Abstract
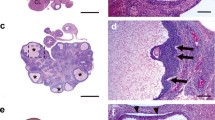
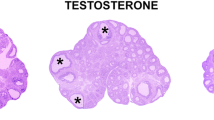
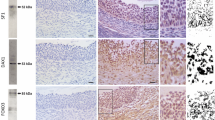
Androgens are relevant in order to achieve a normal growth and maturation of the follicle and oocyte, since both excess and absence of androgens may affect the correct ovarian function. The current study analyzes the impact of neonatal androgenization in the first ovulation and oocyte maturation in response to exogenous gonadotrophin stimulation. Neonatal rats were daily treated with testosterone, dihydrotestosterone, or vehicle during follicle assembly period (days 1 to 5). At juvenile period, rats were stimulated sequentially with PMSG and hCG. Ovulation, ovarian histology, hormonal milieu, morphological characteristics of meiotic spindle, and in vitro fertilization rate in oocytes were analyzed. Our data shows that oocytes from androgenized rats displayed a major proportion of aberrant spindles and altered meiotic advance that control animals. These alterations were accompanied with an increase in both fertilization rate and aberrant embryos after 48 h of culture. Our findings showed a direct impact of neonatal androgens on oocyte development; their effects may be recognized at adulthood, supporting the idea of a programming effect exerted by neonatal androgens. These results could be relevant to explain the low fertility rate seen in polycystic ovary syndrome patients after in vitro fertilization procedures.





Similar content being viewed by others
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
References
Liu Y-X, Zhang Y, Li Y-Y, et al. Regulation of follicular development and differentiation by intra-ovarian factors and endocrine hormones. Front Biosci. 2019;24:983–93.
Conti M, Franciosi F. Acquisition of oocyte competence to develop as an embryo: integrated nuclear and cytoplasmic events. Hum Reprod Update. 2018;24:245–66.
Astapova O, Minor BMN, Hammes SR. Physiological and pathological androgen actions in the ovary. Endocrinology. 2019;160:1166–74.
Prizant H, Gleicher N, Sen A. Androgen actions in the ovary: balance is key. Journal of Endocrinology. 2014;222:R141–51.
Galas J, Słomczyńska M, Knapczyk-Stwora K, Durlej M, Starowicz A, Tabarowski Z, et al. Steroid levels and the spatiotemporal expression of steroidogenic enzymes and androgen receptor in developing ovaries of immature rats. Acta Histochem. 2012;114:207–16.
Li M, Ai J-S, Xu B-Z, Xiong B, Yin S, Lin SL, et al. Testosterone potentially triggers meiotic resumption by activation of intra-oocyte SRC and MAPK in porcine oocytes. Biol Reprod. 2008;79:897–905.
Tarumi W, Itoh MT, Suzuki N. Effects of 5α-dihydrotestosterone and 17β-estradiol on the mouse ovarian follicle development and oocyte maturation. PLoS One. 2014;9:e99423.
Yeh S, Tsai M-Y, Xu Q, Mu XM, Lardy H, Huang KE, et al. Generation and characterization of androgen receptor knockout (ARKO) mice: an in vivo model for the study of androgen functions in selective tissues. Proc Natl Acad Sci U S A. 2002;99:13498–503.
Walters KA, Middleton LJ, Joseph SR, et al. Targeted loss of androgen receptor signaling in murine granulosa cells of preantral and antral follicles causes female subfertility. Biol Reprod. 2012;87:151.
Abbott DH, Dumesic DA, Levine JE. Hyperandrogenic origins of polycystic ovary syndrome - implications for pathophysiology and therapy. Expert Rev Endocrinol Metab. 2019;14:131–43.
Walters KA, Rodriguez Paris V, Aflatounian A, Handelsman DJ. Androgens and ovarian function: translation from basic discovery research to clinical impact. J Endocrinol. 2019;242:R23–50.
