Abstract
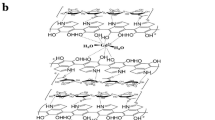
The development of nanoparticles incorporating Gd3+ has attracted interest in the field of contrasting nanomedicine for magnetic resonance (MR) and computed tomography (CT). In order to achieve an effective contrasting performance, the amount and stability of Gd incorporated as well as particle-size control of the nanoparticles should be considered simultaneously. In the current study, Gd-diethylenetriaminepentaacetate(Gd-DTPA) was incorporated into layered double hydroxide (LDH) to meet the physicochemical properties required for MR-CT dual contrasting nanomedicine. Strategically, the particle size and nanometer incorporation of Gd-DTPA into LDH (GL-R hybrid) were controlled homogeneously using the reverse micelle method. X-ray diffraction showed that the hybrid obtained possessed a hydrotalcite phase. Dynamic light scattering and electron microscopic analyses showed that the hybrid had a controlled size of <200 nm with significant homogeneity. Fast Fourier-transform of transmission electron microscopy confirmed that the platelets of the GL-R hybrids were oriented randomly to form inter-particle space, enabling the Gd-DTPA moiety to be encapsulated stably. The encapsulation efficiency of Gd-DTPA was 20.8%, which was sufficiently high compared with other Gd-DTPA-incorporatedLDH. According to X-ray photoelectron spectroscopy and Fourier-transform infrared spectroscopy, Gd-DTPA incorporated in the hybrid preserved its structure intact. Its potential as a dual modal contrast agent was demonstrated by measuring the concentration-dependent Hounsfield unit and magnetic resonance relaxivity, which were determined to be 230 at 2 mg/mL and 5.8 in the range ~0.2–1 mM-Gd, respectively.









Similar content being viewed by others
REFERENCE
Aime, S., & Caravan, P. (2009). Biodistribution of gadolinium-based contrast agents, including gadolinium deposition. Journal of Magnetic Resonance Imaging, 30, 1259–1267.
Alauddin, M. M. (2012). Positron emission tomography (PET) imaging with 18F-based radiotracers. American Journal of Nuclear Medicine and Molecular Imaging, 2, 55–76.
Bloem, J. L., & Wondergem, J. (1989). Gd-DTPA as a contrast agent in CT. Radiology, 171, 578–579.
Caravan, P., Ellison, J. J., McMurry, T. J., & Lauffer, R. B. (1999). Gadolinium(III) chelates as MRI contrast agents: Structure, dynamics, and applications. Chemical Review, 99, 2293–2352.
Choi, S. J., Oh, J. M., & Choy, J. H. (2010). Biocompatible nanoparticles intercalated with anticancer drug for target delivery: Pharmacokinetic and biodistribution study. Journal of Nanoscience and Nanotechnology, 10, 2913–2916.
Choy, J.-H. (2004). Intercalative route to heterostructured nanohybrid. Journal of Physics and Chemistry of Solids, 65, 373–383.
Choy, J.-H., Jung, J.-S., Oh, J.-M., Park, M., Jeong, J., Kang, Y.-K., & Han, O.-J. (2004). Layered double hydroxide as an efficient drug reservoir for folate derivatives. Biomaterials, 25, 3059–3064.
Choy, J.-H., Kim, Y.-K., Son, Y.-H., Choy, Y. B., Oh, J.-M., Jung, H., & Hwang, S.-J. (2008). Nanohybrids of edible dyes intercalated in znal layered double hydroxides. Journal of Physics and Chemistry of Solids, 69, 1547–1551.
Choy, J.-H., Kwak, S.-Y., Jeong, Y.-J., & Park, J.-S. (2000). Inorganic layered double hydroxides as nonviral vectors. Angewandte Chemie International Edition, 39, 4041–4045.
Choy, J.-H., Kwon, Y.-M., Han, K.-S., Song, S.-W., & Chang, S. H. (1998). Intra- and inter-layer structures of layered hydroxy double salts, Ni1–xZn2x(OH)2(CH3CO2)2x·nH2O. Materials Letters, 34, 356–363.
