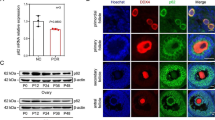
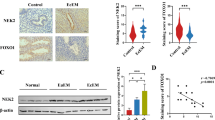
Abstract
Endometriosis (EMS) is a common benign gynecological disease affecting women of reproductive age. It is characterized by abnormal growth of endometrial tissue outside the uterine cavity, resulting in chronic pelvic pain and infertility. Endometrial physiological and pathological processes are intimately connected to autophagy. Mitophagy is an essential selective mode that protects cells from metabolic stress and hypoxia. Mitochondrial autophagy mediated by prohibitin 2 (PHB2) is dependent on the PRKN/Parkin pathway and is involved in numerous human diseases. Uncertainty remains as to whether mitophagy regulation by PHB2 contributes to the occurrence and progression of EMS. This study aims to investigate the mechanism underlying the role of PHB2 in EMS. This study detected the protein and mRNA expression of PHB2 in ectopic and normal endometrial tissues of ovarian EMS, in addition to ectopic endometrial cell line 12Z and endometrial stromal cell line KC02-44D for gene overexpression or knockdown. Cell function experiments and mitochondrial function experiments were conducted to investigate the role of PHB2 in the endometrium. Bioinformatic analysis and experiments were also used to investigate the upstream transcription factors that influence PHB2 expression. PHB2 was downregulated in ectopic endometrium, and PHB2 overexpression inhibited cell proliferation, migration, and invasion and promoted apoptosis. The upregulation of mitophagy markers, including Parkin and LC3II/I, and the downregulation of autophagy degradation markers P62 and TOMM20 in EMS suggest that PHB2 may contribute to cell proliferation, migration, invasion, and apoptosis via PRKN/Parkin-mediated mitophagy. Analysis and validation of bioinformatics data revealed that the transcription factor GABPA binds directly to the PHB2 promoter region and controls the transcriptional expression of PHB2. This study investigated the role of PHB2 in the onset of EMS. It inhibits EMS growth via PRKN/Parkin-mediated mitophagy, and GABPA controls the transcriptional disorder of PHB2. This study’s findings suggest a novel method for investigating the clinical potential of PHB2 in EMS.







Similar content being viewed by others
References
Falcone T, Flyckt R. Clinical management of endometriosis. Obstet Gynecol. 2018;131(3):557–71.
Giudice LC, Kao LC. Endometriosis. Lancet (London, England). 2004;364(9447):1789–99.
Simoens S, Dunselman G, Dirksen C, Hummelshoj L, Bokor A, Brandes I, Brodszky V, Canis M, Colombo GL, DeLeire T, Falcone T, Graham B, Halis G, Horne A, Kanj O, Kjer JJ, Kristensen J, Lebovic D, Mueller M, Vigano P, Wullschleger M, D’Hooghe T. The burden of endometriosis: Costs and quality of life of women with endometriosis and treated in referral centres. Human Reprod (Oxford, England). 2012;27(5):1292–9.
Glick D, Barth S, Macleod KF. Autophagy: Cellular and molecular mechanisms. J Pathol. 2010;221(1):3–12.
Shen HH, Zhang T, Yang HL, Lai ZZ, Zhou WJ, Mei J, Shi JW, Zhu R, Xu FY, Li DJ, Ye JF, Li MQ. Ovarian hormones-autophagy-immunity axis in menstruation and endometriosis. Theranostics. 2021;11(7):3512–26.
Lu H, Yang HL, Zhou WJ, Lai ZZ, Qiu XM, Fu Q, Zhao JY, Wang J, Li DJ, Li MQ. Rapamycin prevents spontaneous abortion by triggering decidual stromal cell autophagy-mediated NK cell residence. Autophagy. 2021;17(9):2511–27.
Zhan L, Li J, Wei B. Autophagy in endometriosis: Friend or foe? Biochem Biophys Res Commun. 2018;495(1):60–3.
