Abstract
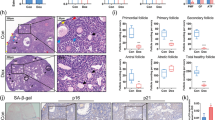
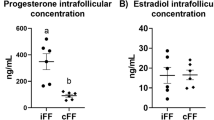
Human placenta mesenchymal stem cells (hPMSCs), have been extensively investigated on the treatment of many diseases. This study was to explore the effects of hPMSCs treatment on the chemotherapy drug-induced premature ovarian insufficiency (POI) mice. Cyclophosphamide (120 mg/kg) and busulfan (30 mg/kg) or cyclophosphamide (70 mg/kg) induced POI mice were used and hPMSCs were injected through tail vein. The hormone levels of serum, morphological changes, the expression and quantitative analysis of inhibin B (INHBB) and FSHR protein, and apoptosis of granulosa cells in ovary were detected. The granulosa cells (GCs) were detected from ovaries of mice and the different concentration of cyclophosphamide on GCs were detected by MTT assay. The apoptosis of GCs was detected by FITC Annexin V Apoptosis Detection Kit. The significant increase in FSH and decrease in E2 and INHBB were observed. Expression of human nuclei was observed in the stroma of ovaries. INHBB and FSHR levels of ovaries were reduced in the POI mice. Following hPMSCs treatment, the amounts of INHBB and FSHR significantly increased close to normal levels. The granulosa cells apoptosis increased in the POI ovaries but decreased after hPMSCs treatment. Moreover, cyclophosphamide has no effect on the GCs and no statistic difference was measured in vitro. The effects of hPMSCs treatment reduce the apoptosis of granulosa cells and restore the ovarian reserve capacity in chemotherapeutic drug–induced POI mice. The data help to further explore new potential clinical therapeutic approach for POI patients.






Similar content being viewed by others
References
Santoro N. Mechanisms of premature ovarian failure. Ann Endocrinol. 2003;64(2):87–92.
Laml T, Schulz-Lobmeyr I, Obruca A, Huber JC, Hartmann BW. Premature ovarian failure: etiology and prospects. Gynecol Endocrinol. 2000;14(4):292–302.
Webber L, Davies M, Anderson R, et al. ESHRE Guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926–37.
Skillern A, Rajkovic A. Recent developments in identifying genetic determinants of premature ovarian insufficiency. Sex Dev. 2008;2(4–5):228–43.
Knauff EA, Eijkemans MJ, Lambalk CB, ten Kate-Booij M, Hoek A, Beerendonk CC, et al. Anti-Mullerian hormone, inhibin B, and antral follicle count in young women with ovarian failure. J Clin Endocrinol Metab. 2009;94(3):786–92.
Palhares MB, Ferriani RA, Martins WP, Navarro PA. A formula combining age, AMH, AFC and FSH is more accurate than individual markers in predicting poor response to controlled ovarian stimulation in good prognosis patients. Fertil Steril. 2015;104(3):e330–1.
Peymani R, Najmabadi S. Anti-mullerian hormone (AMH), inhibin B, follicle stimulating hormone (FSH) and antral follicle count (AFC); which one is the best predictor of ovarian response in assisted reproductive technology? Fertil Steril. 2009;92(3):S168.
MeirellesLda S, Fontes AM, Covas DT, Caplan AI. Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev. 2009;20(5–6):419–27.
Shi Y, Hu G, Su J, et al. Mesenchymal stem cells: a new strategy for immunosuppression and tissue repair. Cell Res. 2010;20(5):510.
Chen CP. Placental villous mesenchymal cells trigger trophoblast invasion. Cell Adhes Migr. 2014;8(2):94–7.
Mohammadi Z, Afshari JT, Keramati MR, Alamdari DH, Ganjibakhsh M, Zarmehri AM, et al. Differentiation of adipocytes and osteocytes from human adipose and placental mesenchymal stem cells. Iran J Basic Med Sci. 2015;18(3):259–66.
Zhang Q, Xu M, Yao X, Li T, Wang Q, Lai D. Human amniotic epithelial cells inhibit granulosa cell apoptosis induced by chemotherapy and restore the fertility. Stem Cell Res Ther. 2015;6(1):152.
Bao R, Xu P, Wang Y, Wang J, Xiao L, Li G, et al. Bone marrow derived mesenchymal stem cells transplantation rescues premature ovarian insufficiency induced by chemotherapy. Gynecol Endocrinol. 2018;34(4):320–6.
Mohamed SA, Shalaby S, Brakta S, et al. Umbilical cord blood mesenchymal stem cells as an infertility treatment for chemotherapy induced premature ovarian insufficiency. Biomedicines. 2019;7(1):E7.
Ovalle WK, Nahirney PC. Netter’s essential histology E-book. Elsevier Health Sciences, 2nd ed. Philadelphia, PA, xv, 517. 2013.
Moore KL, Persaud TVN, Torchia MG. The developing human. Philadelphia: Saunders; 2011.
Pedersen T, Peters H. Proposal for a classification of oocytes and follicles in the mouse ovary. J Reprod Fertil. 1968;17(3):555–7.
Edwards RG, Fowler RE, Gore-Langton RE, Gosden RG, Jones EC, Readhead C, et al. Normal and abnormal follicular growth in mouse, rat and human ovaries. J Reprod Fertil. 1977;51(1):237–63.
Gougeon A. Regulation of ovarian follicular development in primates: facts and hypotheses. Endocr Rev. 1996;17(2):121–55.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Ana Bioch. 1976;7(72):248–54.