Danaei, M., Dehghankhold, M., Ataei, S., Hasanzadeh Davarani, F., Javanmard, R., Dokhani, A., Khorasani, S., & Mozafari, M. R. (2018). Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics, 10, 57.
Deacon, G. B., & Phillips, R. J. (1980). Relationships between the carbon-oxygen stretching frequencies of carboxylato complexes and the type of carboxylate coordination. Coordination Chemistry Reviews, 33, 227–250.
Gao, L., Zhou, J., Yu, J., Li, Q., Liu, X., Sun, L., et al. (2017). A novel Gd-DTPA-conjugated poly(l-γ-glutamyl-glutamine)-paclitaxel polymeric delivery system for tumor theranostics. Scientific Reports,7, 3799.
Gursky, J. A., Blough, S. D., Luna, C., Gomez, C., Luevano, A. N., & Gardner, E. A. (2006). Particle−particle interactions between layered double hydroxide nanoparticles. Journal of the American Chemical Society, 128, 8376–8377.
Hoshyar, N., Gray, S., Han, H., & Bao, G. (2016). The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine (Lond), 11, 673–692.
Hu, G., Wang, N., O'Hare, D., & Davis, J. (2007). Synthesis of magnesium aluminium layered double hydroxides in reverse microemulsions. Journal of Materials Chemistry, 17, 2257–2266.
Ionescu, E., Li, W., Wiehl, L., Mera, G., & Riedel, R. (2017). Synthesis of nanocrystalline Gd2O2NCN from a versatile single-source precursor. Zeitschrift für anorganische und allgemeine Chemie, 643, 1681–1691.
Kim, H. J., Ryu, K., Kang, J. H., Choi, A. J., Kim, T. I., & Oh, J. M. (2013). Anticancer activity of ferulic acid-inorganic nanohybrids synthesized via two different hybridization routes, reconstruction and exfoliation-reassembly. Scientific World Journal, 2013, 421967.
Kim, S. Y., Oh, J. M., Lee, J. S., Kim, T. J., & Choy, J. H. (2008). Gadolinium (III) diethylenetriamine pentaacetic acid/ layered double hydroxide nanohybrid as novel T1-magnetic resonant nanoparticles. Journal of Nanoscience and Nanotechnology, 8, 5181–5184.
Kim, T.-H., Kim, H., & Oh, J.-M. (2012). Interlayer structure of bioactive molecule, 2-aminoethanesulfonate, intercalated into calcium-containing layered double hydroxides. Journal of Nanoscience and Nanotechnology, 2012, article 21. https://doi.org/10.1155/2012/987938
Kim, T.-H., Lee, G. J., Kang, J.-H., Kim, H.-J., Kim, T.-I., & Oh, J.-M. (2014). Anticancer drug-incorporated layered double hydroxide nanohybrids and their enhanced anticancer therapeutic efficacy in combination cancer treatment. BioMed Research International, 2014, 193401.
Kim, T.-H., Lee, J. Y., Kim, M.-K., Park, J. H., & Oh, J.-M. (2016). Radioisotope Co-57 incorporated layered double hydroxide nanoparticles as a cancer imaging agent. RSC Advances, 6, 48415–48419.
Kriven, W. M., Kwak, S.-Y., Wallig, M. A., & Choy, J.-H. (2004). Bio-resorbable nanoceramics for gene and drug delivery. MRS Bulletin, 29, 33–37.
Li, L., Jiang, W., Luo, K., Song, H., Lan, F., Wu, Y., & Gu, Z. (2013a). Superparamagnetic iron oxide nanoparticles as mri contrast agents for non-invasive stem cell labeling and tracking. Theranostics, 3, 595–615.
Li, Z., Wang, S. X., Sun, Q., Zhao, H. L., Lei, H., Lan, M. B., Cheng, Z. X., Wang, X. L., Dou, S. X., & Lu, G. Q. (2013b). Ultrasmall manganese ferrite nanoparticles as positive contrast agent for magnetic resonance imaging. Advanced Healthcare Materials, 2, 958–964.
Li, B., Gu, Z., Kurniawan, N., Chen, W., & Xu, Z. P. (2017). Manganese-based layered double hydroxide nanoparticles as a T(1) -mri contrast agent with ultrasensitive pH response and high relaxivity. Advanced Materials, 29, 1700373.
