Abstract
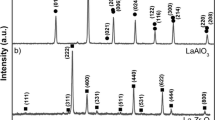
The substrate heating method was used to crystallize coatings containing faujasite zeolite on stainless steel plates from a clear reaction mixture. Conventional synthesis was also applied under similar conditions for comparison. The coatings obtained were characterized by X-ray diffraction (XRD), field emission gun scanning electron microscopy (FEGSEM), energy dispersive X-ray spectroscopy (EDX) and thermal gravimetry (TG). In general, mixed phases containing faujasite were determined to exist in the coatings prepared. The crystallinities as well as the phases of the zeolite coatings varied with the synthesis conditions used. The Si/Al ratios of the coatings were relatively low when substrate heating was applied, indicating the presence of zeolite X type faujasite, instead of the Y type obtained from conventional synthesis. When the substrate heating method was used, the mass equivalent coating thickness increased from about 30–150 µm, under similar synthesis conditions with the conventional method. Additionally, enhancements of up to about 4% and 23% were obtained in the water capacities of the coatings at 350 °C and 100 °C, respectively.






Similar content being viewed by others
Data availability
Data obtained in this study are available from the corresponding author on request.
References
Atakan A, Atalay-Oral C, Tatlier M, Erciyes T, Erdem-Şenatalar A (2012) Post-synthesis treatment for improving zeolite coating stability. Microporous Mesoporous Mater 156:262–269
Belviso C (2020) Zeolite for potential toxic metal uptake from contaminated soil: a brief review. Processes 8:820
Bonaccorsi L, Proverbio E (2004) Synthesis of thick zeolite 4A coatings on stainless steel. Microporous Mesoporous Mater 74:221–229
Bonaccorsi L, Freni A, Proverbio E, Restuccia G, Russo F (2006) Zeolite coated copper foams for heat pumping applications. Microporous Mesoporous Mater 91:7–14
Chanda R, Selvam T, Herrmann R, Schwieger W (2018) Reactive coating process for binder-free zeolite FAU films on metallic aluminum supports. Mater Lett 211:103–106
Clet G, Jansen JC, Van Bekkum H (1999) Synthesis of a zeolite Y coating on stainless steel support. Chem Mater 11:1696–1702
Dagli Z, Atalay-Oral C, Tasdelen-Yucedag C, Tatlier M (2022) How may preferential heating of the substrate aid template-free preparation of EMT zeolite and its coatings? Microporous Mesoporous Mater 337:111905
Diboune M, Nouali H, Soulard M, Patarin J, Rioland G, Faye D, Daou TJ (2020) Efficient removal of volatile organic compounds by FAU-type zeolite coatings. Molecules 25:3336
Dwyer J, Rawlence DJ (1993) Fluid catalytic cracking—chemistry. Catal Today 18:487–507
Erdem-Senatalar A, Tatlier M, Ürgen M (1999) Preparation of zeolite coatings by direct heating of the substrates. Microporous Mesoporous Mater 32:331–343
Firouzjaee M, Taghizadeh M (2020) Synthesis procedure and industrial applications of NaY zeolite for various processes: a review. Mini Rev Org Chem 17:795–804
Freni A, Bonaccorsi L, Calabrese L, Capri A, Frazzica A, Sapienza A (2015) SAPO-34 coated adsorbent heat exchanger for adsorption chillers. Appl Therm Eng 82:1–7
Ibrahim KM, Akashah T (2004) Lead removal from wastewater using faujasite tuff. Environ Geol 46:865–870
Kuzniatsova T, Kim Y, Shqau K, Dutta PK, Verweij H (2007) Zeta potential measurements of zeolite Y: application in homogeneous deposition of particle coatings. Microporous Mesoporous Mater 103:102–107
Lassinantti M, Hedlund J, Sterte J (2000) Faujasite-type films synthesized by seeding. Microporous Mesoporous Mater 38:25–34
Maras T, Nerat EY, Erdem A, Tatlier M (2021) Preparation of zeolite coatings by induction heating of the substrate. J Solgel Sci Technol 98:54–67
Reiprich B, Weissenberger T, Schwieger W, Inayat A (2020) Layer-like FAU-type zeolites: a comparative view on different preparation routes. Front Chem Sci Eng 14:127–142
Robson H, Lillerud KP (2016) Verified syntheses of zeolitic materials. Elsevier, Amsterdam
Schnabel L, Tatlier M, Schmidt F, Erdem-Şenatalar A (2010) Adsorption kinetics of zeolite coatings directly crystallized on metal supports for heat pump applications. Appl Therm Eng 30:1409–1416
Tatlier M, Atalay-Oral C (2022) Selection of a favorable zeolite for solar adsorption cooling: How straightforward is it? Chem Eng Commun. https://doi.org/10.1080/00986445.2022.2067750
Tatlier M, Munz G, Fueldner G, Henninger S (2014) Effect of zeolite A coating thickness on adsorption kinetics for heat pump applications. Microporous Mesoporous Mater 193:115–121
Tatlier M, Rustam L, Munz G (2019) Tailoring the reaction mixture composition for preparing zeolite coatings on aluminum supports in alkaline environments. Chem Eng Comm 206:953–966
Tatlier M, Atalay-Oral C, Bayrak A, Maraş T, Erdem A (2022) Impact of ion exchange on zeolite hydrophilicity/hydrophobicity monitored by water capacity using thermal analysis. Therm Acta 713:179240
Treacy MMJ, Higgins JB (2001) Collection of simulated XRD powder patterns for zeolites. Elsevier, Amsterdam
Yoshida S, Ogawa N, Kamioka K, Hirano S, Mori T (1999) Study of zeolite molecular sieves for production of oxygen by using pressure swing adsorption. Adsorption 5:57–61
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of ınterest
On behalf of all authors, the corresponding author states that there is no confict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dagli, Z., Atalay-Oral, C. & Tatlier, M. Effects of using substrate heating method for the preparation of zeolite coatings containing faujasite. Braz. J. Chem. Eng. 40, 817–825 (2023). https://doi.org/10.1007/s43153-022-00284-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-022-00284-w