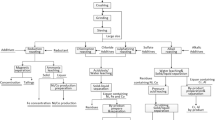
Abstract
The solvent extraction of Fe (residual), Mn, and Co from a real sulfuric Ni lateritic liquor (initial pH = 3) containing Ca and Mg was evaluated using D2EHPA and Cyanex 272 as extractants (A/O ratio = 1, T = 50 °C, one contact stage). Fe was practically extracted by both extractants at pH > 3. While Mn was selectively separated from Co and Ni with D2EHPA (5% v/v) at pH 3 (βMn/Co = 167), it was co-extracted with Co with Cyanex 272, which in turns is much more effective than D2EHPA in separating Co and Ni (βCo/Ni = 697 with 15% v/v of Cyanex 272 at pH 4.5). In addition, D2EHPA presented high affinity to Ca at low pH conditions, while Cyanex 272 extracted Ca and Mg at similar extents but at a higher pH level. Therefore, two SX circuits at staged operations, the first with D2EHPA and the second with Cyanex 272, can produce Co and Ni rich-streams. It was verified that Fe, Mn, as well as Ca, and part of Mg may be removed from the liquor at pH 3 with D2EHPA (5% v/v), while Co and remaining Mg could be separated from Ni with Cyanex 272 (15% v/v) at pH 4.5. Subsequent stripping and raffinate treatment strategies were presented. Data fitting using the slope analysis method revealed that coordinate complexes of Mn with D2EHPA, as well as Mn and Co with Cyanex 272, has the exact same MR2(RH)2 molecular structure.









Similar content being viewed by others
References
Agatzini-Leonardou S, Tsakiridis PE, Oustadakis P, Karidakis T, Katsiapi A (2009) Hydrometallurgical process for the separation and recovery of nickel from sulphate heap leach liquor of nickeliferrous laterite ores. Min Eng 22(14):1181–1192. https://doi.org/10.1016/j.mineng.2009.06.006
Chang Y, Zhai X, Li B, Fu Y (2010) Removal of iron from acidic leach liquor of lateritic nickel ore by goethite precipitate. Hydrometallurgy 101(1–2):84–87. https://doi.org/10.1016/j.hydromet.2009.11.014
Cheng CY (2000) Purification of synthetic laterite leach solution by solvent extraction using D2EHPA. Hydrometallurgy 56(3):369–386. https://doi.org/10.1016/S0304-386X(00)00095-5
Chong S, Hawker W, Vaughan J (2013) Selective reductive leaching of oxidised cobalt containing residue. Min Eng 54:82–87. https://doi.org/10.1016/j.mineng.2013.04.004
Donegan S (2006) Direct solvent extraction of nickel at Bulong operations. Min Eng 19(12):1234–1245. https://doi.org/10.1016/j.mineng.2006.03.003
Flett DS (2005) Solvent extraction in hydrometallurgy: the role of organophosphorus extractants. J Organomet Chem 690(10):2426–2438. https://doi.org/10.1016/j.jorganchem.2004.11.037
Guan Q-J, Sun W, Zhou G-Y, Liu J-P, Yin Z-G (2016) Recovery of cobalt and nickel in the presence of magnesium and calcium from sulfate solutions by Versatic 10 and mixtures of Versatic 10 and Cyanex 301. Trans Nonferrous Metals Soc China 26(3):865–873. https://doi.org/10.1016/S1003-6326(16)64178-X
Guimarães AS, Mansur MB (2017) Solvent extraction of calcium and magnesium from concentrate nickel sulfate solutions using D2EHPA and Cyanex 272 extractants. Hydrometallurgy 173:91–97. https://doi.org/10.1016/j.hydromet.2017.08.005
Guimarães AS, Mansur MB (2018) Selection of a synergistic solvent extraction system to remove calcium and magnesium from concentrated nickel sulfate solutions. Hydrometallurgy 175:250–256. https://doi.org/10.1016/j.hydromet.2017.12.001
Guimarães AS, Silva PS, Mansur MB (2014) Purification of nickel from multicomponent aqueous sulfuric solutions by synergistic solvent extraction using Cyanex 272 and Versatic 10. Hydrometallurgy 150:173–177. https://doi.org/10.1016/j.hydromet.2014.10.005
Guimarães AS, Silva LA, Pereira AM, Correia JCG, Mansur MB (2020) Purification of concentrated nickel sulfuric liquors via synergistic solvent extraction of calcium and magnesium using mixtures of D2EHPA and Cyanex 272. Sep Purif Technol 239:116570. https://doi.org/10.1016/j.seppur.2020.116570
Harvey R, Hannah R, Vaughan J (2011) Selective precipitation of mixed nickel-cobalt hydroxide. Hydrometallurgy 105(3–4):222–228. https://doi.org/10.1016/j.hydromet.2010.10.003
Ismael MRC, Carvalho JMR (2003) Iron recovery from sulphate leach liquors in zinc hydrometallurgy. Min Eng 16(1):31–39. https://doi.org/10.1016/S0892-6875(02)00310-2
Kolarik Z (1982) Critical evaluation of some equilibrium constants involving acidic organophosphorous extractants. Pure Appl Chem 54(12):2593–2674. https://doi.org/10.1351/pac198254122593
Kursunoglu S, Ichlas ZT, Kaya M (2017) Solvent extraction process for the recovery of nickel and cobalt from Caldag laterite leach solution: the first bench scale study. Hydrometallurgy 169:135–141. https://doi.org/10.1016/j.hydromet.2017.01.001
