Abstract
Mucin 16 (MUC16) participates in the process of embryo implantation, but few studies have examined the association between MUC16 and pregnancy loss. To investigate this association, the expression of MUC16 in serum and decidua was compared between women with pregnancy loss and ongoing pregnancies. In vitro experiments and animal models were used to explore the role and underlying mechanisms of MUC16 in pregnancy loss. In human study, the expression of MUC16 in serum and decidua was both consistently lower in the women with pregnancy loss compared with those in women with ongoing pregnancies. In vitro experiments revealed the interaction of MUC16 with peripheral blood natural killer (pNK) cells. MUC16 changed the phenotype and reduced the pro-inflammation ability of pNK cells. MUC16 also inhibited the cytotoxicity of pNK cells through the Src homology region 2 domain-containing phosphatase-1/extracellular signal-regulated kinase (SHP-ERK) pathway. Furthermore, MUC16 promoted the migration, invasion and tube formation of trophoblast cells by co-culturing together with pNK cells. In vivo experiments, the mouse model of abortion was used to further confirm that intraperitoneal administration of MUC16 could rescue the pregnancy loss. This study reveals the still-unknown connection between MUC16 and pNK cells and indicates that MUC16 provides a novel method for future prediction and treatment of unfavorable pregnancy outcomes.
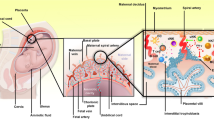
Graphical Abstract
Representative cartoon illustrating the effects of MUC16. A. The direct effects of MUC16 were to shape the phenotype and decrease TNF-α secretion of pNK cells, while inhibiting their cytotoxicity through the SHP-ERK pathway. B. The indirect effects of MUC16 were to promote the tube formation and invasion, migration of extravillous trophoblast cells (EVTs) by co-culturing with pNK cells.








Similar content being viewed by others
Data Availability
All data and materials support the published claims and comply with field standards.
Code availability
Software application or custom code support the published claims and comply with field standards.
References
Felder M, Kapur A, Gonzalez-Bosquet J, et al. MUC16 (CA125): tumor biomarker to cancer therapy, a work in progress. Mol Cancer. 2014;13(1):129.
Gipson IK, Mandel U, Menon B, et al. Generation and characterization of a monoclonal antibody to the cytoplasmic tail of MUC16. Glycobiology. 2017;27(10):920–6.
Perez BH, Gipson IK. Focus on Molecules: human mucin MUC16. Exp Eye Res. 2008;87(5):400–1.
Gipson IK, Blalock T, Tisdale A, et al. MUC16 is lost from the uterodome (pinopode) surface of the receptive human endometrium: in vitro evidence that MUC16 is a barrier to trophoblast adherence. Biol Reprod. 2008;78(1):134–42.
Liu L, Wang Y, Chen X, et al. Evidence from three cohort studies on the expression of MUC16 around the time of implantation suggests it is an inhibitor of implantation. J Assist Reprod Genet. 2020;37(5):1105–15.
Seki K, Kikuchi Y, Uesato T, et al. Increased serum CA 125 levels during the first trimester of pregnancy. Acta Obstet Gynecol Scand. 1986;65(6):583–5.
Bon GG, Kenemans P, Verstraeten AA, et al. Maternal serum Ca125 and Ca15-3 antigen levels in normal and pathological pregnancy. Fetal Diagn Ther. 2001;16(3):166–72.
Shehata NAA, Ali HAA, Hassan A, et al. Doppler and biochemical assessment for the prediction of early pregnancy outcome in patients experiencing threatened spontaneous abortion. Int J Gynaecol Obstet. 2018;143(2):150–5.
Erlebacher A. Immunology of the maternal-fetal interface. Annu Rev Immunol. 2013;31:387–411.
Triggianese P, Perricone C, Chimenti MS, et al. Innate Immune System at the Maternal-Fetal Interface: Mechanisms of Disease and Targets of Therapy in Pregnancy Syndromes. Am J Reprod Immunol. 2016;76(4):245–57.