Anesetti G, Chávez-Genaro R. Ovarian follicular dynamics after aromatizable or non aromatizable neonatal androgenization. J Mol Histol. 2016;47:491–501.
Chávez-Genaro R, Anesetti G. First ovarian response to gonadotrophin stimulation in rats exposed to neonatal androgen excess. J Mol Histol. 2018;49:631–7.
Escobar-Morreale HF. Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat Rev Endocrinol. 2018;14:270–84.
Legro RS, Arslanian SA, Ehrmann DA, Hoeger KM, Murad MH, Pasquali R, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2013;98:4565–92.
Decanter C (2018) Oocyte quality in PCOS. Infertility in Women with Polycystic Ovary Syndrome 31–39
Sigala J, Sifer C, Dewailly D, Robin G, Bruyneel A, Ramdane N, et al. Is polycystic ovarian morphology related to a poor oocyte quality after controlled ovarian hyperstimulation for intracytoplasmic sperm injection? Results from a prospective, comparative study. Fertil Steril. 2015;103:112–8.
Wood JR, Dumesic DA, Abbott DH, Strauss JF 3rd. Molecular abnormalities in oocytes from women with polycystic ovary syndrome revealed by microarray analysis. J Clin Endocrinol Metab. 2007;92:705–13.
Ryu Y, Kim SW, Kim YY, Ku S-Y (2019) Animal models for human polycystic ovary syndrome (PCOS) focused on the use of indirect hormonal perturbations: a review of the literature. Int J Mol Sci 20. https://doi.org/10.3390/ijms20112720
Anesetti G, Chávez-Genaro R. Neonatal testosterone exposure induces early development of follicular cysts followed by sympathetic ovarian hyperinnervation. Reprod Fertil Dev. 2016;28:1753.
Sotomayor-Zárate R, Tiszavari M, Cruz G, Lara HE. Neonatal exposure to single doses of estradiol or testosterone programs ovarian follicular development-modified hypothalamic neurotransmitters and causes polycystic ovary during adulthood in the rat. Fertil Steril. 2011;96:1490–6.
Arai Y, Yamanouchi K, Mizukami S, Yanai R, Shibata K, Nagasawa H. Induction of anovulatory sterility by neonatal treatment with 5 beta-dihydrotestosterone in female rats. Acta Endocrinol. 1981;96:439–43.
Dai X, Zhang M, Lu Y, Miao Y, Zhou C, Xiong B. Cullin9 protects mouse eggs from aneuploidy by controlling microtubule dynamics via Survivin. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 2016;1863:2934–41.
Sanfins A, Lee GY, Plancha CE, Overstrom EW, Albertini DF. Distinctions in meiotic spindle structure and assembly during in vitro and in vivo maturation of mouse oocytes. Biol Reprod. 2003;69:2059–67.
Can A, Semiz O. Diethylstilbestrol (DES)-induced cell cycle delay and meiotic spindle disruption in mouse oocytes during in-vitro maturation. Mol Hum Reprod. 2000;6:154–62.
Stener-Victorin E, Padmanabhan V, Walters KA, Campbell RE, Benrick A, Giacobini P, et al. Animal models to understand the etiology and pathophysiology of polycystic ovary syndrome. Endocr Rev. 2020;41:538–76. https://doi.org/10.1210/endrev/bnaa010.
Vendola KA, Zhou J, Adesanya OO, Weil SJ, Bondy CA. Androgens stimulate early stages of follicular growth in the primate ovary. J Clin Invest. 1998;101:2622–9.
Forsdike RA, Hardy K, Bull L, Stark J, Webber LJ, Stubbs S, et al. Disordered follicle development in ovaries of prenatally androgenized ewes. J Endocrinol. 2007;192:421–8.
Romero-Ruiz A, Skorupskaite K, Gaytan F, Torres E, Perdices-Lopez C, Mannaerts BM, et al. Kisspeptin treatment induces gonadotropic responses and rescues ovulation in a subset of preclinical models and women with polycystic ovary syndrome. Hum Reprod. 2019;34:2495–512.