Devis-Jauregui L, Eritja N, Davis ML, Matias-Guiu X, Llobet-Navàs D. Autophagy in the physiological endometrium and cancer. Autophagy. 2021;17(5):1077–95.
Zhou Z, Wang H, Zhang X, Song M, Yao S, Jiang P, Liu D, Wang Z, Lv H, Li R, Hong Y, Dai J, Hu Y, Zhao G. Defective autophagy contributes to endometrial epithelial-mesenchymal transition in intrauterine adhesions. Autophagy. 2022;18(10):2427–42.
Tosti C, Pinzauti S, Santulli P, Chapron C, Petraglia F. Pathogenetic mechanisms of deep infiltrating endometriosis. Reprod Sci (Thousand Oaks, Calif). 2015;22(9):1053–9.
Zhao Y, Chen Y, Kuang Y, Bagchi MK, Taylor RN, Katzenellenbogen JA, Katzenellenbogen BS. Multiple beneficial roles of repressor of estrogen receptor activity (REA) in suppressing the progression of endometriosis. Endocrinology. 2016;157(2):900–12.
Yoshimaru T, Komatsu M, Matsuo T, Chen YA, Murakami Y, Mizuguchi K, Mizohata E, Inoue T, Akiyama M, Yamaguchi R, Imoto S, Miyano S, Miyoshi Y, Sasa M, Nakamura Y, Katagiri T. Targeting BIG3-PHB2 interaction to overcome tamoxifen resistance in breast cancer cells. Nat Commun. 2013;4:2443.
Wei Y, Chiang WC, Sumpter R, Mishra P, Levine B. Prohibitin 2 is an inner mitochondrial membrane mitophagy receptor. Cell. 2017;168(1–2):224-238.e10.
Saunders PTK, Horne AW. Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell. 2021;184(11):2807–24.
Ristevski S, O’Leary DA, Thornell AP, Owen MJ, Kola I, Hertzog PJ. The ETS transcription factor GABPalpha is essential for early embryogenesis. Mol Cell Biol. 2004;24(13):5844–9.
Khan A, Fornes O, Stigliani A, Gheorghe M, Castro-Mondragon JA, van der Lee R, Bessy A, Chèneby J, Kulkarni SR, Tan G, Baranasic D, Arenillas DJ, Sandelin A, Vandepoele K, Lenhard B, Ballester B, Wasserman WW, Parcy F, Mathelier A. JASPAR 2018: Update of the open-access database of transcription factor binding profiles and its web framework. Nucleic Acids Res. 2018;46(D1):D260–6.
Sampson JA. Metastatic or embolic endometriosis, due to the menstrual dissemination of endometrial tissue into the venous circulation. Am J Pathol. 1927;3(2):93.
Nisenblat V, Bossuyt PMM, Shaikh R, Farquhar C, Jordan V, Scheffers CS, Mol BWJ, Johnson N, Hull ML. 2016 Blood biomarkers for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;5:CD012179.
Nisenblat V, Bossuyt PMM, Farquhar C, Johnson N, Hull ML. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;2(2):CD009591.
Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382(13):1244–56.
Horne AW, Saunders PTK. SnapShot: Endometriosis. Cell. 2019;179(7):1677-1677.e1.
Guo SW. Recurrence of endometriosis and its control. Hum Reprod Update. 2009;15(4):441–61.
Vercellini P, Viganò P, Somigliana E, Fedele L. Endometriosis: Pathogenesis and treatment. Nat Rev Endocrinol. 2014;10(5):261–75.
Fung C, Lock R, Gao S, Salas E, Debnath J. Induction of autophagy during extracellular matrix detachment promotes cell survival. Mol Biol Cell. 2008;19(3):797–806.
