Abstract
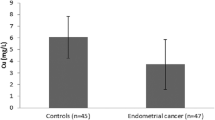
The objective of present study was to examine endometrial tissue Be, As, Cr, Mo, Sr, Ti, Tl, Cu, Co, Se, Zn, Mn, Fe, Cd, Pb, Mg, P, erythrocytes CAT, SOD, GSH-Px, GSH, MDA, serum retinol, cholecalciferol, phylloquinone, TSA, LSA, TOS, and TAS status and to evaluate the relationships between the variables. The study had 110 participants; of these, 50 were women with uterine myoma (UM), 10 were women with endometrial cancer (EC), and 50 were healthy female subjects. In the study, vitamin analyses by HPLC and element analyses were determined using ICP-OES method. It was observed that EC group was significantly lower than healthy group in terms of levels of cholecalciferol (p < 0.05), phylloquinone (p < 0.01), GSH (p < 0.05), Fe (p < 0.05), and had a significant rise in Mg/Fe (p < 0.01) and Zn/Fe (p < 0.05) in preoperative period. UM group had significantly lower retinol (p < 0.05), phylloquinone (p < 0.001), GSH-Px (p < 0.01), GSH (p < 0.01), Cr (p < 0.01), Cu (p < 0.05), Mg (p < 0.01), and Zn (p < 0.01) levels than control group in preoperative period and significantly higher levels of MDA (p < 0.01), TSA (p < 0.01), and LSA (p < 0.01) than control group. It was found that significant associations were observed between Cu–CA 15–3 (r = 0.558, p = 0.016), Mn–CA 15–3 (r = 0.511, p = 0.030), P–CA 15–3(r = − 0.502, p = 0.034) and with UM, also between GSH–CA-125 (r = − 0.825, p = 0.022) and with EC group. The results of correlation analysis observed that concentrations of Cu, Mn, P, and GSH together with CA 15–3 and CA-125 levels might be important for monitoring patients with UM and EC before surgery.
Graphical abstract










Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Caglayan A, Katlan DC, Tuncer ZS, Yuce K, Sayal HB, Kocer-Gumusel B. Assessment of oxidant-antioxidant status alterations with tumor biomarkers and reproductive system hormones in uterine myomas. Eur J Obstet Gynecol Reprod Biol. 2018;229:1–7. https://doi.org/10.1016/j.ejogrb.2018.07.031.
Tinelli A, Vinciguerra M, Malvasi A, Andjić M, Babović I, Sparić R. Uterine fibroids and diet. Int J Environ Res Public Health. 2021;18:1066. https://doi.org/10.3390/ijerph18031066.
Dalvi TB, Canchola AJ, Horn-Ross PL. Dietary patterns, Mediterranean diet, and endometrial cancer risk. Cancer Causes Control. 2007;18:957–66. https://doi.org/10.1007/s10552-007-9037-1.
Vural M, Camuzcuoglu H, Toy H, Camuzcuoglu A, Aksoy N. Oxidative stress and prolidase activity in women with uterine fibroids. J Obstet Gynaecol. 2012;32:68–72. https://doi.org/10.3109/01443615.2011.633718.
Rossi M, Edefonti V, Parpinel M, Lagiou P, Franchi M, Ferraroni M, et al. Proanthocyanidins and other flavonoids in relation to endometrial cancer risk: a case–control study in Italy. Br J Cancer. 2013;109:1914–20. https://doi.org/10.1038/bjc.2013.447.
Strzelczyk JK, Wiczkowski A. Oxidative damage and carcinogenesis. Contemp Oncol/WspółczesnaOnkol. 2012;16:230–233. https://doi.org/10.5114/wo.2012.29290.
Fletcher NM, Saed MG, Abu-Soud HM, Al-Hendy A, Diamond MP, Saed GM. Uterine fibroids are characterized by an impaired antioxidant cellular system: potential role of hypoxia in the pathophysiology of uterine fibroids. J Assist Reprod Genet. 2013;30:969–74. https://doi.org/10.1007/s10815-013-0029-7.
Manoharan S, Kolanjiappan K, Kayalvizhi M. Enhanced lipid peroxidation and impaired enzymic antioxidant activities in the erythrocytes of patients with cervical carcinoma. Cell MolBiol Lett. 2004;9:699–707.
Ay A, Gulyasar T, Alkanli N, Sipahi T, Cicin I, Kocak Z, et al. Investigation of the relationship between GSTM1 gene variations and serum trace elements, plasma malondialdehyde levels in patients with colorectal cancer. Mol Biol Rep. 2021;48:6911–21. https://doi.org/10.1007/s11033-021-06694-2.