Lu J, Dreisinger D, Glück T (2016) Electrolytic manganese metal production from manganese carbonate precipitate. Hydrometallurgy 161:45–53. https://doi.org/10.1016/j.hydromet.2016.01.010
Mansur MB, Slater MJ, Biscaia EC Jr (2002) Equilibrium analysis of the reactive liquid–liquid test system ZnSO4/D2EHPA/n-heptane. Hydrometallurgy 63(2):117–126. https://doi.org/10.1016/S0304-386X(01)00211-0
Mansur MB, Slater MJ, Biscaia Junior EC (2003) Reactive extraction of zinc sulfate with bis (2-ethylhexyl) phosphoric acid in a short Kühni column used in batch mode. Ind Eng Chem Res 42(17):4068–4076. https://doi.org/10.1021/ie020883y
Mansur MB, Guimarães AS, Petraniková M (2021) An overview on the recovery of cobalt from end-of-life lithium ion batteries. Miner Process Extr Metall Rev. https://doi.org/10.1080/08827508.2021.1883014
Mantuano DP, Dorella G, Elias RCA, Mansur MB (2006) Analysis of a hydrometallurgical route to recover base metals from spent rechargeable batteries by liquid–liquid extraction with Cyanex 272. J Power Sources 159(2):1510–1518. https://doi.org/10.1016/j.jpowsour.2005.12.056
Ribeiro PPM, Santos ID, Neumann R, Fernandes A, Dutra AJB (2021) Roasting and leaching behavior of nickel laterite ore. Metall Mater Trans B 52:1739–1754. https://doi.org/10.1007/s11663-021-02141-6
Ritcey GM (2006) Solvent extraction - Principles and applications to process metallurgy, 2nd edn. GM Ritcey & Associates Inc., Ottawa
Roriz ERR, Espinosa DCR, Tenório JAS (2016) Battery recycling: effect of current density on manganese recovery through electrolytic process. Braz J Chem Eng 33(2):271–277. https://doi.org/10.1590/0104-6632.20160332s00003588
Santos LH, Carvalho PLG, Rodrigues GD, Mansur MB (2015) Selective removal of calcium from sulfate solutions containing magnesium and nickel using aqueous two phases systems (ATPS). Hydrometallurgy 156:259–263. https://doi.org/10.1016/j.hydromet.2015.06.010
Sicupira DC, Tolentino Silva T, Leão VA, Mansur MB (2014) Batch removal of manganese from acid mine drainage using bone char. Braz J Chem Eng 31(1):195–204. https://doi.org/10.1590/S0104-66322014000100018
Silva MF, Oliveira MRS, Santos ID, Radino-Rouse P, Mansur MB (2022) Iron precipitation strategies from nickel laterite ore sulfuric acid leach liquor. Min Process Extr Metall Rev 43(1):28–39. https://doi.org/10.1080/08827508.2020.1809392
Sist S, Demopoulos GP (2003) Nickel hydroxide precipitation from aqueous sulfate media. JOM 55:42–46. https://doi.org/10.1007/s11837-003-0104-0
Souza MFA, Mansur MB (2019) Competing solvent extraction of calcium and/or nickel with Cyanex 272 and/or D2EHPA. Braz J Chem Eng 36(1):541–547. https://doi.org/10.1590/0104-6632.20190361s20170527
Tait BK (1993) Cobalt-nickel separation: the extraction of cobalt(II) and nickel(II) by Cyanex 301, Cyanex 302 and Cyanex 272. Hydrometallurgy 32(3):365–372. https://doi.org/10.1016/0304-386X(93)90047-H
Tsakiridis PE, Agatizini-Leonardou S (2004) Process for recovery of cobalt and nickel in the presence of magnesium from sulphate solutions by Cyanex 272 and Cyanex 302. Min Eng 17(7–8):913–923. https://doi.org/10.1016/j.mineng.2004.03.010
Tsakiridis PE, Agatzini SL (2004) Process for the recovery of cobalt and nickel in the presence of magnesium and calcium from sulphate solutions by Versatic 10 and Cyanex 272. Min Eng 17(4):535–543. https://doi.org/10.1016/j.mineng.2003.12.003
Whittington BI, Muir D (2000) Pressure acid leaching of nickel laterites: A review. Min Process Extr Metall Rev 21(6):527–599. https://doi.org/10.1080/08827500008914177
Williams C, Hawker W, Vaughan JW (2013) Selective leaching of nickel from mixed nickel cobalt hydroxide precipitate. Hydrometallurgy 138:84–92. https://doi.org/10.1016/j.hydromet.2013.05.015
Zhu Z, Pranolo Y, Zhang W, Wang W, Cheng CY (2010) Precipitation of impurities from synthetic laterite leach solutions. Hydrometallurgy 104(1):81–85. https://doi.org/10.1016/j.hydromet.2010.05.003
Ritcey GM, Lucas BH (1974) Diluent and modifiers – Their effects on mass transfer and separation. In: Proc. of the Int Solvent Extraction Conf, p. 2437–2481, Lyon, France
Acknowledgements
The authors wish to thank CAPES (Finance Code 001) and CNPq (PQ1C grant nº 304018/2020-1) for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing interests that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guimarães, A.S., Silva, M., Resende, G. et al. Solvent extraction of metals from a Brazilian nickel lateritic liquor with D2EHPA and Cyanex 272. Braz. J. Chem. Eng. 40, 599–606 (2023). https://doi.org/10.1007/s43153-022-00252-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-022-00252-4