Mao Y, Qiao JD, Chen S, Zhou X, Wang Z, Cai S, Li L, Luo Y. Kallistatin inhibits anoikis resistance and metastasis of ectopic endometrium cells by modulating MnSOD and caspase 3 signaling. Reprod Sci (Thousand Oaks, Calif). 2021;28(4):1012–9.
Zhao Q, Ye M, Yang W, Wang M, Li M, Gu C, Zhao L, Zhang Z, Han W, Fan W, Meng Y. Effect of Mst1 on Endometriosis apoptosis and migration: Role of Drp1-related mitochondrial fission and Parkin-required mitophagy. Cell Physiol Biochem. 2018;45(3):1172–90.
Ruiz A, Rockfield S, Taran N, Haller E, Engelman RW, Flores I, Panina-Bordignon P, Nanjundan M. Effect of hydroxychloroquine and characterization of autophagy in a mouse model of endometriosis. Cell Death Dis. 2016;7(1):e2059.
Siracusa R, D’Amico R, Impellizzeri D, Cordaro M, Peritore AF, Gugliandolo E, Crupi R, Salinaro AT, Raffone E, Genovese T, Cuzzocrea S, Fusco R, Di Paola R. Autophagy and mitophagy promotion in a rat model of endometriosis. Int J Mol Sci. 2021;22(10):5074.
D’Amico R, Impellizzeri D, Cordaro M, Siracusa R, Interdonato L, Marino Y, Crupi R, Gugliandolo E, Macrì F, Di Paola D, Peritore AF, Fusco R, Cuzzocrea S, Di Paola R. Complex interplay between autophagy and oxidative stress in the development of endometriosis. Antioxidants (Basel, Switzerland). 2022;11(12):2484.
Pickles S, Vigié P, Youle RJ. Mitophagy and quality control mechanisms in mitochondrial maintenance. Curr Biol CB. 2018;28(4):R170–85.
Liu L, Sakakibara K, Chen Q, Okamoto K. Receptor-mediated mitophagy in yeast and mammalian systems. Cell Res. 2014;24(7):787–95.
Cheng J, Gao F, Chen X, Wu J, Xing C, Lv Z, Xu W, Xie Q, Wu L, Ye S, Xie H, Zheng S, Zhou L. Prohibitin-2 promotes hepatocellular carcinoma malignancy progression in hypoxia based on a label-free quantitative proteomics strategy. Mol Carcinog. 2014;53(10):820–32.
Shen Y, Gao Y, Yuan H, Cao J, Jia B, Li M, Peng Y, Du X, Zhang J, Shi J. Prohibitin-2 negatively regulates AKT2 expression to promote prostate cancer cell migration. Int J Mol Med. 2017;41(2):1147–55. https://doi.org/10.3892/ijmm.2017.3307.
Wu B, Chang N, Xi H, Xiong J, Zhou Y, Wu Y, Wu S, Wang N, Yi H, Song Y, Chen L, Zhang J. PHB2 promotes tumorigenesis via RACK1 in non-small cell lung cancer. Theranostics. 2021;11(7):3150–66.
Cai XW, Yu WW, Yu W, Zhang Q, Feng W, Liu MN, Sun MH, Xiang JQ, Zhang YW, Fu XL. Tissue-based quantitative proteomics to screen and identify the potential biomarkers for early recurrence/metastasis of esophageal squamous cell carcinoma. Cancer Med. 2018;7(6):2504–17.
Ferro F, Servais S, Besson P, Roger S, Dumas JF, Brisson L. Autophagy and mitophagy in cancer metabolic remodelling. Semin Cell Dev Biol. 2020;98:129–38.
Xu Y, Wang J, Xu W, Ding F, Ding W. Prohibitin 2-mediated mitophagy attenuates renal tubular epithelial cells injury by regulating mitochondrial dysfunction and NLRP3 inflammasome activation. Am J Physiol Renal Physiol. 2019;316(2):F396–407.
