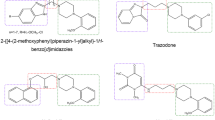
Abstract
Background
In our previous work, spiroxatrine was taken as reference compound to develop selective NOP ligands. Therefore, several triazaspirodecanone derivatives were synthesized. Here, we verify their selectivity towards other 5-HT1 receptor subtypes and with respect to α2-AR (Adrenergic Receptors).
Methods
Binding affinities were determined on cells expressing human cloned receptors for 5-HT1A/B/D and α2A/B/C subtypes. The Ki values were determined for those with at least 50% radioligand inhibition.
Results
All our derivatives show a moderate affinity for α2 subtypes, spanning from 5 to 7.5 pKi values. Moreover, they show affinity values in a μM–nM range at the 5-HT1A receptor, while they are practically inactive at 5-HT1B and 5-HT1D subtypes. Compound 11, the best of the series, has a 5-HT1A pKi value of 8.43 similar to spiroxatrine but, notably, it has a 5-HT1A favorable selectivity ratio of 52, 8 and 29, respectively over α2A, α2B and α2C adrenoceptor subtypes.
Conclusions
In this SAR study, a 5-HT1A selective ligand has been identified in which a tetralone moiety replaced the 1,4-benzodioxane of spiroxatrine and the methylene linker to the triazaspirodecanone portion was maintained in position 2.
Graphic abstract


Similar content being viewed by others
References
Caldwell JP, Matasi JJ, Zhang H, Fawzi A, Tulshian DB. Synthesis and structure-activity relationships of N-substituted spiropiperidines as nociceptin receptor ligands. Bioorg Med Chem Lett. 2007;17:2281–4. https://doi.org/10.1016/j.bmcl.2007.01.069.
Zaveri N, Polgar WE, Olsen CM, Kelson AB, Grundt P, Lewis JW, et al. Characterization of opiates, neuroleptics, and synthetic analogs at ORL1 and opioid receptors. Eur J Pharmacol. 2001;428:29–36. https://doi.org/10.1016/S0014-2999(01)01282-1.
Wichmann J, Adam G, Röver S, Henniq M, Scalone M, Cesura AM, et al. Synthesis of (1S,3aS)-8-(2,3,3a,4,5,6-hexahydro-1H-phenalen-1-yl)-1-phenyl-1,3,8-triaza-spiro[4.5]decan-4-one, a potent and selective orphanin FQ (OFQ) receptor agonist with anxiolytic-like properties. Eur J Med Chem. 2000;35(9):839–51. https://doi.org/10.1016/S0223-5234(00)00171-9.
Röver S, Adam G, Cesura AM, Galley G, Jenck F, Monsma FJ, et al. High-affinity, non-peptide agonists for the ORL1 (orphanin FQ/nociceptin) receptor. J Med Chem. 2000;43(7):1329–38. https://doi.org/10.1021/jm991129q.
Zaveri N, Jiang F, Olsen CM, Polgar WE, Toll L. Small-molecule agonists and antagonists of the opioid receptor-like receptor (ORL1, NOP): ligand-based analysis of structural factors influencing intrinsic activity at NOP. AAPS J. 2005;7(2):E345–52. https://doi.org/10.1208/aapsj070234.
Battisti UM, Corrado S, Sorbi C, Cornia A, Tait A, Malfacini D, et al. Synthesis, enantiomeric separation and docking studies of spiropiperidine analogues as ligands of the nociceptin/orphanin FQ receptor. MEDCHEMCOMM. 2014;5:973–83. https://doi.org/10.1039/C4MD00082J.
Corrado S, Sorbi C, Tait A, Battisti UM, Camarda V, Malfacini D, et al. 1,4-Dioxolane-triazaspirodecanone derivatives as nociceptin/orphanin FQ receptor ligands. Med Chem Res. 2014;23:4642–56. https://doi.org/10.1007/s00044-014-1032-y.
Corrado S, Battisti UM, Sorbi C, Tait A, Malfacini D, Camarda V, et al. Synthesis and structure-activity relationships of triazaspirodecanone derivatives as nociceptin/orphanin FQ receptor ligands. Chem Biol Drug Des. 2015;86:447–58. https://doi.org/10.1111/cbdd.12505.
Newman-Tancredi A. Biased agonism at serotonin 5-HT1A receptors: preferential postsynaptic activity for improved therapy of CNS disorders. Neuropsychiatry (London). 2011;1(2):149–64. https://doi.org/10.2217/npy.11.12.
Del Bello F, Ambrosini D, Bonifazi A, Newman AH, Keck TM, Giannella M, et al. Multitarget 1,4-dioxane compounds combining favorable D 2 -like and 5-HT 1A receptor interactions with potential for the treatment of Parkinson’s Disease or Schizophrenia. ACS Chem Neurosci. 2019;10(5):2222–8. https://doi.org/10.1021/acschemneuro.8b00677.