Zhu L, Aly M, Wang H, et al. Increased natural killer cell subsets with inhibitory cytokines and inhibitory surface receptors in patients with recurrent miscarriage and decreased or normal subsets in kidney transplant recipients late post-transplant. Clin Exp Immunol. 2018;193(2):241–54.
El-Badawy O, Helmy AS, Abbas AM, et al. Concordance between peripheral and decidual NK cell subsets and killer immunoglobulin-like receptors in women with recurrent spontaneous miscarriages. J Reprod Immunol. 2020;140:103130.
Kuon RJ, Vomstein K, Weber M, et al. The “killer cell story” in recurrent miscarriage: Association between activated peripheral lymphocytes and uterine natural killer cells. J Reprod Immunol. 2017;119:9–14.
Gubbels JA, Felder M, Horibata S, et al. MUC16 provides immune protection by inhibiting synapse formation between NK and ovarian tumor cells. Mol Cancer. 2010;9(1):11.
Lee CL, Vijayan M, Wang X, et al. Glycodelin-A stimulates the conversion of human peripheral blood CD16-CD56bright NK cell to a decidual NK cell-like phenotype. Hum Reprod. 2019;34(4):689–701.
Peng P, Song H, Xie C, et al. miR-146a-5p-mediated suppression on trophoblast cell progression and epithelial-mesenchymal transition in preeclampsia. Biol Res. 2021;54(1):30.
Taga S, Hayashi M, Nunode M, et al. miR-486-5p inhibits invasion and migration of HTR8/SVneo trophoblast cells by down-regulating ARHGAP5. Placenta. 2022;123:5–11.
Maia J, Fonseca BM, Teixeira N, et al. The endocannabinoids anandamide and 2-arachidonoylglycerol modulate the expression of angiogenic factors on HTR8/SVneo placental cells. Prostaglandins Leukot Essent Fatty Acids. 2022;180:102440.
Zhang Q, Liu RX, Chan KW, et al. Exosomal transfer of p-STAT3 promotes acquired 5-FU resistance in colorectal cancer cells. J Exp Clin Cancer Res. 2019;38(1):320.
Long Y, Li Y, Wang T, et al. Inflammation-related proteomics demonstrate landscape of fracture blister fluid in patients with acute compartment syndrome. Front Immunol. 2023;14:1161479.
Wang X, Yip KC, He A, et al. Plasma Olink Proteomics Identifies CCL20 as a Novel Predictive and Diagnostic Inflammatory Marker for Preeclampsia. J Proteome Res. 2022;21(12):2998–3006.
Bao XH, Chen BF, Liu J, et al. Olink proteomics profiling platform reveals non-invasive inflammatory related protein biomarkers in autism spectrum disorder. Front Mol Neurosci. 2023;16:1185021.
Ansariniya H, Zare F, Mosaffa N, et al. Immunologic deviations in recurrent spontaneous abortion mouse model. Am J Reprod Immunol. 2022;88(6):e13631.
Li YH, Zhou WH, Tao Y, et al. The Galectin-9/Tim-3 pathway is involved in the regulation of NK cell function at the maternal-fetal interface in early pregnancy. Cell Mol Immunol. 2016;13(1):73–81.
Felder M, Kapur A, Rakhmilevich AL, et al. MUC16 suppresses human and murine innate immune responses. Gynecol Oncol. 2019;152(3):618–28.
Gallazzi M, Baci D, Mortara L, et al. Prostate Cancer Peripheral Blood NK Cells Show Enhanced CD9, CD49a, CXCR4, CXCL8, MMP-9 Production and Secrete Monocyte-Recruiting and Polarizing Factors. Front Immunol. 2020;11:586126.
Jabrane-Ferrat N. Features of Human Decidual NK Cells in Healthy Pregnancy and During Viral Infection. Front Immunol. 2019;10:1397.