Xiao Y, Zhou Y, Lu Y, Zhou K, Cai W. PHB2 interacts with LC3 and SQSTM1 is required for bile acids-induced mitophagy in cholestatic liver. Cell Death Dis. 2018;9(2):160.
Xiang X, Fu Y, Zhao K, Miao R, Zhang X, Ma X, Liu C, Zhang N, Qu K. Cellular senescence in hepatocellular carcinoma induced by a long non-coding RNA-encoded peptide PINT87aa by blocking FOXM1-mediated PHB2. Theranostics. 2021;11(10):4929–44.
Zhang H, Yin C, Liu X, Bai X, Wang L, Xu H, Ju J, Zhang L. Prohibitin 2/PHB2 in Parkin-mediated mitophagy: A potential therapeutic target for non-small cell lung carcinoma. Med Sci Monit. 2020;26:e923227-1. https://www.medscimonit.com/abstract/index/idArt/923227.
Yan C, Gong L, Chen L, Xu M, Abou-Hamdan H, Tang M, Désaubry L, Song Z. PHB2 (prohibitin 2) promotes PINK1-PRKN/Parkin-dependent mitophagy by the PARL-PGAM5-PINK1 axis. Autophagy. 2020;16(3):419–34.
Zhao Y, Park S, Bagchi MK, Taylor RN, Katzenellenbogen BS. The coregulator, repressor of estrogen receptor activity (REA), is a crucial regulator of the timing and magnitude of uterine decidualization. Endocrinology. 2013;154(3):1349–60.
Sizemore GM, Pitarresi JR, Balakrishnan S, Ostrowski MC. The ETS family of oncogenic transcription factors in solid tumours. Nat Rev Cancer. 2017;17(6):337–51.
Yuan X, Dai M, Xu D. TERT promoter mutations and GABP transcription factors in carcinogenesis: More foes than friends. Cancer Lett. 2020;493:1–9.
Guo Y, Yuan X, Li K, Dai M, Zhang L, Wu Y, Sun C, Chen Y, Cheng G, Liu C, Strååt K, Kong F, Zhao S, Bjorkhölm M, Xu D. GABPA is a master regulator of luminal identity and restrains aggressive diseases in bladder cancer. Cell Death Differ. 2020;27(6):1862–77.
Yuan X, Mu N, Wang N, Strååt K, Sofiadis A, Guo Y, Stenman A, Li K, Cheng G, Zhang L, Kong F, Ekblad L, Wennerberg J, Nilsson IL, Juhlin CC, Larsson C, Xu D. GABPA inhibits invasion/metastasis in papillary thyroid carcinoma by regulating DICER1 expression. Oncogene. 2019;38(7):965–79.
Paulsson JO, Wang N, Gao J, Stenman A, Zedenius J, Mu N, Lui WO, Larsson C, Juhlin CC. GABPA-dependent down-regulation of DICER1 in follicular thyroid tumours. Endocr Relat Cancer. 2020;27(5):295–308.
Zhang S, Zhang K, Ji P, Zheng X, Jin J, Feng M, Liu P. GABPA predicts prognosis and inhibits metastasis of hepatocellular carcinoma. BMC Cancer. 2017;17(1):380.
Ma X, Lin Q, Cui G, Zhao J, Wei X, Li R, Mao H, Ma Y, Liu P, Pang Y. GABPA Expression in Endometrial carcinoma: A prognostic marker. Dis Markers. 2021;2021:5552614.
Funding
This study was supported by funding from the National Key Research and Development Program of China (No. 2017YFC1001204).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Lili Kong and Chongdong Liu are the co-corresponding authors of this manuscript.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, Y., Lou, T., Kong, L. et al. Prohibitin2/PHB2, Transcriptionally Regulated by GABPA, Inhibits Cell Growth via PRKN/Parkin-dependent Mitophagy in Endometriosis. Reprod. Sci. 30, 3629–3640 (2023). https://doi.org/10.1007/s43032-023-01316-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01316-7