Li, X., Liu, Y., Fu, F., Cheng, M., Liu, Y., Yu, L., Wang, W., Wan, Y., & Yuan, Z. (2019). Single nir laser-activated multifunctional nanoparticles for cascaded photothermal and oxygen-independent photodynamic therapy. Nano-Micro Letters, 11, 68.
Lusic, H., & Grinstaff, M. W. (2013). X-ray-computed tomography contrast agents. Chemical Reviews, 113, 1641–1666.
Ma, S., & Liu, Y. (2020). Diagnostic value of fluorine-18 fluorodeoxyglucose positron emission tomography/computed tomography in sublingual and submandibular salivary gland tumors. Molecular and Clinical Oncology, 13, 27.
Meng, D., Cui, X., Bai, C., Yu, Z., Xin, L., Fu, Y., Wang, S., Du, Y., Gao, Z., & Ye, Z. (2019). Application of low-concentration contrast agents and low-tube-voltage computed tomography to chest enhancement examinations: A multicenter prospective study. Science Progress, 103, 0036850419892193.
Musumeci, A., Schiller, T., Xu, Z., Minchin, R., Martin, D., & Smith, S. (2010). Synthesis and characterization of dual radiolabeled layered double hydroxide nanoparticles for use in in vitro and in vivo nanotoxicology studies. The Journal of Physical Chemistry C, 114(2), 734–740.
Na, H., Jung, L., An, K., Park, Y. I., Park, M., Lee, I., Nam, D.-H., Kim, S. T., Kim, S.-H., Kim, S.-W., Lim, K.-H., Kim, K., Kim, S.-O., & Hyeon, T. (2007). Development of at1 contrast agent for magnetic resonance imaging using mno nanoparticles. Angewandte Chemie (International ed. in English), 46, 5397–5401.
Oh, J.-M., Kwak, S.-Y., & Choy, J.-H. (2006a). Intracrystalline structure of DNA molecules stabilized in the layered double hydroxide. Journal of Physics and Chemistry of Solids, 67, 1028–1031.
Oh, J.-M., Park, M., Kim, S.-T., Jung, J.-Y., Kang, Y.-G., & Choy, J.-H. (2006b). Efficient delivery of anticancer drug MTX through MTX-LDH nanohybrid system. Journal of Physics and Chemistry of Solids, 67, 1024–1027.
Oh, J. M., Choi, S. J., Lee, G. E., Kim, J. E., & Choy, J. H. (2009). Inorganic metal hydroxide nanoparticles for targeted cellular uptake through clathrin-mediated endocytosis. Chemistry: An Asian Journal, 4, 67–73.
Pan, Y., Yang, J., Fang, Y., Zheng, J., Song, R., & Yi, C. (2017). One-pot synthesis of gadolinium-doped carbon quantum dots for high-performance multimodal bioimaging. Journal of Materials Chemistry B, 5, 92–101.
Pedrosa, P., & Baptista, P. V. (2015). Chapter 1 - gold and silver nanoparticles for diagnostics of infection. In M. Rai & K. Kon (Eds.), Nanotechnology in diagnosis, treatment and prophylaxis of infectious diseases (pp. 1–18). Academic Press.
Peters, A., Veronesi, B., Calderón-Garcidueñas, L., Gehr, P., Chen, L. C., Geiser, M., Reed, W., Rothen-Rutishauser, B., Schürch, S., & Schulz, H. (2006). Translocation and potential neurological effects of fine and ultrafine particles a critical update. Particle and Fibre Toxicology, 3, 13.
Ribeiro, L. N. M., Breitkreitz, M. C., Guilherme, V. A., da Silva, G. H. R., Couto, V. M., Castro, S. R., de Paula, B. O., Machado, D., & de Paula, E. (2017). Natural lipids-based nlc containing lidocaine: From pre-formulation to in vivo studies. European Journal of Pharmaceutical Sciences, 106, 102–112.
Seltzer, S. E., Shulkin, P. M., Adams, D. F., Davis, M. A., Hoey, G. B., Hopkins, R. M., & Bosworth, M. E. (1984). Usefulness of liposomes carrying losefamate for CT opacification of liver and spleen. American Journal of Roentgenology, 143, 575–579.