Osuchowska-Grochowska I, Blicharska E, Gogacz M, Nogalska A, Winkler I, Szopa A, et al. Brief review of endometriosis and the role of trace elements. Int J Mol Sci. 2021;22:11098. https://doi.org/10.3390/ijms222011098.
Michalczyk K, Cymbaluk-Płoska A. The role of zinc and copper in gynecological malignancies. Nutrients. 2020;12:3732. https://doi.org/10.3390/nu12123732.
Ciebiera M, Ali M, Zgliczyńska M, Skrzypczak M, Al-Hendy A. Vitamins and uterine fibroids: current data on pathophysiology and possible clinical relevance. Int J Mol Sci. 2020;21:5528. https://doi.org/10.3390/ijms21155528.
Yasin HK, Taylor AH, Ayakannu T. A narrative review of the role of diet and lifestyle factors in the development and prevention of endometrial cancer. Cancers. 2021;13:2149. https://doi.org/10.3390/cancers13092149.
Karagul MI, Aktas S, Yilmaz SN, Yetkin D, Celikcan HD, Cevik OS. Perifosine and vitamin D combination induces apoptotic and non-apoptotic cell death in endometrial cancer cells. EXCLI J. 2020;19:532–546. https://doi.org/10.17179/excli2019-1834.
McCullough ML, Bandera EV, Moore DF, Kushi LH. Vitamin D and calcium intake in relation to risk of endometrial cancer: a systematic review of the literature. Prev Med. 2008;46:298–302. https://doi.org/10.1016/j.ypmed.2007.11.010.
Zhang Z, Wuhrer M, Holst S. Serum sialylation changes in cancer. Glycoconj J. 2018;35:139–60. https://doi.org/10.1007/s10719-018-9820-0.
Olaru OG, Constantin GI, Pena CM. Variation of total serum sialic acid concentration in postmenopausal women. Exp Ther Med. 2020;20:2455–9. https://doi.org/10.3892/etm.2020.8873.
Pecorelli S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol Obstet. 2009;105:103–4. https://doi.org/10.1016/j.ijgo.2009.02.012.
Rizzi R, Caroli A, Bolla P, Acciaioli A, Pagnacco G. Variability of reduced glutathione levels in Massese ewes and its effect on daily milk production. J Dairy Res. 1988;55:345–53. https://doi.org/10.1017/s0022029900028600.
Jain SK, McVie R, Duett J, Herbst JJ. Erythrocyte membrane lipid peroxidation and glycosylated hemoglobin in diabetes. Diabetes. 1989;38:1539–43. https://doi.org/10.2337/diab.38.12.1539.
Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70:158–69.
Sun YI, Oberley LW, Li Y. A simple method for clinical assay of superoxide dismutase. ClinChem. 1988;34:497–500.
Aebi H. Catalase in vitro. In: Packer L (ed) Methods in Enzymol. 1984;105:121–126. San Diego: Academic Press. https://doi.org/10.1016/S0076-6879(84)05016-3.
Sydow GA. Simplified quick method for determination of sialic acid in serum. Biomed Biochim Acta. 1985;44:1721–3.
Katopodis N, Hirshaut Y, Geller NL, Stock CC. Lipid-associated sialic acid test for the detection of human cancer. Cancer Res. 1982;42:5270–5.
Erel O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem. 2004;37:277–85. https://doi.org/10.1016/j.clinbiochem.2003.11.015.
Erel O. A new automated colorimetric method for measuring total oxidant status. Clin Biochem. 2005;38:1103–11. https://doi.org/10.1016/j.clinbiochem.2005.08.008.
Su Q, Rowley KG, Balazs ND. Carotenoids: separation methods applicable to biological samples. J Chromatogr B. 2002;781:393–418. https://doi.org/10.1016/s1570-0232(02)00502-0.
Siluk D, Oliveira RV, Esther-Rodriguez-Rosas M, Ling S, Bos A, Ferrucci L, et al. A validated liquid chromatography method for the simultaneous determination of vitamins A and E in human plasma. J Pharm Biomed Anal. 2007;44:1001–7. https://doi.org/10.1016/j.jpba.2007.03.033.
Lima MSO, da Silva BB, de Medeiros ML, Dos Santos AR, do Nascimento Brazil ED, et al. Evaluation of vitamin D receptor expression in uterine leiomyoma and nonneoplastic myometrial tissue: a cross‐sectional controlled study. Reprod Biol Endocrinol. 2021;19:67. https://doi.org/10.1186/s12958-021-00752-x.