Chilmonczyk Z, Bojarski AJ, Pilc A, Sylte I. Serotonin transporter and receptor ligands with antidepressant activity as neuroprotective and proapoptotic agents. Pharmacol Rep. 2017;69(3):469–78. https://doi.org/10.1016/j.pharep.2017.01.011.
Staroń J, Bugno R, Hogendorf AS, Bojarski AJ. 5-HT1A receptor ligands and their therapeutic applications: review of new patents. Expert Opin Ther Pat. 2018;28(9):679–89. https://doi.org/10.1080/13543776.2018.1514011.
Rapalli A, Bertoni S, Arcaro V, Saccani F, Grandi A, Vivo V, et al. Dual role of endogenous serotonin in 2,4,6-trinitrobenzene sulfonic acid-induced colitis. Front Pharmacol. 2016;7:68. https://doi.org/10.3389/fphar.2016.00068.
Haleem DJ. Serotonin-1A receptor dependent modulation of pain and reward for improving therapy of chronic pain. Pharmacol Res. 2018;134:212–9. https://doi.org/10.1016/j.phrs.2018.06.030.
Lindberg A, Lu S, Nag S, Schou M, Liow JS, Zoghbi SS, et al. Synthesis and evaluation of two new candidate high-affinity full agonist PET radioligands for imaging 5-HT1B receptors. Nucl Med Biol. 2019;70:1–13. https://doi.org/10.1016/j.nucmedbio.2019.01.005.
Amidfar M, Colic L, Walter M, Kim YK. Biomarkers of major depression related to serotonin receptors. Curr Psychiatry Rev. 2018;14(4):239–44. https://doi.org/10.2174/1573400514666181016115747.
Goadsby PJ, Holland PR, Martins-Oliveira M, Hoffmann J, Schankin C, Akerman S. Pathophysiology of migraine: a disorder of sensory processing. Physiol Rev. 2017;97(2):553–622. https://doi.org/10.1152/physrev.00034.2015.
Negro A, Koverech A, Martelletti P. Serotonin receptor agonists in the acute treatment of migraine: a review on their therapeutic potential. J Pain Res. 2018;11:515–26. https://doi.org/10.2147/JPR.S132833.
Millan M. α2-Adrenergic mechanisms of analgesia: strategies for improving their therapeutic window and identification of the novel, potent α2A-adrenergic receptor agonist, S 1861. In: Goldstein DS, Eisenhofer G, McCarty R, editors. Advances in pharmacology, vol. 42. New York: Academic Press; 1997. p. 575–9. https://doi.org/10.1016/S1054-3589(08)60816-9.
Hisamieva LI, Zefirov AL, Ziyatdinova NI, Zefirov TL. Age features of α2C adrenoceptor JP-1302 selective blockade influence on rat myocardium inotropy. Int J Adv Biotechnol Res. 2016;7(4):1316–21.
Khisamieva LI, Ziyatdinova NI, Zefirov TL. The effect of blockade of α2A/D-adrenoreceptors on myocardial contractility in developing rats. Int J Pharm Technol. 2016;8(3):14980–7.
Duan L, Chen J, Razavi M, Wei Y, Tao Y, Rao X, Zhong J. Alpha2B-adrenergic receptor regulates neutrophil recruitment in MSU-induced peritoneal inflammation. Front Immunol. 2019;10:501. https://doi.org/10.3389/fimmu.2019.00501.
Cheng Y, Prusoff WH. Relationship between the inhibition constant (KI) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol. 1973;22:3099–108. https://doi.org/10.1016/0006-2952(73)90196-2.
Brasili L, Sorbi C, Franchini S, Manicardi M, Angeli P, Marucci G, et al. 1,3-dioxolane-based ligands as a novel class of α1-adrenoceptor antagonists. J Med Chem. 2003;46(8):1504–11. https://doi.org/10.1021/jm021078o.
Franchini S, Tait A, Prandi A, Sorbi C, Gallesi R, Buccioni M, et al. (2,2-Diphenyl-[1,3]oxathiolan-5-ylmethyl)-(3-phenyl-propyl)-amine: a potent and selective 5-HT(1A) receptor agonist. ChemMedChem. 2009;4(2):196–203. https://doi.org/10.1002/cmdc.200800276.
Acknowledgements
Receptor binding profiles were generously provided by the National Institute of Mental Health’s Psychoactive Drug Screening Program. Contract # HHSN-271-2018-00023-C (NIMH PDSP). The NIMH PDSP is directed by Bryan L. Roth MD, PhD at the University of North Carolina Chapel Hill and the Project Officer is Jamie Driscoll at the NIMH Bethesda (MD, USA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sorbi, C., Tait, A., Battisti, U.M. et al. Spiroxatrine derivatives towards 5-HT1A receptor selectivity. Pharmacol. Rep 72, 427–434 (2020). https://doi.org/10.1007/s43440-019-00039-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43440-019-00039-4