Yun YW, Yu FH, Yuen BH, Moon YS. Effects of a superovulatory dose of pregnant mare serum gonadotropin on follicular steroid contents and oocyte maturation in rats. Gamete Res. 1989;23:289–98.
Lee ST, Kim TM, Cho MY, et al. Development of a hamster superovulation program and adverse effects of gonadotropins on microfilament formation during oocyte development. Fertil Steril. 2005;83(Suppl 1):1264–74.
Ibáñez E, Sanfins A, Combelles CMH, et al. Genetic strain variations in the metaphase-II phenotype of mouse oocytes matured in vivo or in vitro. Reproduction. 2005;130:845–55.
Huang Y, Yu Y, Gao J, Li R, Zhang C, Zhao H, et al. Impaired oocyte quality induced by dehydroepiandrosterone is partially rescued by metformin treatment. PLoS One. 2015;10:e0122370.
Tarumi W, Tsukamoto S, Okutsu Y, Takahashi N, Horiuchi T, Itoh MT, et al. Androstenedione induces abnormalities in morphology and function of developing oocytes, which impairs oocyte meiotic competence. Fertil Steril. 2012;97:469–76.
Makita M, Miyano T. Androgens promote the acquisition of maturation competence in bovine oocytes. J Reprod Dev. 2015;61:211–7.
Hassold T, Hunt P. To err (meiotically) is human: the genesis of human aneuploidy. Nat Rev Genet. 2001;2:280–91.
Wang H, Höög C. Structural damage to meiotic chromosomes impairs DNA recombination and checkpoint control in mammalian oocytes. J Cell Biol. 2006;173:485–95.
Carbone L, Chavez SL. Mammalian pre-implantation chromosomal instability: species comparison, evolutionary considerations, and pathological correlations. Syst Biol Reprod Med. 2015;61:321–35.
Wei Y, Multi S, Yang C-R, Ma J, Zhang QH, Wang ZB, et al. Spindle assembly checkpoint regulates mitotic cell cycle progression during preimplantation embryo development. PLoS ONE. 2011;6:e21557.
Ma J-Y, Ou Yang Y-C, Wang Z-W, et al. The effects of DNA double-strand breaks on mouse oocyte meiotic maturation. Cell Cycle. 2013;12:1233–41.
Marangos P, Stevense M, Niaka K, Lagoudaki M, Nabti I, Jessberger R, et al. DNA damage-induced metaphase I arrest is mediated by the spindle assembly checkpoint and maternal age. Nat Commun. 2015;6:8706.
Yang K-T, Inoue A, Lee Y-J, Jiang CL, Lin FJ. Loss of Ikbkap/Elp1 in mouse oocytes causes spindle disorganization, developmental defects in preimplantation embryos and impaired female fertility. Sci Rep. 2019;9:18875.
Han L, Ge J, Zhang L, Ma R, Hou X, Li B, et al. Sirt6 depletion causes spindle defects and chromosome misalignment during meiosis of mouse oocyte. Sci Rep. 2015;5:15366.
Acknowledgements
We gratefully acknowledge Karina Hernández for histotechnical assistance, Mariela Santos for animal care support, and Dana Kimelman for the revision of the English manuscript.
Funding
This research was partially supported by the PEDECIBA, Universidad de la República, Montevideo, Uruguay.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Animal care and protocols were in accordance with international guides for use of laboratory animals and approved by the local Experimental Animal Committee (Comisión Honoraria de Experimentación Animal, CHEA, Universidad de la República, Protocol number: 070153-000358-18), Montevideo, Uruguay.
Consent to Participate
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Anesetti, G., Chávez-Genaro, R. Neonatal androgenization in rats affects oocyte maturation. Reprod. Sci. 28, 2799–2806 (2021). https://doi.org/10.1007/s43032-021-00559-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-021-00559-6