Sun Zhou, X.D., Marzke, R., Peng, Z., Szilágyi, I., & Dey, S.K. (2019). Understanding the high longitudinal relaxivity of Gd(DPTA)-intercalated (Zn,Al)-layered double hydroxide nanoparticles. Inorganic Chemistry, 58, 12112–12121.
Ullah, N., Imran, M., Liang, K., Yuan, C.-Z., Zeb, A., Jiang, N., Qazi, U., Sahar, S., & Xu, A.-W. (2017). Highly dispersed ultra-small pd nanoparticles on gadolinium hydroxide nanorods for efficient hydrogenation reactions. Nanoscale, 9, 13800–13807.
Vithanarachchi, S. M., & Allen, M. J. (2012). Strategies for target-specific contrast agents for magnetic resonance imaging. Current molecular imaging, 1, 12–25.
Wattjes, M. P., Steenwijk, M. D., & Stangel, M. (2015). Mri in the diagnosis and monitoring of multiple sclerosis: An update. Clinical Neuroradiology, 25, 157–165.
Xie, W., Guo, Z., Cao, Z., Gao, Q., Wang, D., Boyer, C., Kavallaris, M., Sun, X., Wang, X., Zhao, L., & Gu, Z. (2019). Manganese-based magnetic layered double hydroxide nanoparticle: A pH-sensitive and concurrently enhanced T1/T2-weighteddual-mode magnetic resonance imaging contrast agent. ACS Biomaterials Science & Engineering, 5, 2555–2562.
Xu, C., Akakuru, O. U., Zheng, J., & Wu, A. (2019). Applications of iron oxide-based magnetic nanoparticles in the diagnosis and treatment of bacterial infections. Frontiers in Bioengineering and Biotechnology, 7, 141–141.
Xu, Z. P., Kurniawan, N. D., Bartlett, P. F., & Lu, G. Q. (2007). Enhancement of relaxivity rates of Gd-dtpa complexes by intercalation into layered double hydroxide nanoparticles. Chemistry – A European Journal, 13, 2824–2830.
Yang, J.-H., Han, Y.-S., Park, M., Park, T., Hwang, S.-J., & Choy, J.-H. (2007). New inorganic-based drug delivery system of indole-3-aceticacid-layered metal hydroxide nanohybrids with controlled release rate. Chemistry of Materials, 19, 2679–2685.
Zatsepin, D. A., Boukhvalov, D. W., Zatsepin, A. F., Kuznetsova, Y. A., Mashkovtsev, M. A., Rychkov, V. N., Shur, V. Y., Esin, A. A., & Kurmaev, E. Z. (2018). Electronic structure, charge transfer, and intrinsic luminescence of gadolinium oxide nanoparticles: Experiment and theory. Applied Surface Science, 436, 697–707.
Zhang, J., Ju, X., Wu, Z. Y., Liu, T., Hu, T. D., Xie, Y. N., & Zhang, Z. L. (2001). Structural characteristics of cerium oxide nanocrystals prepared by the microemulsion method. Chemistry of Materials, 13, 4192–4197.
Zhang, Y., Li, B., Wu, B., Yu, H., Song, J., Xiu, Y., & Shi, H. (2020). Diagnostic performance of whole-body bone scintigraphy in combination with spect/ct for detection of bone metastases. Annals of Nuclear Medicine, 34, 549–558.
ACKNOWLEDGMENT
This research was supported by the Dongguk University Research Fund of 2019.
Funding
Funding sources are as stated in the Acknowledgment.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
This paper is based on a presentation made during the 4th Asian Clay Conference, Thailand, June 2020.
Supplementary Information
ESM 1
(PDF 62 kb)
Rights and permissions
About this article
Cite this article
Jung, SY., Park, J.K. & Oh, JM. LAYERED DOUBLE HYDROXIDE-BASED MRI/CT DUAL MODAL CONTRASTING AGENT WITH HOMOGENEOUS PARTICLE SIZE. Clays Clay Miner. 69, 425–433 (2021). https://doi.org/10.1007/s42860-021-00142-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42860-021-00142-9