu Y, Cai J (2013) Chinese medicine injection shuanghuanglian for treatment of acute upper respiratory tract infection: a systematic review of randomized controlled trials. Evid Based Complement Alternat Med 2013:987326. https://doi.org/10.1155/2013/987326
Zhang J, Cai W, Zhou Y, Liu Y, Wu X, Li Y, Lu J, Qiao Y (2015) Profiling and identification of the metabolites of baicalin and study on their tissue distribution in rats by ultra-high-performance liquid chromatography with linear ion trap-Orbitrap mass spectrometer. J Chromatogr B Analyt Technol Biomed Life Sci 985:91–102. https://doi.org/10.1016/j.jchromb.2015.01.018
Zhang J, Zhang S, Teng S, Zhai L (2016) An LC-MS/MS method for simultaneous determination of four flavonoids from Semen Oroxyli in rat plasma and its application to a pharmacokinetic study. J Chromatogr B Analyt Technol Biomed Life Sci 1020:96–102. https://doi.org/10.1016/j.jchromb.2016.03.030
Zhang P, Peng HJ, Gao CL, Ren XG, Xia ZK (2018) Effects of baicalin on the expression of I-IFN and SOCS1/3 in RSV infected rats. China J Tradit Chin Med Pharm 33:328–332
Zhang XH, Zhao QQ, Ci XT, Chen S, Chen LY, Lian JM, Xie Z, Ye YQ, Lv HY, Li HX, Lin WC, Zhang HM, Xie QM (2020) Effect of baicalin on bacterial secondary infection and inflammation caused by H9N2 AIV infection in chickens. BioMed Res Int 18:2524314. https://doi.org/10.1155/2020/2524314
Zhao Y, Kong H, Sun Y, Feng H, Zhang Y, Su X, Qu H, Wang Q (2014) Assessment of baicalin in mouse blood by monoclonal antibody-based icELISA. Biomed Chromatogr 28:1864–1868. https://doi.org/10.1002/bmc.3235
Zhi HJ, Zhu HY, Zhang YY, Lu Y, Li H, Chen DF (2019) In vivo effect of quantified flavonoids-enriched extract of Scutellaria baicalensis root on acute lung injury induced by influenza A virus. Phytomedicine 57:105–116. https://doi.org/10.1016/j.phymed.2018.12.009
Zhu Z, Zhao L, Liu X, Chen J, Zhang H, Zhang G, Chai Y (2010) Comparative pharmacokinetics of baicalin and wogonoside by liquid chromatography-mass spectrometry after oral administration of Xiaochaihu Tang and Radix scutellariae extract to rats. J Chromatogr B Analyt Technol Biomed Life Sci 878:2184–2190. https://doi.org/10.1016/j.jchromb.2010.06.021
Zhu H, Qian Z, He F, Liu M, Pan L, Zhang Q, Tang Y (2013) Novel pharmacokinetic studies of the Chinese formula Huang-Lian-Jie-Du-Tang in MCAO rats. Phytomedicine 20:767–774. https://doi.org/10.1016/j.phymed.2012.11.012
Zhu HY, Han L, Shi XL, Wang BL, Huang H, Wang X, Chen DF, Ju DW, Feng MQ (2015) Baicalin inhibits autophagy induced by influenza A virus H3N2. Antiviral Res 113:62–70. https://doi.org/10.1016/j.antiviral.2014.11.003
Acknowledgements
The authors thank the Shandong University of Traditional Chinese Medicine Library for providing access to ancient Materia Medica books.
Author information
Authors and Affiliations
Contributions
KL and YL conceived and designed the review. AC, QW, YL, and HW collected and analyzed literature information. KL and YL wrote the manuscript. CZ and XW reviewed the manuscript. All authors have read the final manuscript and approved its submission.
Corresponding authors
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, K., Liang, Y., Cheng, A. et al. Antiviral Properties of Baicalin: a Concise Review. Rev. Bras. Farmacogn. 31, 408–419 (2021). https://doi.org/10.1007/s43450-021-00182-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-021-00182-1