Sun M, Gao J, Meng T, et al. Cyclin G2 upregulation impairs migration, invasion, and network formation through RNF123/Dvl2/JNK signaling in the trophoblast cell line HTR8/SVneo, a possible role in preeclampsia. FASEB J. 2021;35(2):e21169.
Keppel MP, Yang L, Cooper MA. Murine NK cell intrinsic cytokine-induced memory-like responses are maintained following homeostatic proliferation. J Immunol. 2013;190(9):4754–62.
King K, Smith S, Chapman M, et al. Detailed analysis of peripheral blood natural killer (NK) cells in women with recurrent miscarriage. Hum Reprod. 2010;25(1):52–8.
Raghupathy R. Th1-type immunity is incompatible with successful pregnancy. Immunol Today. 1997;18(10):478–82.
Yui J, Garcia-Lloret M, Wegmann TG, et al. Cytotoxicity of tumour necrosis factor-alpha and gamma-interferon against primary human placental trophoblasts. Placenta. 1994;15(8):819–35.
Azargoon A, Mirrasouli Y, Shokrollahi Barough M, et al. The State of Peripheral Blood Natural Killer Cells and Cytotoxicity in Women with Recurrent Pregnancy Loss and Unexplained Infertility. Int J Fertil Steril. 2019;13(1):12–7.
Brosens I, Pijnenborg R, Vercruysse L, et al. The “Great Obstetrical Syndromes” are associated with disorders of deep placentation. Am J Obstet Gynecol. 2011;204(3):193–201.
Chaouat G, Cayol V, Mairovitz V, et al. Localization of the Th2 cytokines IL-3, IL-4, IL-10 at the fetomaternal interface during human and murine pregnancy and lack of requirement for Fas/Fas ligand interaction for a successful allogeneic pregnancy. Am J Reprod Immunol. 1999;42(1):1–13.
Muzikova E, Clark DA. Is spontaneous resorption in the DBA/2-mated CBA/J mouse due to a defect in “seed” or in “soil”? Am J Reprod Immunol. 1995;33(1):81–5.
Acknowledgements
We thank former members of the Key Laboratory of Reproductive Dysfunction Management of Zhejiang Province, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine for technical assistance.This research was supported by the Huadong Medicine Joint Funds of the Zhejiang Provincial Natural Science Foundation of China (Grant No. LHDMZ22H040001). We thank Liwen Bianji (Edanz) (www.liwenbianji.cn/) for editing the English text of a draft of this manuscript.
Funding
This research was supported by the Huadong Medicine Joint Funds of the Zhejiang Provincial Natural Science Foundation of China (Grant No. LHDMZ22H040001).
Author information
Authors and Affiliations
Contributions
Conceptualization, Xiaona Huang, Liu Liu; methodology, Xiaona Huang, Huizhen Lin, Yue Zhao; Data analysis, Xiaona Huang, Huizhen Lin, Peixin Wang, Hanqi Ying; Supervision: LiuLiu; Writing – Original Draft: Xiaona Huang, Huizhen Lin; Writing – Review & Editing: Songying Zhang, Liu Liu.
Corresponding author
Ethics declarations
Financial interests
The authors declare they have no financial interests.
Conflict of Interest
The authors declare no competing interests.
Ethical Approval
All experimental work included in this work was performed in compliance with ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments, and the study protocol was approved by approved by the institutional review board of Sir Run Run Shaw hospital (No.2023–0309). The animal experiment protocols were approved by the Zhejiang University Institutional Animal Care and Use Committee (No.26165).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Competing Interests
The authors declare no competing interests.
Ethics Approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee (IRB) of Zhejiang University approved this study.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
The authors affirm that human research participants provided informed consent for publication of the images in Fig. 1E and 1F.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Lin, H., Zhao, Y. et al. MUC16 can Predict the Pregnancy Outcomes in Human and Intraperitoneal Administration of MUC16 can Rescue Pregnancy Losses in Mouse Models. Reprod. Sci. (2024). https://doi.org/10.1007/s43032-024-01550-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43032-024-01550-7