Ding S, Madu CO, Lu Y. The impact of hormonal imbalances associated with obesity on the incidence of endometrial cancer in postmenopausal women. J Cancer. 2020;11:5456–65. https://doi.org/10.7150/jca.47580.
Karaman E, Karaman Y, Numanoglu C, Ark HC. Evaluation of hemoglobin A1c levels in endometrial cancer patients: a retrospective study in Turkey. Asian Pac J Cancer Prev. 2015;16:1817–20. https://doi.org/10.7314/APJCP.2015.16.5.1817.
Roškar L, Roškar I, Rižner TL, Smrkolj Š. Diagnostic and therapeutic values of angiogenic factors in endometrial cancer. Biomolecules. 2022;12:7. https://doi.org/10.3390/biom12010007.
Özcan HÇ, Öztürk E, Sucu S, Uğur MG, Kutlar İ, Balat Ö, et al. Evaluation of sialic acid levels in patients with hydatidiform mole: a preliminary study. Clin Exp Obstet Gynecol. 2016;43:414–416. https://doi.org/10.12891/ceog2156.2016.
Todorović A, Pejić S, Gavrilović L, Pavlović I, Stojiljković V, Popović N, et al. Expression of antioxidant enzymes in patients with uterine polyp, myoma, hyperplasia, and adenocarcinoma. Antioxidants. 2019;8:97. https://doi.org/10.3390/antiox8040097.
Paszkowska A, Berbeć H, Semczuk A, Cybulski M. Sialic acid concentration in serum and tissue of endometrial cancer patients. Eur J Obstet Gynecol Reprod Biol. 1998;76:211–5. https://doi.org/10.1016/s0301-2115(97)00176-0.
Berghuis AY, Pijnenborg JF, Boltje TJ, Pijnenborg JM. Sialic acids in gynecological cancer development and progression: impact on diagnosis and treatment. Int J Cancer. 2022;150:678–87. https://doi.org/10.1002/ijc.33866.
Paffoni A, Somigliana E, Vigano’ P, Benaglia L, Cardellicchio L, Pagliardini L, et al. Vitamin D status in women with uterine leiomyomas. J Clin Endocrinol Metab. 2013;98:E1374-E1378. https://doi.org/10.1210/jc.2013-1777.
Fichera M, Török P, Tesarik J, Della Corte L, Rizzo G, Garzon S, et al. Vitamin D, reproductive disorders and assisted reproduction: evidences and perspectives. Int J Food Sci Nutr. 2020;71:276–85. https://doi.org/10.1080/09637486.2019.1661978.
Xu WH, Dai Q, Xiang YB, Zhao GM, Ruan ZX, Cheng JR, et al. Nutritional factors in relation to endometrial cancer: a report from a population-based case-control study in Shanghai. China Int J Cancer. 2007;120:1776–81. https://doi.org/10.1002/ijc.22456i.
Pelucchi C, Dal Maso L, Montella M, Parpinel M, Negri E, Talamini R, et al. Dietary intake of carotenoids and retinol and endometrial cancer risk in an Italian case–control study. Cancer Causes Control. 2008;19:1209–15. https://doi.org/10.1007/s10552-008-9190-1.
Liu JJ, Bertrand KA, Karageorgi S, Giovannucci E, Hankinson SE, Rosner B, et al. Prospective analysis of vitamin D and endometrial cancer risk. Ann Oncol. 2013;24:687. https://doi.org/10.1093/annonc/mds509.
Iyoti S, Neelima S, Biharilal SS, Achala S. Study of blood levels of antioxidant enzymes and erythrocyte malondialdehyde (MDA) in ovarian, cervical and uterine cancer at stage I. J Obstet Gynecol India. 2009;59:242–5.
Ngô C, Chéreau C, Nicco C, Weill B, Chapron C, Batteux F. Reactive oxygen species controls endometriosis progression. Am J Clin Pathol. 2009;175:225–34. https://doi.org/10.2353/ajpath.2009.080804.
Beevi SS, Rasheed MH, Geetha A. Evidence of oxidative and nitrosative stress in patients with cervical squamous cell carcinoma. Anal Chim Acta. 2007;375:119–23. https://doi.org/10.1016/j.cca.2006.06.028.
Pejic S, Kasapovic J, Todorovic A, Stojiljkovic V, Pajovic SB. Lipid peroxidation and antioxidant status in blood of patients with uterine myoma, endometrial polypus, hyperplastic and malignant endometrium. Biol Res. 2006;39:619–29. https://doi.org/10.4067/S0716-97602006000500005.