Zhang HQ, Luo QQ, Lu XY, Yin N, Zhou D, Zhang L, et al. Effects of hPMSCs on granulosa cell apoptosis and AMH expression and their role in the restoration of ovary function in premature ovarian insufficiency mice. Stem Cell Res Ther. 2018;9(1):20.
Zhang Q, Bu S, Sun J, Xu M, Yao X, He K, et al. Paracrine effects of human amniotic epithelial cells protect against chemotherapy-induced ovarian damage. Stem Cell Res Ther. 2017;8(1):270.
Ling L, Feng X, Wei T, Wang Y, Wang Y, Wang Z, et al. Human amnion-derived mesenchymal stem cell (hAD-MSC) transplantation improves ovarian function in rats with premature ovarian insufficiency (POI) at least partly through a paracrine mechanism. Stem Cell Res Ther. 2019;10(1):46.
Randolph JF Jr, Harlow SD, Helmuth ME, Zheng H, McConnell DS. Updated assays for inhibin B and AMH provide evidence for regular episodic secretion of inhibin B but not AMH in the follicular phase of the normal menstrual cycle. Hum Reprod. 2014;29(3):592–600.
Dixit H, Deendayal M, Singh L. Mutational analysis of the mature peptide region of inhibin genes in Indian women with ovarian failure. Hum Reprod. 2004;19(8):1760–4.
MacNaughton J, Banah M, McCloud P, Hee J, Burger H. Age related changes in follicle stimulating hormone, luteinizing hormone, oestradiol and immunoreactive inhibin in women of reproductive age. Clin Endocrinol. 1992;36(4):339–45.
Shahara FI, Scott RT, Seifer DB. The detection of diminished ovarian reserve in infertile women. Am J Obstet Gynecol. 1998;179(3 Pt 1):804–12.
Nadel MV. Aging and infertility in women: a committee opinion. Fertil Steril. 2002;78(1):215–9.
Findlay JK, Drummond AE, Dyson M, Baillie AJ, Robertson DM, Ethier JF. Production and actions of inhibin and activin during folliculogenesis in the rat. Mol Cell Endocrinol. 2001;180(1):139–44.
Bernard DJ, Woodruff TK, Plant TM. Cloning of a novel inhibin alpha cDNA from rhesus monkey testis. Reprod Biol Endocrinol. 2004;2(1):71.
Hugon-Rodin J, Kalhorpour N, Borghese B, Bordonne C, Just PA, Gompel A, et al. Inhibin a and inhibin B producing ovarian fibrothecoma revealed by suppression of follicle stimulating hormone (FSH) in a post-menopausal woman: report of the first case. Gynecol Endocrinol. 2016;32(11):872–4.
Karakaya C, Kayisli OG, Rebecca JH, et al. Follicle-stimulating hormone receptor (FSHR) alternative skipping of exon 2 or 3 affects ovarian response to FSH. Mol Hum Reprod. 2014;20(7):630–43.
Lussiana C, Guani B, Mari C, Restagno G, Massobrio M, Revelli A. Mutations and polymorphisms of the FSH receptor (FSHR) gene: clinical implications in female fecundity and molecular biology of FSHR protein and gene. Obstet Gynecol Surv. 2008;63(12):785–95.
Pu D, Xing Y, Gao Y, Gu L, Wu J. Gene variation and premature ovarian failure: a meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2014;182:226–37.
M’baye M, Hua G, Khan HA, Yang L. RNAi-mediated knockdown of INHBB increases apoptosis and inhibits steroidogenesis in mouse granulosa cells. J Reprod Dev. 2015;61(5):391–7.
Tu F, Pan ZX, Yao Y, et al. miR-34a targets the inhibin beta B gene, promoting granulosa cell apoptosis in the porcine ovary. Genet Mol Res. 2014;13(2):2504–12.
Baufeld A, Vanselow J. Increasing cell plating density mimics an early post-LH stage in cultured bovine granulosa cells. Cell Tissue Res. 2013;354(3):869–80.
Meirow D, Lewis H, Nugent D, Epstein M. Subclinical depletion of primordial follicular reserve in mice treated with cyclophosphamide: clinical importance and proposed accurate investigative tool. Hum Reprod. 1999;14(7):1903–7.
Funding
This research was supported by Natural Science Foundation of Shandong Province (No. ZR2016HM77, No. ZR2016HP05) and a Project of Shandong Province Higher Educational Science and Technology Program (No. J16LK01).
Author information
Authors and Affiliations
Contributions
Qianqian Luo and Ranran Liu contributed equally to the work. All authors read and approved the final version of the manuscript. Qianqian Luo, Ranran Liu, and Hongqin Zhang were in charge of study design and literature research. Qianqian Luo and Li Wang performed the experiments and were in charge of manuscript preparation. Yun Hou and Qianqian Luo were responsible for revision and the final version approval of the manuscript.
Corresponding authors
Ethics declarations
The use of human placentas tissue was permitted to the Institutional Ethics Committee of Binzhou Medical University.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Luo, Q., Liu, R., Wang, L. et al. The Effects of Inhibin B in the Chemotherapy Drug-Induced Premature Ovarian Insufficiency Mice and hPMSCs Treatment. Reprod. Sci. 27, 1148–1155 (2020). https://doi.org/10.1007/s43032-019-00128-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-019-00128-y