Ying JF, Lu ZB, Fu LQ, Tong Y, Wang Z, Li WF, et al. The role of iron homeostasis and iron-mediated ROS in cancer. Am J Cancer Res. 2021;11:1895.
Nayki C, Nayki U, Gunay M, Kulhan M, Çankaya M, Kafa A, Balci G. Oxidative and antioxidative status in the endometrium of patients with benign gynecological disorders. J Gynecol Obstet Hum Reprod. 2017;46:243–7. https://doi.org/10.1016/j.jogoh.2017.02.002.
Nasiadek M, Krawczyk T, Sapota A. Tissue levels of cadmium and trace elements in patients with myoma and uterine cancer. Hum Exp Toxicol. 2005;24:623–30. https://doi.org/10.1191/0960327105ht575oa.
Atakul T, Altinkaya SO, Abas BI, Yenisey C. Serum copper and zinc levels in patients with endometrial cancer. Biol Trace Elem Res. 2020;195:46–54. https://doi.org/10.1007/s12011-019-01844-x.
Caglayan A, Katlan DC, Tuncer ZS, Yüce K. Evaluation of trace elements associated with antioxidant enzymes in blood of primary epithelial ovarian cancer patients. J Trace Elem Med Biol. 2019;52:254–62. https://doi.org/10.1016/j.jtemb.2019.01.010.
Lai GL, Yeh CC, Yeh CY, Chen RY, Fu CL, Chen CH, et al. Decreased zinc and increased lead blood levels are associated with endometriosis in Asian Women. Reprod Toxicol. 2017;74:77–84. https://doi.org/10.1016/j.reprotox.2017.09.001.
Rodríguez-Tomàs E, Baiges-Gaya G, Castañé H, Arenas M, Camps J, Joven J. Trace elements under the spotlight: a powerful nutritional tool in cancer. J Trace Elem Med Biol. 2021;68:126858. https://doi.org/10.1016/j.jtemb.2021.126858.
Ciebiera M, Esfandyari S, Siblini H, Prince L, Elkafas H, Wojtyła C, et al. Nutrition in gynecological diseases: current perspectives. Nutrients. 2021;2021(13):1178. https://doi.org/10.3390/nu13041178.
Cunzhi H, Jiexian J, Xianwen Z, Jingang G, Shumin Z, Lili D. Serum and tissue levels of six trace elements and copper/zinc ratio in patients with cervical cancer and uterine myoma. Biol Trace Elem Res. 2003;94:113–22. https://doi.org/10.1385/bter:94:2:113.
Naidu MSK, Suryakar AN, Swami SC, Katkam RV, Kumbar KM. Oxidative stress and antioxidant status in cervical cancer patients. Indian J Clin Biochem. 2007;22:140–4. https://doi.org/10.1007/bf02913333.
Dring JC, Forma A, Chilimoniuk Z, Dobosz M, Teresiński G, Buszewicz G, et al. Essentiality of trace elements in pregnancy, fertility, and gynecologic cancers—a state-of-the-art review. Nutrients. 2021;14:185. https://doi.org/10.3390/nu14010185.
Feng Y, Zeng JW, Ma Q, Zhang S, Tang J, Feng JF. Serum copper and zinc levels in breast cancer: a meta-analysis. J Trace Elem Med Biol. 2020;62:126629. https://doi.org/10.1016/j.jtemb.2020.126629.
Iyshwarya BK, Mohammed V, Veerabathiran R. Genetics of endometriosis and its association with ovarian cancer. Gynecol Obstet Clin Med. 2021;1:177–85. https://doi.org/10.1016/j.gocm.2021.09.001.
Nasiadek M, Kilanowicz A, Darago A, Lazarenkow A, Michalska M. The effect of cadmium on the coagulation and fibrinolytic system in women with uterine endometrial cancer and myoma. Int J Occup Med Environ Health. 2013;26:291–301. https://doi.org/10.2478/s13382-013-0089-z.
Funding
This work was supported by a grant from the Van Yuzuncu Yil University (VanYYU) Scientific Research Projects Presidency (FDK/2020/6594).
Author information
Authors and Affiliations
Contributions
SE contributed to the study design and statistical analysis. FG, DY, and AB conducted the biochemical investigation and material collection. SE and EK performed data interpretation and manuscript preparation. RE contributed to the histopathology. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gök, F., Ekin, S., Karaman, E. et al. Total Sialic Acid, Antioxidant Enzyme Activities, Trace Elements, and Vitamin Status Before and After Surgery in Women with Uterine Myoma and Endometrial Cancer. Reprod. Sci. 30, 2743–2757 (2023). https://doi.org/10.1007/s43032-023-01241-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01241-9